Abstract
Rationale
It is well known that the anterior cingulate cortex (ACC) plays an important role in acute pain perception.
Objectives
In the present study, we aimed to investigate the possible involvement of the ACC dopamine D1 and D2 receptors in nicotine plus morphine-induced analgesia.
Methods
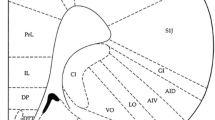
The ACC’s of adult male Wistar rats were bilaterally cannulated by stereotaxic instrument and the tail-flick test was used to measure the thermal pain threshold.
Results
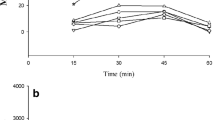
The results indicated that subcutaneous (s.c.) injection of nicotine (0.3 mg/kg) potentiated the analgesic response of intraperitoneal (i.p.) administration of morphine (3 mg/kg). Systemic administration of the same doses of nicotine or morphine alone had no effect on tail-flick latency. Intra-ACC administration of apomorphine (0.3–0.9 μg/rat), the non-selective D1/D2 receptors agonist, plus ineffective doses of nicotine (0.1 mg/kg, s.c.) plus morphine (3 mg/kg, i.p) induced analgesia in rats. In addition, the analgesia induced with co-administration of nicotine and morphine was inhibited via intra-ACC administration of SCH23390 (0.5–1 μg/rat) or sulpiride (0.5–2 μg/rat), the selective antagonists of D1 or D2 receptors, respectively. The intra-ACC microinjection of the same doses of drugs alone had no effect on tail-flick latency. Cubic interpolation analysis also confirmed that activation or inactivation of the ACC D1 and D2 receptors by different doses of drugs can modulate the nicotine-morphine analgesic response.
Conclusions
The findings suggest that the ACC has an important role in acute thermal pain perception and modulates the analgesia induced by nicotine plus morphine via dopaminergic receptors.





Similar content being viewed by others
Abbreviations
- ACC:
-
Anterior cingulate cortex
- ANOVA:
-
Analysis of variance
- AUC:
-
Area under the curve
- CNS:
-
Central nervous system
- i.p.:
-
Intraperitoneal
- MPE:
-
Maximum possible effect
- nAchRs:
-
Nicotinic acetylcholine receptors
- s.c:
-
Subcutaneous
- SCH23390:
-
R( +)-7-chloro-8-hydroxy-3 methyl-1-phenyl-3,4,5-tetrahydro-1H-3-benzazepine hydrochloride
- S.E.M.:
-
Standard error of mean
- TFL:
-
Tail-flick latency
- VTA:
-
Ventral tegmental area
References
Almeida RA, Lauretti GR, Mattos AL (2003) Antinociceptive effect of low-dose intrathecal neostigmine combined with intrathecal morphine following gynecologic surgery. Anesthesiology 98:495–498. https://doi.org/10.1097/00000542-200302000-00031
Bannon AW, Decker MW, Holladay MW, Curzon P, Donnelly-Roberts D, Puttfarcken PS, Bitner RS, Diaz A, Dickenson AH, Porsolt RD, Williams M, Arneric SP (1998) Broad-spectrum, non-opioid analgesic activity by selective modulation of neuronal nicotinic acetylcholine receptors. Science 279:77–81. https://doi.org/10.1126/science.279.5347.77
Bauch EM, Andreou Ch, Rausch VH (2017) Bunzeck N (2017) Neural habituation to painful stimuli is modulated by dopamine: evidence from a pharmacological fMRI study. Front Hum Neurosci 11:630. https://doi.org/10.3389/fnhum.2017.00630
Berrendero F, Kieffer BL, Maldonado R (2002) Attenuation of nicotine induced antinociception, rewarding effects, and dependence in μ-opioid receptor knock-out mice. J Neurosci 22(24):10935–10940. https://doi.org/10.1523/JNEUROSCI.22-24-10935.2002
