Abstract
Rationale
Previous neuroimaging studies of cognition involving nicotinic acetylcholine receptor (nAChR) agonist administration have repeatedly found enhanced task-induced deactivation of regions of the default mode network (DMN), a group of brain systems that is more active at rest and mediates task-independent thought processes. This effect may be related to pro-cognitive nAChR agonist effects
Objectives
The present study sought to test whether nAChR modulation of the DMN is bi-directional, i.e., whether a nAChR antagonist would reduce task-induced deactivation.
Methods
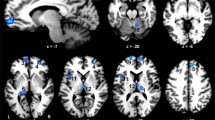
Eighteen healthy non-smokers underwent functional magnetic resonance imaging while performing a letter N-back task. Scans were performed after nicotine administration (7 mg/24 h, transdermally), after administration of the nAChR antagonist mecamylamine (7.5 mg, p.o.), and after double placebo, in counterbalanced sequence. Blood-oxygen-level-dependent (BOLD) signal was analyzed within ventromedial prefrontal cortex (vmPFC) and posterior cingulate cortex (PCC) regions of interest—central hubs of the DMN in which consistent nAChR agonist–induced changes had previously been identified.
Results
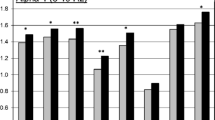
Nicotine enhanced hit rate in both the 0-back and 2-back condition, while mecamylamine slowed reaction time in the 2-back condition. Mecamylamine reduced task-induced deactivation of vmPFC and PCC. Nicotine had no significant effects on the BOLD signal.
Conclusions
The finding that nAChR tone reduction by mecamylamine weakened task-induced DMN deactivation indicates that a constant tone of nAChR activation helps regulate DMN activity in healthy individuals. This suggests that low nAChR tone may play a causal role in DMN dysregulation seen in conditions such as mild cognitive impairment or Alzheimer’s disease.



Similar content being viewed by others
References
Adams CE, Stevens KE (2007) Evidence for a role of nicotinic acetylcholine receptors in schizophrenia. Front Biosci 12:4755–4772
Alvarez-Jimenez R, Baakman AC, Stevens J, Goulooze SC, Hart EP, Rissmann R, van Gerven JMA, Groeneveld GJ (2017) Pharmacokinetics and pharmacodynamics of oral mecamylamine - development of a nicotinic acetylcholine receptor antagonist cognitive challenge test using modelling and simulation. J Psychopharmacol 31:192–203. https://doi.org/10.1177/0269881116681417
Aronson Fischell S, Ross TJ, Deng ZD, Salmeron BJ, Stein EA (2020) Transcranial direct current stimulation applied to the dorsolateral and ventromedial prefrontal cortices in smokers modifies cognitive circuits implicated in the nicotine withdrawal syndrome. Biol Psychiatry Cogn Neurosci Neuroimaging 5:448–460. https://doi.org/10.1016/j.bpsc.2019.12.020
Beaver JD, Long CJ, Cole DM, Durcan MJ, Bannon LC, Mishra RG, Matthews PM (2011) The effects of nicotine replacement on cognitive brain activity during smoking withdrawal studied with simultaneous fMRI/EEG. Neuropsychopharmacology 36:1792–1800. https://doi.org/10.1038/npp.2011.53
Buckner RL, DiNicola LM (2019) The brain’s default network: updated anatomy, physiology and evolving insights. Nat Rev Neurosci 20:593–608. https://doi.org/10.1038/s41583-019-0212-7
Buckner RL, Andrews-Hanna JR, Schacter DL (2008) The brain’s default network: anatomy, function, and relevance to disease. Ann N Y Acad Sci 1124:1–38
Ceko M, Gracely JL, Fitzcharles MA, Seminowicz DA, Schweinhardt P, Bushnell MC (2015) Is a responsive default mode network required for successful working memory task performance? J Neurosci 35:11595–11605. https://doi.org/10.1523/JNEUROSCI.0264-15.2015
Contestabile A (2011) The history of the cholinergic hypothesis. Behav Brain Res 221:334–340. https://doi.org/10.1016/j.bbr.2009.12.044
Cox RW (1996) AFNI: software for analysis and visualization of functional magnetic resonance neuroimages. Comput Biomed Res 29:162–173
Esposito F, Bertolino A, Scarabino T, Latorre V, Blasi G, Popolizio T, Tedeschi G, Cirillo S, Goebel R, di Salle F (2006) Independent component model of the default-mode brain function: assessing the impact of active thinking. Brain Res Bull 70:263–269
