Abstract
Rationale
Strategies are needed to decrease the abuse liability of mu opioid receptor (MOR) agonists. One strategy under consideration is to combine MOR agonists with kappa opioid receptor (KOR) agonists.
Objectives
The effects of KOR agonists (U50488, nalfurafine) on fentanyl-vs.-food choice were compared under conditions where the KOR agonists were added to the intravenously self-administered fentanyl (contingent delivery) or administered as subcutaneous pretreatments (non-contingent delivery) in male and female rats.
Methods
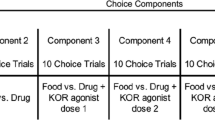
Rats were trained to respond under a concurrent schedule of fentanyl (0, 0.32–10 μg/kg/infusion) and food reinforcement. In experiment 1, U50488 and nalfurafine were co-administered with fentanyl as fixed-proportion mixtures (contingent administration). In experiment 2, U50488 (1–10 mg/kg) and nalfurafine (3.2–32 μg/kg) were administered as acute pretreatments (non-contingent administration). The selective KOR antagonist, nor-BNI (32 mg/kg), was administered prior to contingent and non-contingent KOR-agonist treatment in experiment 3.
Results
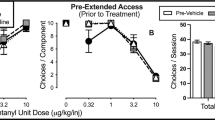
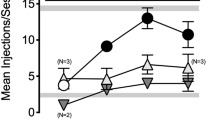
Both U50488 and nalfurafine decreased fentanyl choice when administered contingently, demonstrating that KOR agonists punish opioid choice. However, evidence for punishment corresponded with an elimination of operant responding in the majority of rats. Non-contingent U50488 and nalfurafine administration only decreased the number of choices made during the behavioral session without altering fentanyl choice. Contingent and non-contingent KOR-agonist effects on fentanyl choice were both attenuated by nor-BNI.
Conclusions
These results illustrate that the effects of KOR agonists on fentanyl reinforcement are dependent upon the contingencies under which they are administered.



Similar content being viewed by others
References
Azrin NH (1959) Punishment and recovery during fixed-ratio performance. J Exp Anal Behav 2:301–305. https://doi.org/10.1901/jeab.1959.2-301
Azrin NHH (1966) Punishment. In: Honig WK (ed) Operant behavior: areas of research and application. Appleton-Century-Crofts, New York
Banks ML (2020) The rise and fall of kappa-opioid receptors in drug abuse research. Handb Exp Pharmacol 258:147–165. https://doi.org/10.1007/164_2019_268
Banks ML, Negus SS (2012) Preclinical determinants of drug choice under concurrent schedules of drug self-administration. Adv Pharmacol Sci 2012:281768. https://doi.org/10.1155/2012/281768
Bergman J, Johanson CE (1981) The effects of electric shock on responding maintained by cocaine in rhesus monkeys. Pharmacol Biochem Behav 14:423–426. https://doi.org/10.1016/0091-3057(81)90413-5
Bhargava HN, Matwyshyn GA, Reddy PL, Veeranna (1994) Brain and spinal cord kappa opiate receptors and pharmacological responses to U-50,488H in rats of differing ages. Pharmacol Biochem Behav 48:87–91. https://doi.org/10.1016/0091-3057(94)90502-9
Bowen CA, Negus SS, Zong R, Neumeyer JL, Bidlack JM, Mello NK (2003) Effects of mixed-action kappa/mu opioids on cocaine self-administration and cocaine discrimination by rhesus monkeys. Neuropsychopharmacology 28:1125–1139. https://doi.org/10.1038/sj.npp.1300105
Bruchas MR, Chavkin C (2010) Kinase cascades and ligand-directed signaling at the kappa opioid receptor. Psychopharmacology 210:137–147. https://doi.org/10.1007/s00213-010-1806-y
Bruchas MR, Land BB, Aita M, Xu M, Barot SK, Li S, Chavkin C (2007) Stress-induced p38 mitogen-activated protein kinase activation mediates kappa-opioid-dependent dysphoria. J Neurosci 27:11614–11623. https://doi.org/10.1523/JNEUROSCI.3769-07.2007