Cepeda-Benito A, Reynoso J, McDaniel EH (1998) Associative tolerance to nicotine analgesia in the rat: Tail-flick and hot-plate tests. Exp Clin Psychopharmacol 6:248–254. https://doi.org/10.1037//1064-1297.6.3.248
Dai WL, Xiong F, Yan B, Cao ZY, Liu WT, Liu JH, Yu BY (2016) Blockade of neuronal dopamine D2 receptor attenuates morphine tolerance in mice spinal cord. Sci Rep 6:38746. https://doi.org/10.1038/srep38746
Davenport KE, Houdi AA, Van Loon GR (1990) Nicotine protects against -opioid receptor antagonism by -funaltrexamine: evidence for nicotine-induced release of endogenous opioids in brain. Neurosci Lett 113:40–46. https://doi.org/10.1016/0304-3940(90)90491-Q
Dhatt RK, Gudehithlu KP, Wemlinger TA, Tejwani GA, Neff NH et al (1995) Preproenkephalin mRNA and methionine-enkephalin content are increased in mouse striatum after treatment with nicotine. J Neurochem 64:1878–1883. https://doi.org/10.1046/j.1471-4159.1995.64041878.x
Gaspar P, Bloch B, Le Moine C (1995) D1 and D2 receptor gene expression in the rat frontal cortex: cellular localization in different classes of efferent neurons. Eur J Neurosci 7(5):1050–1063. https://doi.org/10.1111/j.1460-9568.1995.tb01092.x
Hadjiconstantinou M, Neff NH (2011) Nicotine and endogenous opioids: neurochemical and pharmacologicalevidence. Neuropharmacology 60:120920. https://doi.org/10.1016/j.neuropharm.2010.11.010
Hall BJ, Slade S, Allenby C, Kutlu MG, Levin ED (2015) Neuro-anatomic mapping of dopamine D1 receptor involvement in nicotine self-administration in rats. Neuropharmacology 99:689–695. https://doi.org/10.1016/j.neuropharm.2015.03.005
Hnasko TS, Sotak BN, Palmiter RD (2005) Morphine reward in dopamine-deficient mice. Nature 438:854–857. https://doi.org/10.1038/nature04172
Jasinska AJ, Zorick T, Brody AL, Stein EA (2014) Dual role of nicotine in addiction and cognition: a review of neuroimaging studies in humans. Neuropharmacology 84:111–122. https://doi.org/10.1016/j.neuropharm.2013.02.015
Karna SR, Kongara K, Singh PM, Chambers P, Lopez-Villalobos N (2019) Evaluation of analgesic interaction between morphine, dexmedetomidine and maropitant using hot-plate and tail-flick tests in rats. Veterinary Anaesth Analg 46:476–482. https://doi.org/10.1016/j.vaa.2018.12.009
Kiyatkin EA, Zhukov VN (1991) Dopaminergic involvement in the mediation of morphine effects on vocalization and movement reactivity in the rat. Int J Neurosci 58:1–5. https://doi.org/10.3109/00207459108987180
Lazenka MF, Freitas KC, Henck S, Negus SS (2017) Relief of pain-depressed behavior in rats by activation of D1-like dopamine receptors. J Pharmacol Exp Ther 362(1):14–23. https://doi.org/10.1124/jpet.117.240796
Li S, Guan S, Wang Y, Cheng L, Yang Q, Tian Z, Zhao M, Wang X, Feng B (2019) Nicotine inhibits rapamycin-induced pain through activating mTORC1/S6K/IRS-1-related feedback inhibition loop. Brain Res Bull 149:75–85. https://doi.org/10.1016/j.brainresbull.2019.04.016
Liu S, Tang Y, Shu H, Tatum D, Bai Q, Crawford J, Xing Y, Lobo MK, Bellinger L, Kramer P, Tao F (2019) Dopamine receptor D2, but not D1, mediates descending dopaminergic pathway-produced analgesic effect in a trigeminal neuropathic pain mouse model. Pain 160:334–344. https://doi.org/10.1097/j.pain.0000000000001414
