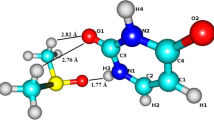
Abstract.
A very diffuse, but spatially confined, electron trapped in a dipole–bound state of a polar molecule provides an excellent target for testing the interaction of a localized electron positioned outside the molecular frame of its host molecule with other atomic or molecular systems. In this work we use ab initio calculations to investigate systems where a dipole–bound electron attached to a uracil molecule is interacting with an N2 molecule and an Ar atom. Neither of the two systems forms a stable anion and in the aducts they form with the dipole–bound electron the electron becomes suspended between the uracil molecule and Ar or N2. Calculations are performed to determine the vertical electron detachment energies of these anions and to determine the molecular rearrangements occurring when the excess electron is removed from them.
Similar content being viewed by others
Acknowledgments.
ensp;We dedicate this work to Prof. Jacopo Tomasi. Through his numerous important contributions to the field of quantum chemistry he has inspired many works of others, including ours. He has been one of the pioneers of quantum chemistry and owing to his dedication and intellectual input to the field it has grown to become one of the leading research branches of chemistry.
Author information
Authors and Affiliations
Corresponding author
Additional information
Contribution to the Jacopo Tomasi Honorary Issue
Rights and permissions
About this article
Cite this article
Jalbout, A., Pichugin, K. & Adamowicz, L. Interaction of the uracil dipole-bound electron with closed-shell systems (Ar and N2). Theor Chem Acc 111, 358–362 (2004). https://doi.org/10.1007/s00214-003-0510-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00214-003-0510-y