Ettinger U, Williams SCR, Patel D, Michel TM, Nwaigwe A, Caceres A, Mehta MA, Anilkumar AP, Kumari V (2009) Effects of acute nicotine on brain function in healthy smokers and non-smokers: estimation of inter-individual response heterogeneity. Neuroimage 45:549–561. https://doi.org/10.1016/j.neuroimage.2008.12.029
Fant RV, Henningfield JE, Shiffman S, Strahs KR, Reitberg DP (2000) A pharmacokinetic crossover study to compare the absorption characteristics of three transdermal nicotine patches. Pharmacol Biochem Behav 67:479–482
Froeliger B, Modlin L, Wang L, Kozink RV, McClernon FJ (2012) Nicotine withdrawal modulates frontal brain function during an affective Stroop task. Psychopharmacology 220:707–718. https://doi.org/10.1007/s00213-011-2522-y
Gorsline J, Gupta SK, Dye D, Rolf CN (1993) Steady-state pharmacokinetics and dose relationship of nicotine delivered from Nicoderm (Nicotine Transdermal System). J Clin Pharmacol 33:161–168
Gupta SK, Benowitz NL, Jacob P 3rd, Rolf CN, Gorsline J (1993) Bioavailability and absorption kinetics of nicotine following application of a transdermal system. Br J Clin Pharmacol 36:221–227
Gusnard DA, Raichle ME (2001) Searching for a baseline: functional imaging and the resting human brain. Nat Rev Neurosci 2:685–694
Hahn B (2015) Nicotinic receptors and attention. Curr Top Behav Neurosci 23:103–135. https://doi.org/10.1007/978-3-319-13665-3_5
Hahn B, Ross TJ, Yang Y, Kim I, Huestis MA, Stein EA (2007) Nicotine enhances visuospatial attention by deactivating areas of the resting brain default network. J Neurosci 27:3477–3489
Hahn B, Ross TJ, Wolkenberg FA, Shakleya DM, Huestis MA, Stein EA (2009) Performance effects of nicotine during selective attention, divided attention, and simple stimulus detection: an fMRI study. Cereb Cortex 19:1990–2000. https://doi.org/10.1093/cercor/bhn226
Hahn B, Olmstead CK, Yuille MB, Chiappelli JJ, Wells AK (2020a) Attention-enhancing effects of propranolol and synergistic effects with nicotine. Cogn Affect Behav Neurosci 20:658–668. https://doi.org/10.3758/s13415-020-00794-5
Hahn B, Shrieves ME, Olmstead CK, Yuille MB, Chiappelli JJ, Pereira EFR, Albuquerque EX, Fawcett WP (2020b) Evidence for positive allosteric modulation of cognitive-enhancing effects of nicotine in healthy human subjects. Psychopharmacology 237:219–230. https://doi.org/10.1007/s00213-019-05363-4
Hasselmo ME, Sarter M (2011) Modes and models of forebrain cholinergic neuromodulation of cognition. Neuropsychopharmacology 36:52–73. https://doi.org/10.1038/npp.2010.104
Heishman SJ, Kleykamp BA, Singleton EG (2010) Meta-analysis of the acute effects of nicotine and smoking on human performance. Psychopharmacology 210:453–469
Kendziorra K, Wolf H, Meyer PM, Barthel H, Hesse S, Becker GA, Luthardt J, Schildan A, Patt M, Sorger D, Seese A, Gertz HJ, Sabri O (2011) Decreased cerebral alpha4beta2* nicotinic acetylcholine receptor availability in patients with mild cognitive impairment and Alzheimer’s disease assessed with positron emission tomography. Eur J Nucl Med Mol Imaging 38:515–525. https://doi.org/10.1007/s00259-010-1644-5
Kriegeskorte N, Simmons WK, Bellgowan PS, Baker CI (2009) Circular analysis in systems neuroscience: the dangers of double dipping. Nat Neurosci 12:535–540. https://doi.org/10.1038/nn.2303
Loughead J, Ray R, Wileyto EP, Ruparel K, Sanborn P, Siegel S, Gur RC, Lerman C (2010) Effects of the alpha4beta2 partial agonist varenicline on brain activity and working memory in abstinent smokers. Biol Psychiatry 67:715–721. https://doi.org/10.1016/j.biopsych.2010.01.016
Loughead J, Wileyto EP, Ruparel K, Falcone M, Hopson R, Gur R, Lerman C (2015) Working memory-related neural activity predicts future smoking relapse. Neuropsychopharmacology 40:1311–1320. https://doi.org/10.1038/npp.2014.318
Lustig C, Snyder AZ, Bhakta M, O'Brien KC, McAvoy M, Raichle ME, Morris JC, Buckner RL (2003) Functional deactivations: change with age and dementia of the Alzheimer type. Proc Natl Acad Sci U S A 100:14504–14509
Mason MF, Norton MI, Van Horn JD, Wegner DM, Grafton ST, Macrae CN (2007) Wandering minds: the default network and stimulus-independent thought. Science 315:393–395
Metzak PD, Riley JD, Wang L, Whitman JC, Ngan ET, Woodward TS (2012) Decreased efficiency of task-positive and task-negative networks during working memory in schizophrenia. Schizophr Bull 38:803–813. https://doi.org/10.1093/schbul/sbq154