López-Avila A, Coffeen U, Ortega-Legaspi MJ, del Angel R, Pellicer F (2004) Dopamine and NMDA systems modulate long-term nociception in the rat anterior cingulate cortex. Pain 111(1–2):136–143. https://doi.org/10.1016/j.pain.2004.06.010
Maggio R, Aloisi G, Silvano E, Rossi M, Millan MJ (2009) Heterodimerization of dopamine receptors: new insights into functional and therapeutic significance. Parkinsonism Relat Disord 15(Suppl 4):S2-7. https://doi.org/10.1016/S1353-8020(09)70826-0
Marinelli M, Aouizerate B, Barrot M, Le Moal M, Piazza PV (1998) Dopamine-dependent responses to morphine depend on glucocorticoid receptors. Proc Natl Acad Sci U S A 95:7742–7747. https://doi.org/10.1073/pnas.95.13.7742
Morgan MJ, Franklin KB (1990) 6-Hydroxydopamine lesions of the ventral tegmentum abolish D-amphetamine and morphine analgesia in the formalin test but not in the tail flick test. Brain Res 519:144–149. https://doi.org/10.1016/0006-8993(90)90072-j
Narita M, Matsushima Y, Niikura K, Narita M, Takagi S, Nakahara K, Kurahashi K, Abe M, Saeki M, Asato M, Imai S, Ikeda K, Kuzumaki N, Suzuki T (2010) Implication of dopaminergic projection from the ventral tegmental area to the anterior cingulate cortex in μ-opioid-induced place preference. Addict Biol 15(4):434–447. https://doi.org/10.1111/j.1369-1600.2010.00249.x
Navratilova E, Porreca F (2014) Reward and motivation in pain and pain relief. Nat Neurosci 17(10):1304–1312. https://doi.org/10.1038/nn.3811
Navratilova E, Atcherley C, Porreca F (2015a) Brain circuits encoding reward from pain relief. Trends Neurosci 38(11):741–750. https://doi.org/10.1016/j.tins.2015.09.003
Navratilova E, Xie JY, Meske D, Qu C, Morimura K, Okun A, Arakawa N, Ossipov M, Fields HL, Porreca F (2015b) Endogenous opioid activity in the anterior cingulate cortex is required for relief of pain. J Neurosci 35(18):7264–7271. https://doi.org/10.1523/JNEUROSCI.3862-14.2015
Nazarinia E, Rezayof A, Sardari M, Yazdanbakhsh N (2017) Contribution of the basolateral amygdala NMDA and muscarinic receptors in rat’s memory retrieval. Neurobiol Learn and Mem 139:28–36. https://doi.org/10.1016/j.nlm.2016.12.008
Nazari-Serenjeh F, Rezayof A, Zarrindast M-R (2011) Functional correlation between GABAergic and dopaminergic systems of dorsal hippocampus and ventral tegmental area in passive avoidance learning in rats. Neuroscience 196:104–114. https://doi.org/10.1016/j.neuroscience.2011.08.073
Norouzi M, Mousavi Z, Shafaroodi H (2017) Aripiprazole prolongs morphine antinociception effect and disrupts acute morphine tolerance. Biomed Pharmacol J 10:1149–1157. https://doi.org/10.13005/bpj/1215
Ortega-Legaspi MJ, de Gortari P, Garduno-Gutierrez R, Amaya MI, Leon-Olea M, Coffeen U, Pellicer F (2011) Expression of the dopaminergic D1 and D2 receptors in the anterior cingulate cortex in a model of neuropathic pain. Mol Pain 7:97. https://doi.org/10.1186/1744-8069-7-97
Ozdemir E, Bagcivan I, Gursoy S (2013) Role of D1/D2 dopamine receptors antagonist perphenazine in morphine analgesia and tolerance in rats. Bosn J Basic Med Sci 13:119–125. https://doi.org/10.17305/bjbms.2013.2394
Paxinos G, Watson C (2007) the rat brain in stereotaxic coordinates in stereotaxic coordinates. Elsevier, pp 139–140
Picard F, Sadaghiani S, Leroy C, Courvoisier DS, Maroy R, Bottlaender M (2013) High density of nicotinic receptors in the cingulo-insular network. Neuroimage 1(79):42–51. https://doi.org/10.1016/j.neuroimage.2013.04.074