CDC, Prevention USCfDCa (2019) Trends in annual opioid prescribing rates by overall and high-dosage prescriptions vol https://www.cdc.gov/drugoverdose/data/prescribing/prescribing-practices.html
CDER (2015) Abuse-deterrent opioids —evaluation and labeling vol https://www.fda.gov/regulatory-information/search-fda-guidance-documents/abuse-deterrent-opioids-evaluation-and-labeling. U.S. Department of Health and Human Services
Cosgrove KP, Carroll ME (2002) Effects of bremazocine on self-administration of smoked cocaine base and orally delivered ethanol, phencyclidine, saccharin, and food in rhesus monkeys: a behavioral economic analysis. J Pharmacol Exp Ther 301:993–1002. https://doi.org/10.1124/jpet.301.3.993
Craft RM, Bernal SA (2001) Sex differences in opioid antinociception: kappa and ‘mixed action’ agonists. Drug Alcohol Depend 63:215–228. https://doi.org/10.1016/s0376-8716(00)00209-x
Crowley RS, Riley AP, Alder AF, Anderson RJ III, Luo D, Kaska S, Maynez P, Kivell BM, Prisinzano TE (2020) Synthetic studies of neoclerodane diterpenes from Salvia divinorum: design, synthesis, and evaluation of analogues with improved potency and G-protein activation bias at the mu opioid receptor. ACS Chem Neurosci 11:1781–1790. https://doi.org/10.1021/acschemneuro.0c00191
Czoty PW, Stoops WW, Rush CR (2016) Evaluation of the “pipeline” for development of medications for cocaine use disorder: a review of translational preclinical, human laboratory, and clinical trial research. Pharmacol Rev 68:533–562. https://doi.org/10.1124/pr.115.011668
Devine DP, Leone P, Pocock D, Wise RA (1993) Differential involvement of ventral tegmental mu, delta and kappa opioid receptors in modulation of basal mesolimbic dopamine release: in vivo microdialysis studies. J Pharmacol Exp Ther 266:1236–1246
Di Chiara G, Imperato A (1988) Opposite effects of mu and kappa opiate agonists on dopamine release in the nucleus accumbens and in the dorsal caudate of freely moving rats. J Pharmacol Exp Ther 244:1067–1080
Donzanti BA, Althaus JS, Payson MM, Von Voigtlander PF (1992) Kappa agonist-induced reduction in dopamine release: site of action and tolerance. Res Commun Chem Pathol Pharmacol 78:193–210
Endoh T, Matsuura H, Tanaka C, Nagase H (1992) Nor-binaltorphimine: a potent and selective kappa-opioid receptor antagonist with long-lasting activity in vivo. Arch Int Pharmacodyn Ther 316:30–42
Escobar ADP, Casanova JP, Andres ME, Fuentealba JA (2020) Crosstalk between kappa opioid and dopamine systems in compulsive behaviors. Front Pharmacol 11:57. https://doi.org/10.3389/fphar.2020.00057
Faunce KE, Banks ML (2020) Effects of repeated kappa-opioid receptor agonist U-50488 treatment and subsequent termination on intracranial self-stimulation in male and female rats. Exp Clin Psychopharmacol 28:44–54. https://doi.org/10.1037/pha0000287
Freeman KB, Naylor JE, Prisinzano TE, Woolverton WL (2014) Assessment of the kappa opioid agonist, salvinorin A, as a punisher of drug self-administration in monkeys. Psychopharmacology 231:2751–2758. https://doi.org/10.1007/s00213-014-3436-2
Glick SD, Maisonneuve IM, Raucci J, Archer S (1995) Kappa opioid inhibition of morphine and cocaine self-administration in rats. Brain Res 681:147–152. https://doi.org/10.1016/0006-8993(95)00306-b
Glick SD, Visker KE, Maisonneuve IM (1998) Effects of cyclazocine on cocaine self-administration in rats. Eur J Pharmacol 357:9–14. https://doi.org/10.1016/s0014-2999(98)00548-2
Grove RN, Schuster CR (1974) Suppression of cocaine self-administration by extinction and punishment. Pharmacol Biochem Behav 2:199–208. https://doi.org/10.1016/0091-3057(74)90053-7
