Abstract
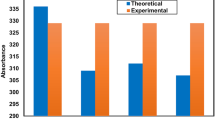
Equilibrium ground state geometry configurations and their relevant electronic properties of four experimentally reported asymmetric spirosilabifluorene derivatives are calculated by the HF(DFT)/6-31G(d) method. Their excited state geometries are investigated using the CIS/6-31G(d) method. The absorption and emission spectra are evaluated using the TD-B3LYP/6-31G(d) and TD-PBE0/6-31+G(d) levels both in gas phase and CHCl3 solvent. Our results show an excellent agreement with the experimental data on their optical properties. To predict the substitution effect, the H/R (R = –NO2, –CN, –NH2 and –OCH3) substituted symmetric and asymmetric spirosilabifluorene derivatives are also investigated, and the optical properties of H/R substituted derivatives are predicted in gas phase and CHCl3 solvent. In comparison with the parent compound, significant red-shift is predicted for the emission spectra of the di-substituted symmetric derivatives with –NH2 (96 nm), –OCH3 (61 nm) and the push–pull (containing both –NH2 and –NO2) derivative (56 nm). It is found that the performance and the optical properties of these derivatives can be improved by adding push–pull substitutents. The largest change in the electronic and optical properties of this system can be obtained upon symmetric di-substitution among mono-, di-, tri- and tetra-substitutions.
Similar content being viewed by others
References
Tang CW, Van Slyke SA (1987) Appl Phys Lett 51: 913
Kulkarni AP, Tonzola CJ, Babel A, Jenekhe SA (2004) Chem Mater 16: 4556
Lee SK, Hwang DH, Jung JB, Cho NS, Lee J, Lee JD, Shim HK (2005) Adv Funct Mater 15: 1647
Liu J, Zhou QG, Cheng YX, Geng YH, Wang LX, Ma DG, Jing X B, Wang FS (2005) Adv Mater 17: 2974
Tu GL, Mei CY, Zhou QG, Cheng YX, Geng YH, Wang LX, Ma DG, Jing XB, Wang FS (2006) Adv Funct Mater 16: 101
Liu J, Zhou QG, Cheng YX, Geng YH, Wang LX, Ma DG, Jing XB, Wang FS (2006) Adv Func Mater 16: 957
Yang R, Wu H, Cao Y, Ba ZG (2006) J Am Chem Soc 128: 14422
Jiang J, Xu Y, Yang W, Guan R, Liu Z, Zhen H, Cao Y (2006) Adv Mater 18: 1769
Wong KT, Chien YY, Chen RT, Wang CF, Lin YT, Chiang HH, Hsieh PY, Wu CC, Chou CH, Su YO, Lee GH, Peng SM (2002) J Am Chem Soc 124: 11576
Steuber F, Staudigel J, Stössel M, Simmerer J, Winnacker A, Spreitzer H, Weissörtel F, Salbeck J (2000) Adv Mater 12: 130
Wu CC, Lin YT, Chiang HH, Cho TY, Chen CW, Wong KT, Liao YL, Lee GH, Peng SM (2002) Appl Phys Lett 81: 577
Schneider D, Rabe T, Riedl T, Dobbertin T, Werner O, Kröger M, Becker E, Johannes HH, Kowalsky W, Weimann T, Wang J, Hinze P, Gerhard A, Stössel P, Vestweber H (2004) Appl Phys Lett 84: 4693
Saragi TPI, Pudzich R, Fuhrmann T, Salbeck J (2004) Appl Phys Lett 84: 2334
Bach U, Lupo D, Comte P, Moser JE, Weissörtel F, Salbeck J, Spreitzer H, Grätzel M (1998) Nature 395: 583
Cabanillas-Gonzalez J, Yeates S, Bradley DDC (2003) Synth Met 139: 637
Kim SY, Lee M, Boo BH (1998) J Chem Phys 109: 2593
Tian H, Chen B, Liu PH (2001) Chem Lett 30: 990
Ghosh P, Shabat D, Kumar S, Sinha SC, Grynszpan F, Li J, Noodleman L, Keinan E (1996) Nature 382: 25
Murata H, Kafafi ZH, Uchida M (2002) Appl Phys Lett 80: 189
Palilis LC, Murata H, Uchida M, Kafafi ZH (2003) Organic Electronics 4: 113
Ohshita J, Lee KH, Hamamoto D, Kunugi Y, Ikadai J, Kwak YW (2004) Chem Lett 33: 892
Mitschke U, Bauerle P (2001) J Chem Soc Perkin Trans 1: 740
Wu RL, Schumm JS, Pearson DL, Tour JM (1996) J Org Chem 61: 6906
Xiao HB, Leng B, Tian H (2005) Polymer 46: 5707
Pei J, Ni J, Zhou XH, Cao XY, Lai YH (2002) J Org Chem 67: 4924