Porreca F, Ossipov MH (2009) Nausea and vomiting side effects with opioid analgesics during treatment of chronic pain: mechanisms, implications, and management options. Pain Med 10:654–662. https://doi.org/10.1111/j.1526-4637.2009.00583.x
Rezayof A, Zarrindast M-R, Sahraei H, Haeri-Rohani A (2003) Involvement of dopamine receptors of the dorsal hippocampus on the acquisition and expression of morphine-induced place preference in rats. J Psychopharmacol 17(415):423. https://doi.org/10.1177/0269881103174005
Suh HW, Song DK, Lee KJ, Choi SR, Kim YH (1996) Intrathecally injected nicotine enhances the antinociception induced by morphine but not beta-endorphin, D-Pen 2,5-enkephalin and U50,488H administered intrathecally in the mouse. Neuropeptides 30:373–8. https://doi.org/10.1016/s0143-4179(96)90027-x
Suh HW, Song DK, Choi SR, Chung KM, Kim YH (1996) Nicotine enhances morphine- and β-endorphin-induced antinociception at the supraspinal level in the mouse. Neuropeptides 30:479–484. https://doi.org/10.1016/s0143-4179(96)90013-x
Taylor BK, Joshi C, Uppal H (2003) Stimulation of dopamine D2 receptors in the nucleus accumbens inhibits inflammatory pain. Brain Res 987:135–143. https://doi.org/10.1016/s0006-8993(03)03318-3
Taylor AMW, Becker S, Schweinhardt P, Cahill C (2016) Mesolimbic dopamine signaling in acute and chronic pain: implications for motivation, analgesia, and addiction. Pain 157(6):1194–1198. https://doi.org/10.1097/j.pain.0000000000000494
Vogt BA (2005) Pain and emotion interactions in subregions of the cingulate gyrus. Nat Rev Neurosci 6:533–544. https://doi.org/10.1038/nrn1704
Wehring HJ, Heishman SJ, McMahon RP, Liu F, FeldmanS RH, Weiner E, Kelly DL (2017) Antipsychotic treatment and tobacco craving in people with schizophrenia. J Dual Diagn 13(1):36–42. https://doi.org/10.1080/15504263.2017.1288946
Wood PB (2008) Role of central dopamine in pain and analgesia. Expert Rev Neurother 8(5):781–797. https://doi.org/10.1586/14737175.8.5.781
Xiao X, Zhang YQ (2018) A new perspective on the anterior cingulate cortex and affective pain. Neurosci Behav Rev 90:200–211. https://doi.org/10.1016/j.neubiorev.2018.03.022
Yam MF, Loh YC, Tan CS, Adam SK, Manan NA, Basir R (2018) General pathways of pain sensation and the major neurotransmitters involved in pain regulation. Int J Mol Sci 19(8):2164. https://doi.org/10.3390/ijms19082164
Yankov K (2010) Dose-effect modeling of experimental data. J Inf Control Manag Syst 8(3):257
Zarrindast MR, Nami AB, Farzin D (1996) Nicotine potentiates morphine antinociception: a possible cholinergic mechanism. Eur Neuropsychopharmacol 6:127–133. https://doi.org/10.1016/0924-977x(96)00002-8
Zarrindast MR, Pazouki M, Nassiri-Rad S (1997) Involvement of cholinergic and opioid receptor mechanisms in nicotine-induced antinociception. Pharmacol Toxicol 81:209–213. https://doi.org/10.1111/j.1600-0773.1997.tb00048.x
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sardari, M., Seddighfar, M. & Sardari, S. Dopamine receptors in the anterior cingulate cortex implicate in nicotine enhanced morphine analgesia. Psychopharmacology 238, 3311–3323 (2021). https://doi.org/10.1007/s00213-021-05947-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-021-05947-z