Gullapalli S, Ramarao P (2002) Role of L-type Ca(2+) channels in pertussis toxin induced antagonism of U50,488H analgesia and hypothermia. Brain Res 946:191–197. https://doi.org/10.1016/s0006-8993(02)02880-9
Haney M, Spealman R (2008) Controversies in translational research: drug self-administration. Psychopharmacology 199:403–419. https://doi.org/10.1007/s00213-008-1079-x
Huskinson SL, Naylor JE, Townsend EA, Rowlett JK, Blough BE, Freeman KB (2017) Self-administration and behavioral economics of second-generation synthetic cathinones in male rats. Psychopharmacology 234:589–598. https://doi.org/10.1007/s00213-016-4492-6
Huskinson SL, Platt DM, Brasfield M, Follett ME, Prisinzano TE, Blough BE, Freeman KB (2020) Quantification of observable behaviors induced by typical and atypical kappa-opioid receptor agonists in male rhesus monkeys. Psychopharmacology. https://doi.org/10.1007/s00213-020-05519-7
Jacobs DS, Moghaddam B (2020) Prefrontal cortex representation of learning of punishment probability during reward-motivated actions. J Neurosci. https://doi.org/10.1523/JNEUROSCI.0310-20.2020
Johanson CE (1975) Pharmacological and environmental variables affecting drug preference in rhesus monkeys. Pharmacol Rev 27:343–355
Johanson CE (1977) The effects of electric shock on responding maintained by cocaine injections in a choice procedure in the rhesus monkey. Psychopharmacology 53:277–282. https://doi.org/10.1007/BF00492364
Kaski SW et al (2019) Preclinical testing of nalfurafine as an opioid-sparing adjuvant that potentiates analgesia by the mu opioid receptor-targeting agonist morphine. J Pharmacol Exp Ther 371:487–499. https://doi.org/10.1124/jpet.118.255661
Kavaliers M, Innes DG (1987) Sex and day-night differences in opiate-induced responses of insular wild deer mice, Peromyscus maniculatus triangularis. Pharmacol Biochem Behav 27:477–482. https://doi.org/10.1016/0091-3057(87)90351-0
Keselman HJ, Cribbie R, Holland B (2002) Controlling the rate of type I error over a large set of statistical tests. Br J Math Stat Psychol 55:27–39. https://doi.org/10.1348/000711002159680
Kozono H, Yoshitani H, Nakano R (2018) Post-marketing surveillance study of the safety and efficacy of nalfurafine hydrochloride (Remitch((R)) capsules 2.5 mug) in 3,762 hemodialysis patients with intractable pruritus. Int J Nephrol Renov Dis 11:9–24. https://doi.org/10.2147/IJNRD.S145720
Kumor KM, Haertzen CA, Johnson RE, Kocher T, Jasinski D (1986) Human psychopharmacology of ketocyclazocine as compared with cyclazocine, morphine and placebo. J Pharmacol Exp Ther 238:960–968
Kutlu MG et al (2020) A novel multidimensional reinforcement task in mice elucidates sex-specific behavioral strategies. Neuropsychopharmacology. https://doi.org/10.1038/s41386-020-0692-1
Kuzmin AV, Semenova S, Gerrits MA, Zvartau EE, Van Ree JM (1997) Kappa-opioid receptor agonist U50,488H modulates cocaine and morphine self-administration in drug-naive rats and mice. Eur J Pharmacol 321:265–271. https://doi.org/10.1016/s0014-2999(96)00961-2
Lazenka ML, Moerke MJ, Townsend EA, Freeman KB, Carroll FI, Negus SS (2018) Dissociable effects of the kappa opioid receptor agonist nalfurafine on pain/itch-stimulated and pain/itch-depressed behaviors in male rats. Psychopharmacology 235:203–213. https://doi.org/10.1007/s00213-017-4758-7
Liu JJ et al (2019) Phosphoproteomic approach for agonist-specific signaling in mouse brains: mTOR pathway is involved in kappa opioid aversion. Neuropsychopharmacology 44:939–949. https://doi.org/10.1038/s41386-018-0155-0
Margolis EB, Hjelmstad GO, Bonci A, Fields HL (2003) Kappa-opioid agonists directly inhibit midbrain dopaminergic neurons. J Neurosci 23:9981–9986
