Abstract
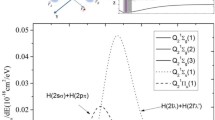
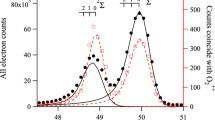
We have studied photodissociation of the A state of the H2S+ ion using the quantum-chemical CAS methods, and the 12 A″ (X 2 B 1) and 14 A″ states are involved in photodissociation of the 12 A′ (A 2 A 1) state (the electronic states in dissociation were studied in the C s symmetry). The CASPT2 S-loss dissociation potential energy curve (PEC) calculations indicate that the 12 A″ and 12 A′ states correlate with the second limit [H2 + S+(2 D)] while the 14 A″ state correlates with the first limit [H2 + S+(4S)] and that there are a transition state and a local minimum along the 12 A′ PEC and the repulsive 14 A″ PEC crosses the 12 A″ and 12 A′ PECs. The CASPT2 H-loss dissociation PEC calculations indicate that the 12 A″ and 14 A″ states correlate with the first limit [HS+(X 3Σ−) + H] while the 12 A′ state correlates with the second limit [HS+(a 1Δ) + H] and that the repulsive 14 A″ PEC crosses the 12 A′ PEC. For the crossing doublet and quartet states in pairs, we performed CASSCF minimum energy crossing point (MECP) calculations, and the CASSCF spin-orbit couplings and CASPT2 energies at the MECP geometries were calculated. We examined the two previously proposed mechanisms (mechanisms I and II) for dissociation of the A state to the S+ ion, based on our calculation results. We suggest processes for dissociation of the A state to the S+ ion (processes I and II, based on mechanisms I and II, respectively) and to the SH+ ion (process III) and conclude that photodissociation of the A state mainly leads to the S+ ion via the most energetically favorable process II: A 2 A 1 (12 A′) (2.38 eV) → barrier at the linearity (2.96 eV) → X 2 B 1 (12 A″) (0.0 eV) → the 12 A″/14 A″ MECP (3.50 eV, large spin-orbit coupling) → H2 \( (X^{ 1} \Upsigma_{\text{g}}^{ + } ) \) + S+(4S) (2.92 eV) (the CASPT2 relative energy values to X 2 B 1 are given in parentheses and the largest value is 3.50 eV at the MECP).





Similar content being viewed by others
References
Hirst DM (2003) J Chem Phys 118:9175–9184. doi:10.1063/1.1568080
Hirsch G, Bruna PJ (1980) Int J Mass Spectrom Ion Phys 36:37–46. doi:10.1016/0020-7381(80)80005-2
Li WZ, Huang MB (2005) Chem Phys 315:133–141. doi:10.1016/j.chemphys.2005.04.005
Webb AD, Dixon RN, Ashfold MNR (2007) J Chem Phys 127:224307. doi:10.1063/1.2800559
Webb AD, Kawanaka N, Dixon RN, Ashfold MNR (2007) J Chem Phys 127:224308. doi:10.1063/1.2800565
Dibeler VH, Rosenstock HM (1963) J Chem Phys 39:3106–3111. doi:10.1063/1.1734150
Dibeler VH, Liston SK (1968) J Chem Phys 49:482–485. doi:10.1063/1.1670100
Eland JHD (1979) Int J Mass Spectrom Ion Phys 31:161–173. doi:10.1016/0020-7381(79)80115-1
Möhlmann GR, Deheer FJ (1975) Chem Phys Lett 15:353–356. doi:10.1016/0009-2614(75)80254-5
Dixon RN, Duxbury G, Horani M, Rostas J (1971) Mol Phys 22:977–992. doi:10.1080/00268977100103311
Edwards CP, Maclean CS, Sarre PJ (1982) Chem Phys Lett 87:11–13. doi:10.1016/0009-2614(82)83542-2
Prest HF, Tzeng WB, Brom JM Jr, Ng CY (1983) Int J Mass Spectrom Ion Phys 50:315–329. doi:10.1016/0020-7381(83)87008-9
Jarrold MF, Illies AJ, Bowers MT (1982) Chem Phys 65:19–28. doi:10.1016/0301-0104(82)85052-0
Ibuki T (1984) J Chem Phys 81:2915–2918. doi:10.1063/1.448038
Tokue I, Yamasaki K, Nanbu S (2003) J Chem Phys 119:5882–5888. doi:10.1063/1.1602064
Carney TE, Baer T (1981) J Chem Phys 75:4422–4429. doi:10.1063/1.442607
Fiquet-Fayard F, Guyon PM (1966) Mol Phys 11:17–30. doi:10.1080/00268976600100811
Feng RF, Cooper G, Brion CE (1999) Chem Phys 249:223–236. doi:10.1016/S0301-0104(99)00232-3
Hochlaf M, Weitzel KM (2004) J Chem Phys 120:6944–6956. doi:10.1063/1.1669386
Tokue I, Yamasaki K, Nanbu S (2003) J Chem Phys 119:5874–5881. doi:10.1063/1.1602063
Roos BO (1987) Adv Chem Phys 69:399–445. doi:10.1002/9780470142943.ch7
Andersson K, Roos BO (1993) Int J Quantum Chem 45:591–607. doi:10.1002/qua.560450610
Andersson K, Malmqvist P, Roos BO (1995) In: Yarkony DR (ed) Modern electronic structure theory. World Scientific, Singapore, vol 1, p 55
Chang HB, Chen BZ, Huang MB (2008) J Phys Chem A 112:1688–1693. doi:10.1021/jp710633s
Andersson K et al (2005) MOLCAS (version 6.2). University of Lund, Sweden
Almlof J, Taylor PR (1987) J Chem Phys 86:4070–4077. doi:10.1063/1.451917
Widmark PO, Malmqvist PA, Roos BO (1990) Theor Chim Acta 77:291–306. doi:10.1007/BF01120130
Widmark PO, Persson BJ, Roos BO (1991) Theor Chim Acta 79:419–432. doi:10.1007/BF01112569
Huber KP, Herzberg G (1979) Molecular spectra and molecular structure IV. Constants of diatomic molecules. Van Nostrand Reinhold Co., New York
Dunlavey SJ, Dyke JM, Fayad NK, Jonathan N, Morris A (1979) Mol Phys 38:729. doi:10.1080/00268977900102001
Turner DW, Baker C, Baker AD, Brundle CR (1970) Molecular photoelectron spectroscopy. Wiley, London
Duxbury G, Horani M, Rostas J (1972) Proc R Soc Lond A 331:109
Duxbury G, Jungen C, Rostas J (1983) Mol Phys 48:719. doi:10.1080/00268978300100541
Potts AW, Price WC (1972) Proc R Soc A 326:181–197. doi:10.1098/rspa.1972.0004
Acknowledgment
We appreciate the financial support of this work that was provided by National Natural Science Foundation of China through Contract No. 20773161.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chang, HB., Huang, MB. A theoretical study on photodissociation of the A state of the H2S+ ion. Theor Chem Account 122, 189–196 (2009). https://doi.org/10.1007/s00214-008-0498-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-008-0498-4