Tamao K, Uchida M, Izumizawa T, Furukawa K, Yamaguchi S (1996) J Am Chem Soc 118: 11974
Yamaguchi S, Endo T, Uchida M, Izumizawa T, Furukawa K, Tamao K (2000) Chem Eur J 6: 1683
Watkins NJ, Mdkinen AJ, Gao Y, Uchida M, Kafafi ZH (2004) Proc SPIE Int Soc Opt Engr 5214: 368
Yin SW, Yi YP, Li QX, Yu G, Liu YQ, Shuai ZG (2006) J Phys Chem A 110: 7138
Murata H, Malliaras GG, Uchida M, Shen Y, Kafafi ZH (2001) Chem Phys Lett 339: 161
Maslak P, Chopra A, Moylan CR, Wortmann R, Lebus S, Rheingold AL (1996) J Am Chem Soc 118: 1471
Wu CC, Liu TL, Hung WY, Lin YT, Wong KT, Chen RT, Chen YM, Chien YY (2003) J Am Chem Soc 125: 3710
Xiao HB, Shen H, Lin YG, Su JH, Tian H (2007) Dyes Pigments 73: 224
Lee SH, Jang BB, Kafafi ZH (2005) J Am Chem Soc 127: 9071
Lukeš V, Pálszegir T, Milota F, Sperling J, Kauffmann HF (2006) J Phys Chem A 110: 1775
Yang GC, Su ZM, Qin CS (2006) J Phys Chem A 110: 4817
Sun XB, Liu Y, Xu XJ, Yang CH, Yu G, Zhu DB (2005) J Phys Chem B 109: 10786
Belletête M, Blouin N, Boudreault PT, Leclerc M, Durocher G (2006) J Phys Chem A 110: 13696
Ortí E, Viruela PM, Viruela R, Effenberger F (2005) J Phys Chem A 109: 8724
Jacquemin D, Preat J, Wathelet V, Michèle F, Perpète EA (2006) J Am Chem Soc 128: 2072
Belletête M, Morin JF, Leclerc M, Durocher G (2005) J Phys Chem A 109: 6953
Wu C, Tretiak S, Chernyak VY (2007) Chem Phys Lett 433: 305
Masunov A, Tretiak S (2004) J Chem Phys B 108: 899
Hariharan PC, Pople JA (1974) Mol Phys 27: 209
Gordon MS (1980) Chem Phys Lett 76: 163
Frisch MJ, Pople JA, Binkley JS (1984) J Chem Phys 80: 3265
Foresman JB, Head-Gordon M, Pople JA, Frisch MJ (1992) J Phys Chem 96: 135
Becke AB (1993) J Chem Phys 98: 5648
Lee C, Yang W, Parr RG (1988) Phys Rev B 37: 785
Stephens PJ, Devlin FJ, Chabalowski CF, Frisch M (1994) J Phys Chem 98: 11623
Foresman JB, Head-Gordon M, Pople JA, Frish MJ (1992) J Phys Chem 96: 135
Stratman RE, Scuseria GE, Frisch MJ (1998) J Chem Phys 109: 8218
Bacon AD, Zerner MC (1979) Theor Chim Acta 53: 21
Adamo C, Barone V (1999) J Chem Phys 110: 6158
Clark T, Chandrasekhar J, Spitznagel GW, Schleyer PvR (1983) J Comp Chem 4: 294
Cancès E, Mennucci B, Tomasi J (1997) J Chem Phys 107: 3032
Cornard JP, Lapouge C (2006) J Phys Chem A 110: 7159
Cossi M, Barone V (2001) J Chem Phys 115: 4708
Becke AD (1993) J Chem Phys 98: 1372
Gaussian 03, Revision B.03 (2003) Gaussian, Pittsburgh
Gaussview 3.0 Reference (2003) Gaussian, Wallingford
Stephan Portmann (2002) CSCS/ETHZ http://www.cscs.ch/molekel
Marcos AD, Duarte HA, Pernaut JM, Wagner BD (2000) J Phys Chem A 104: 8256
Halls MD, Schlegel HB (2001) Chem Mater 13: 2632
Foresman JB, Schlegel HB (1933) In: Gausto R, Hollas JM (eds) Recent experimental and computational advances in molecular spectroscopy, vol 406. Kluwer, Dordrecht, p 11
Tirapattur S, Belletête M, Leclerc M, Durocher G (2003) TheoChem 625: 141
Zhang JP, Frenking G (2004) J Phys Chem A 108: 10269
Hu B, Gahungu G, Zhang JP (2007) J Phys Chem A 111: 4965
Magyar RJ, Tretiak S (2007) J Chem Theory Comput 3: 976
Reed AE, Weinhold F (1983) J Chem Phys 78: 4066
Mulliken RS (1955) J Chem Phys 23: 1833
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link for the Electronic supplementary material
Rights and permissions
About this article
Cite this article
Sun, M., Niu, B. & Zhang, J. Theoretical design of blue emitting materials based on symmetric and asymmetric spirosilabifluorene derivatives. Theor Chem Account 119, 489–500 (2008). https://doi.org/10.1007/s00214-008-0410-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-008-0410-2