Abstract
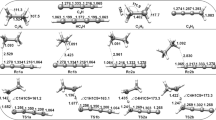
The multiple-channel reactions OH + SiH(CH3)3 → products (R1) and the single-channel reaction OH + Si(CH3)4 → Si(CH3)3CH2 + H2O (R2) have been studied by means of the direct dynamics method at the BMC-CCSD//MP2/6-311+G(2d,2p) level. The optimized geometries, frequencies and minimum energy path are all obtained at the MP2/6-311+G(2d,2p) levels, and energy information is further refined by the BMC-CCSD (single-point) level. The rate constants for every reaction channels are calculated by canonical variational transition states theory (CVT) with small-curvature tunneling (SCT) contributions over the temperature range 200–2,000 K. The theoretical total rate constants are in good agreement with the available experimental data, and the three-parameter expression k 1 = 2.53×10−21 T 3.14 exp(1, 352.86/T), k 2 = 6.00 × 10−19 T 2.54 exp(−106.11/T) (in unit of cm3 molecule−1 s−1) over the temperature range 200–2,000 K are given. Our calculations indicate that at the low temperature range, for reaction R1, H-abstraction is favored for the SiH group, while the abstraction from the CH3 group is a minor channel.
Similar content being viewed by others
References
Calvert JG, Atkinson R, Kerr JA, Madronich S, Moortgat GK, Wallington TJ and Yarwood G (2000). The mechanisms of atmospheric oxidation of the alkenes. Oxford, New York
Calvert JG, Atkinson R, Becker KH, Kamens RH, Seinfeld JH, Wallington TJ and Yarwood G (2002). The mechanisms of atmospheric oxidation of the aromatic hydrocarbons. Oxford, New York
Goumri A, Yuan J, Hommel EL and Marshall P (2003). Chem Phys Lett 375: 149
Atkinson R (1991). Environ Sci Technol 25: 863
Sommerlade R, Parlar H, Wrobel D and Kochs P (1993). Environ Sci Technol 27: 2435
Tuazon EC, Aschmann SM and Atkinson R (2000). Environ Sci Technol 34: 1970
Truhlar DG (1995). Direct dynamics method for the calculation of reaction rates. In: Heidrich, D (eds) The reaction path in chemistry: current approaches and perspectives, pp 229. Kluwer, Dordrecht
Truhlar DG, Garrett BC and Klippenstein SJ (1996). J Phys Chem 100: 12771
Hu WP and Truhlar DG (1996). J Am Chem Soc 118: 860
Corchado JC, Espinosa-Garcia J, Hu W-P, Rossi L and Truhlar DG (1995). J Phys Chem 99: 687
Lynch BJ, Zhao Y and Truhlar DG (2005). J Phys Chem A 109: 1643
Corchado JC, Chuang Y-Y, Fast PL, Villa J, Hu W-P, Liu Y-P, Lynch GC, Nguyen KA, Jackels CF, Melissas VS, Lynch BJ, Rossi I, Coitino EL, Ramos AF, Pu J and Albu TV (2002). POLYRATE version 9.1. Department of Chemistry and Supercomputer Institute. University of Minnesota, Minneapolis
Truhlar DG and Garrett BC (1980). Acc Chem Res 13: 440
Truhlar DG, Isaacson AD, Garrett BC (1985). In: Baer M (eds) The theory of chemical reaction dynamics, vol 4, CRC P, Boca Raton, p. 65
Duncan WT and Truong TN (1995). J Chem Phys 103: 9642
Frisch MJ, Head-Gordon M and Pople JA (1990). Chem Phys Lett 166: 275
Head-Gordon M, Pople JA and Frisch MJ (1988). Chem Phys Lett 153: 503
Boys SF and Bernardi F (1970). Mol Phys 19: 553
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA and Montgomery JA Jr (2003). Gaussian 03. Gaussian, Inc., Pittsburgh
Steckler R, Hu W-P, Liu Y-P, Lynch GC, Garrett BC, Isaacson AD, Melissas VS, Lu D-P, Troung TN, Rai SN, Hancock GC, Lauderdale JG, Joseph T and Truhlar DG (1995). Comput Phys Commun 88: 341
Lu DH, Truong TN, Melissas VS, Lynch GC, Liu YP, Grarrett BC, Steckler R, Issacson AD, Rai SN, Hancock GC, Lauderdale JG, Joseph T and Truhlar DG (1992). Comput Phys Commun 71: 235
Liu Y-P, Lynch GC, Truong TN, Lu D-H, Truhlar DG and Garrett BC (1993). J Am Chem Soc 115: 2408
Truhlar DG (1991). J Comput Chem 12: 266
Chuang YY and Truhlar DG (2000). J Chem Phys 112: 1221
Huber KP, Herzberg G (2005) NIST chemistry webbook, nist standard reference database number 69, June 2005 release constants of diatomic molecules date
Kuchitsu K (1998) In: Structure of free polyatomic molecules basic data. Springer, Berlin, p 58
Hammond GS (1955). J Am Chem Soc 77: 334
Shimanouchi T (1972) Tables of molecular vibrational frequencies consolidated volume I. National Bureau of Standards, US GPO, Washington, DC
Coblentz Society, Inc (2005) NIST chemistry webbook, NIST standard reference database number 69, June 2005 release, Vibrational frequency date
Shimanouchi T (2005) NIST chemistry webbook, NIST standard reference database number 69, June 2005 release, Vibrational frequency date
Walsh R (1992) In: Martinho Simões JA (eds) Energetica of organometallic species; NATO-ASI series C, 367 Kluwer, Dordrecht Chapter 11
Chase MW Jr (1998). NIST-JANAF themochemical tables, 4th edn. J Phys Chem Ref Data Monogr 9: 1–1951
Ding L and Marshall P (1992). J Am Chem Soc 114: 5754
Kalinovski IJ, Gutman D, Krasnoperov LN, Goumri A, Yuan W-J and Marshall P (1994). J Phys Chem 98: 9551
Goumri A, Yuan W-J and Marshall P (1993). J Am Chem Soc 115: 2539
Ellul R, Potzinger P, Reimann B and Camilleri P (1981). Ber Bunsen-Ges Phys Chem 85: 407
Doncaster AM and Walsh R (1979). J Chem Soc, Faraday Trans 1(75): 1126
Zhang QZ, Wang SK and Gu YS (2002). J Phys Chem A 106: 3796
Taghikhani M and Parsafar GA (2005). J Phys Chem A 109: 8158
Ji YM, Wang L, Li ZS, Liu JY and Sun CC (2006). Chem Phys Chem 7: 1741
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Zhang, H., Zhang, GL., Wang, Y. et al. Theoretical studies on the reactions of hydroxyl radicals with trimethylsilane and tetramethylsilane. Theor Chem Account 119, 319–327 (2008). https://doi.org/10.1007/s00214-007-0387-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-007-0387-2