Nakaizumi K, Ouchi Y, Terada T, Yoshikawa E, Kakimoto A, Isobe T, Bunai T, Yokokura M, Suzuki K, Magata Y (2018) In vivo Depiction of alpha7 nicotinic receptor loss for cognitive decline in Alzheimer’s disease. J Alzheimers Dis 61:1355–1365. https://doi.org/10.3233/JAD-170591
Newhouse PA, Potter A, Corwin J, Lenox R (1992) Acute nicotinic blockade produces cognitive impairment in normal humans. Psychopharmacology 108:480–484
Newhouse PA, Potter A, Kelton M, Corwin J (2001) Nicotinic treatment of Alzheimer’s disease. Biol Psychiatry 49:268–278. https://doi.org/10.1016/s0006-3223(00)01069-6
Newhouse PA, Potter A, Singh A (2004) Effects of nicotinic stimulation on cognitive performance. Curr Opin Pharmacol 4:36–46
Newhouse PA, Potter AS, Dumas JA, Thiel CM (2011) Functional brain imaging of nicotinic effects on higher cognitive processes. Biochem Pharmacol 82:943–951. https://doi.org/10.1016/j.bcp.2011.06.008
Palmer KJ, Buckley MM, Faulds D (1992) Transdermal nicotine. A review of its pharmacodynamic and pharmacokinetic properties, and therapeutic efficacy as an aid to smoking cessation. Drugs 44:498–529
Perkins KA, Grobe JE, Fonte C, Goettler J, Caggiula AR, Reynolds WA, Stiller RL, Scierka A, Jacob RG (1994) Chronic and acute tolerance to subjective, behavioral and cardiovascular effects of nicotine in humans. J Pharmacol Exp Ther 270:628–638
Perry E, Martin-Ruiz C, Lee M, Griffiths M, Johnson M, Piggott M, Haroutunian V, Daniel Buxbaum J, Nãsland J, Davis K, Gotti C, Clementi F, Tzartos S, Cohen O, Soreq H, Jaros E, Perry R, Ballard C, McKeith I, Court J (2000) Nicotinic receptor subtypes in human brain ageing, Alzheimer and Lewy body diseases. Eur J Pharmacol 393:215–222
Petrovsky N, Quednow BB, Ettinger U, Schmechtig A, Mössner R, Collier DA, Kühn KU, Maier W, Wagner M, Kumari V (2010) Sensorimotor gating is associated with CHRNA3 polymorphisms in schizophrenia and healthy volunteers. Neuropsychopharmacology 35:1429–1439. https://doi.org/10.1038/npp.2010.12
Pickworth WB, Fant RV, Butschky MF, Henningfield JE (1997) Effects of mecamylamine on spontaneous EEG and performance in smokers and non-smokers. Pharmacol Biochem Behav 56:181–187
Raichle ME, MacLeod AM, Snyder AZ, Powers WJ, Gusnard DA, Shulman GL (2001) A default mode of brain function. Proc Natl Acad Sci U S A 98:676–682
Sabri O, Meyer PM, Gräf S, Hesse S, Wilke S, Becker GA, Rullmann M, Patt M, Luthardt J, Wagenknecht G, Hoepping A, Smits R, Franke A, Sattler B, Tiepolt S, Fischer S, Deuther-Conrad W, Hegerl U, Barthel H, Schönknecht P, Brust P (2018) Cognitive correlates of alpha4beta2 nicotinic acetylcholine receptors in mild Alzheimer’s dementia. Brain 141:1840–1854. https://doi.org/10.1093/brain/awy099
Shulman GL, Fiez JA, Corbetta M, Buckner RL, Miezin FM, Raichle ME, Petersen SE (1997) Common blood flow changes across visual tasks: II. Decreases in cerebral cortex. J Cogn Neurosci 9:648–663
Singh A, Das DK, Kelley ME (2006) Mecamylamine (Targacept) IDrugs 9:205–217
Sonuga-Barke EJ, Castellanos FX (2007) Spontaneous attentional fluctuations in impaired states and pathological conditions: a neurobiological hypothesis. Neurosci Biobehav Rev 31:977–986
Stolerman IP, Goldfarb T, Fink R, Jarvik ME (1973) Influencing cigarette smoking with nicotine antagonists. Psychopharmacologia 28:247–259
Stolerman IP, Mirza NR, Shoaib M (1995) Nicotine psychopharmacology: addiction, cognition and neuroadaptation. Med Res Rev 15:47–72
Sutherland MT, Ray KL, Riedel MC, Yanes JA, Stein EA, Laird AR (2015) Neurobiological impact of nicotinic acetylcholine receptor agonists: an activation likelihood estimation meta-analysis of pharmacologic neuroimaging studies. Biol Psychiatry 78:711–720. https://doi.org/10.1016/j.biopsych.2014.12.021
Talairach J, Tournoux P (1988) Co-planar stereotaxic atlas of the human brain. Thieme, New York
Tanabe J, Nyberg E, Martin LF, Martin J, Cordes D, Kronberg E, Tregellas JR (2011) Nicotine effects on default mode network during resting state. Psychopharmacology 216:287–295. https://doi.org/10.1007/s00213-011-2221-8
Tregellas JR, Tanabe J, Rojas DC, Shatti S, Olincy A, Johnson L, Martin LF, Soti F, Kem WR, Leonard S, