Abstract
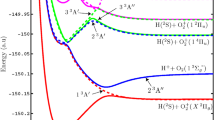
Diabatic potential energy surfaces are a convenient starting point for dynamics calculations of photochemical processes, and they can be calculated by the fourfold way direct diabatization scheme. Here we present an improved definition of the reference orbital for applying the fourfold way direct diabatization scheme to ammonia. The improved reference orbital is a geometry-dependent hybrid orbital that allows one to define consistent dominant configuration lists at all geometries important for photodissociation. Using diabatic energies calculated with the new reference orbital and consistent dominant configuration lists, we have refitted the analytical representations of the ground and the first electronically excited singlet-state potential energy surfaces and the diabatic coupling surface. Improved functional forms were used to reproduce the experimental dissociation energies and excitation energies, which will be important for subsequent simulations of photochemical dynamics. We find that the lowest-energy conical intersection point is at 5.16 eV, with C 2v symmetry.
Article PDF
Similar content being viewed by others
Avoid common mistakes on your manuscript.
References
Nangia S, Truhlar DG (2006). J Chem Phys 124:124309
Nakamura H, Truhlar DG (2001). J Chem Phys 115:10353
Nakamura H, Truhlar DG (2002). J Chem Phys 117:5576
Nakamura H, Truhlar DG (2003). J Chem Phys 118:6816
Atchity GJ, Ruedenberg K (1993). J Chem Phys 99:3790
Ruedenberg K, Atchity GJ (1993). J Chem Phys 99:3799
Nakano H (1993). J Chem Phys 99:7983
Nakano H (1993). Chem Phys Lett 207:372
Nakano H, Nakajima T, Tsuneda T, Hirao K (2001). J Mol Struct 573:91
Ruedenberg K, Sundberg R (1976) In: Calais, J-L, Goscinski O, Linderberg J, Öhrn Y (Eds) Quantum Science. Plenum, New York, p 505
Roos, BO (1987). Adv Chem Phys 69:399
Witek HA, Choe Y-K, Finley JP, Hirao K (2002). J Comput Chem 10:957
Krishnan R, Binkley JS, Seeger R, Pople JA (1980). J Chem Phys 72:650
Frisch MJ, Pople JA, Binkley JS (1984). J Chem Phys 80:3265
Nakamura H, Xidos JD, Chamberlin AC, Kelly CP, Valero R, Thompson JD , Li J, Hawkins GD, Zhu T, Lynch BJ, Volobuev Y, Rinaldi D, Liotard DA, Cramer CJ, Truhlar DG (2007). HONDOPLUS v5.1, based on HONDO-v99.6. University of Minnesota, Minneapolis
Dupuis M, Marquez A, Davidson ER (1999) HONDO 99.6, based on Dupuis M, Marquez A, Davidson ER, HONDO 95.3, Quantum Chemistry Program Exchange, Indiana University, Bloomington, IN 47405
Schmidt MW, Baldridge KK, Boatz JA, Elbert ST, Gordon MS, Jensen JH, Koseki S, Matsunaga N, Nguyen KA, Su SJ, Windus TL, Dupuis M, Montgomery JA (1993). J Comput Chem 14:1347
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery Jr. JA, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2004) Gaussian 03, Revision D.01, Gaussian, Inc., Wallingford CT
Léonard C, Handy NC, Carter S, Bowman JM (2002). Spectrochim Acta Part A 58:825
Landolt-Börnstein (1976) Numerical Data and Function Relationships in Science and Technology II(7,2). Springer, Heidelberg. Cited in Pesonen J, Miani A, Halonen L (2001) J Chem Phys 115:1243
Coy SL, Lehmann KK (1989). Spectrochim Acta Part A 45:47
Martin JML, Lee TJ, Taylor PR (1992). J Chem Phys 97:8361
Handy NC, Carter S, Colwell SM (1999). Mol Phys 96:477
Mordaunt DH, Dixon RN, Ashfold MNR (1996). J Chem Phys 104:6472
Henck SA, Mason MA, Yan WB, Lehmann KK, Coy SL (1995). J Chem Phys 102:4772
Bach A, Hutchison JM, Holiday RJ, Crim FF (2002). J Chem Phys 116:9315
Morino I, Kawaguchi KJ (1997). Mol Spectrosc 182:428
Gabriel W, Chambaud G, Rosmus P, Carter S, Handy NC (1994). Mol Phys 81:1445
Jacox ME (1994) J Phys Chem Ref Data Monograph 3
Herzberg G (1966) Molecular Spectra and Molecular structure, vol. 3. Van Nostrand Reinhold, New York. Cited in McCarthy MI, Rosmus P, Werner H-J, Botschwina P, Vaida V (1987) J Chem Phys 86:6693
As reported in http://webbook.nist.gov/chemistry
Ruscic B, Boggs JE, Burcat A, Császár AG, Demaison J, Janoschek R, Martin JML, Morton ML, Rossi MJ, Stanton JF, Szalay PG, Westmoreland PR, Zabel F, Bérces T (2005) J Phys Chem Ref Data 34:573 and references therein
Hargiss LO, Ermler WC (1988). J Phys Chem 92:300
Gatti F, Lung C, Leforestier C, Chapuisat X (1999). J Chem Phys 111:7236
Demaison J, Margulès L, Boggs JE (2003). Phys Chem Chem Phys 5:3359
Barone V (2004). J Chem Phys 120:3059
Douglas AE (1963). Discuss Faraday Chem Soc 35:158
Dixon RA (1996). Mol Phys 88:949
Duchovic RJ, Volobuev YL, Lynch GC, Truhlar DG, Allison TC, Wagner F, Garrett BC, Corchado JC (2002) Comput Phys Commun 144:169
Duchovic RJ, Volobuev Y L, Lynch GC, Truhlar DG, Allison TC, Wagner F, Garrett BC, Corchado JC (2004) 156:319(E)
Duchovic RJ, Volobuev YL, Jasper AW, Lynch GC, Truhlar DG, Allison TC, Wagner F, Garrett BC, Espinosa-García J, Corchado JC POTLIB-online. http://comp.chem.umn.edu/ potlib
Yarkony DR (2004). J Chem Phys 121:628
Bach A, Hutchison JM, Holiday RJ, Crim FF (2003). J Phys Chem A 107:10490
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the special issue dedicated to the memory of the late Professor Fernando Bernardi.
Electronic supplementary material
Below is the electronic supplementary material.
Rights and permissions
Open Access This is an open access article distributed under the terms of the Creative Commons Attribution Noncommercial License ( https://creativecommons.org/licenses/by-nc/2.0 ), which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
About this article
Cite this article
Li, Z.H., Valero, R. & Truhlar, D.G. Improved direct diabatization and coupled potential energy surfaces for the photodissociation of ammonia. Theor Chem Account 118, 9–24 (2007). https://doi.org/10.1007/s00214-006-0237-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-006-0237-7