Mello NK, Negus SS (1996) Preclinical evaluation of pharmacotherapies for treatment of cocaine and opioid abuse using drug self-administration procedures. Neuropsychopharmacology 14:375–424. https://doi.org/10.1016/0893-133X(95)00274-H
Mello NK, Negus SS (1998) Effects of kappa opioid agonists on cocaine- and food-maintained responding by rhesus monkeys. J Pharmacol Exp Ther 286:812–824
Minervini V, Osteicoechea DC, Casalez A, France CP (2019) Punishment and reinforcement by opioid receptor agonists in a choice procedure in rats. Behav Pharmacol 30:335–342. https://doi.org/10.1097/FBP.0000000000000436
Mores KL, Cummins BR, Cassell RJ, van Rijn RM (2019) A review of the therapeutic potential of recently developed G protein-biased kappa agonists. Front Pharmacol 10:407. https://doi.org/10.3389/fphar.2019.00407
Negus SS (2004) Effects of the kappa opioid agonist U50,488 and the kappa opioid antagonist nor-binaltorphimine on choice between cocaine and food in rhesus monkeys. Psychopharmacology 176:204–213. https://doi.org/10.1007/s00213-004-1878-7
Negus SS (2005) Effects of punishment on choice between cocaine and food in rhesus monkeys. Psychopharmacology 181:244–252. https://doi.org/10.1007/s00213-005-2266-7
Negus SS, Mello NK, Portoghese PS, Lin CE (1997) Effects of kappa opioids on cocaine self-administration by rhesus monkeys. J Pharmacol Exp Ther 282:44–55
Negus SS, Schrode K, Stevenson GW (2008) Micro/kappa opioid interactions in rhesus monkeys: implications for analgesia and abuse liability. Exp Clin Psychopharmacol 16:386–399. https://doi.org/10.1037/a0013088
Orsini CA, Willis ML, Gilbert RJ, Bizon JL, Setlow B (2016) Sex differences in a rat model of risky decision making. Behav Neurosci 130:50–61. https://doi.org/10.1037/bne0000111
Pergolizzi JV Jr, Rosenblatt M, LeQuang JA (2019) Three years down the road: the aftermath of the CDC guideline for prescribing opioids for chronic pain. Adv Ther 36:1235–1240. https://doi.org/10.1007/s12325-019-00954-1
Pfeiffer A, Brantl V, Herz A, Emrich HM (1986) Psychotomimesis mediated by kappa opiate receptors. Science 233:774–776. https://doi.org/10.1126/science.3016896
Russell SE, Rachlin AB, Smith KL, Muschamp J, Berry L, Zhao Z, Chartoff EH (2014) Sex differences in sensitivity to the depressive-like effects of the kappa opioid receptor agonist U-50488 in rats. Biol Psychiatry 76:213–222. https://doi.org/10.1016/j.biopsych.2013.07.042
Schattauer SS, Kuhar JR, Song A, Chavkin C (2017) Nalfurafine is a G-protein biased agonist having significantly greater bias at the human than rodent form of the kappa opioid receptor. Cell Signal 32:59–65. https://doi.org/10.1016/j.cellsig.2017.01.016
Schenk S, Partridge B, Shippenberg TS (1999) U69593, a kappa-opioid agonist, decreases cocaine self-administration and decreases cocaine-produced drug-seeking. Psychopharmacology 144:339–346. https://doi.org/10.1007/s002130051016
Schenk S, Partridge B, Shippenberg TS (2001) Effects of the kappa-opioid receptor agonist, U69593, on the development of sensitization and on the maintenance of cocaine self-administration. Neuropsychopharmacology 24:441–450. https://doi.org/10.1016/S0893-133X(00)00190-1
Spanagel R, Herz A, Shippenberg TS (1990) The effects of opioid peptides on dopamine release in the nucleus accumbens: an in vivo microdialysis study. J Neurochem 55:1734–1740. https://doi.org/10.1111/j.1471-4159.1990.tb04963.x
Tejeda HA, Bonci A (2019) Dynorphin/kappa-opioid receptor control of dopamine dynamics: implications for negative affective states and psychiatric disorders. Brain Res 1713:91–101. https://doi.org/10.1016/j.brainres.2018.09.023
Thompson AC, Zapata A, Justice JB Jr, Vaughan RA, Sharpe LG, Shippenberg TS (2000) Kappa-opioid receptor activation modifies dopamine uptake in the nucleus accumbens and opposes the effects of cocaine. J Neurosci 20:9333–9340
Townsend EA et al (2017) Effects of nalfurafine on the reinforcing, thermal antinociceptive, and respiratory-depressant effects of oxycodone: modeling an abuse-deterrent opioid analgesic in rats. Psychopharmacology 234:2597–2605. https://doi.org/10.1007/s00213-017-4652-3
Townsend EA et al (2019a) Conjugate vaccine produces long-lasting attenuation of fentanyl vs. food choice and blocks expression of opioid withdrawal-induced increases in fentanyl choice in rats. Neuropsychopharmacology. https://doi.org/10.1038/s41386-019-0385-9
Townsend EA, Negus SS, Caine SB, Thomsen M, Banks ML (2019b) Sex differences in opioid reinforcement under a fentanyl vs. food choice procedure in rats. Neuropsychopharmacology. https://doi.org/10.1038/s41386-019-0356-1
Tsuji M, Takeda H, Matsumiya T, Nagase H, Narita M, Suzuki T (2001) The novel kappa-opioid receptor agonist TRK-820 suppresses the rewarding and locomotor-enhancing effects of morphine in mice. Life Sci 68:1717–1725. https://doi.org/10.1016/s0024-3205(01)00957-2
Verharen JPH, Luijendijk MCM, Vanderschuren L, Adan RAH (2020) Dopaminergic contributions to behavioral control under threat of punishment in rats. Psychopharmacology. https://doi.org/10.1007/s00213-020-05497-w
Walker JM, Thompson LA, Frascella J, Friederich MW (1987) Opposite effects of mu and kappa opiates on the firing-rate of dopamine cells in the substantia nigra of the rat. Eur J Pharmacol 134:53–59. https://doi.org/10.1016/0014-2999(87)90130-0
Walsh SL, Geter-Douglas B, Strain EC, Bigelow GE (2001a) Enadoline and butorphanol: evaluation of kappa-agonists on cocaine pharmacodynamics and cocaine self-administration in humans. J Pharmacol Exp Ther 299:147–158
Walsh SL, Strain EC, Abreu ME, Bigelow GE (2001b) Enadoline, a selective kappa opioid agonist: comparison with butorphanol and hydromorphone in humans. Psychopharmacology 157:151–162. https://doi.org/10.1007/s002130100788
Wilson N, Kariisa M, Seth P, Smith H, Davis NL (2020) Drug and opioid-involved overdose deaths - United States, 2017-2018. MMWR Morb Mortal Wkly Rep 69:290–297. https://doi.org/10.15585/mmwr.mm6911a4
Zamarripa CA, Naylor JE, Huskinson SL, Townsend EA, Prisinzano TE, Freeman KB (2020) Kappa opioid agonists reduce oxycodone self-administration in male rhesus monkeys. Psychopharmacology. https://doi.org/10.1007/s00213-020-05473-4
Acknowledgments
The manuscript content is solely the responsibility of the author and does not necessarily reflect the official views of the National Institutes of Health. The author would like to thank Dr. Matthew L. Banks, Dr. Kevin B. Freeman, and Mr. C. Austin Zamarripa for comments on an earlier version of the manuscript.
Funding
This work was supported by (1) a Research Grant awarded by the Virginia Commonwealth University Postdoctoral Association, (2) the National Institute on Drug Abuse of the National Institutes of Health under Award Numbers F32DA047026 and P30DA033934, and (3) the National Institute on Alcohol Abuse and Alcoholism and the National Institute on Drug Abuse Intramural Research Programs.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The manuscript content is solely the responsibility of the author and does not necessarily reflect the official views of the National Institutes of Health.
Conflict of interest
The author declares that he has no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Townsend, E.A. Effects of kappa opioid receptor agonists on fentanyl vs. food choice in male and female rats: contingent vs. non-contingent administration. Psychopharmacology 238, 1017–1028 (2021). https://doi.org/10.1007/s00213-020-05749-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-020-05749-9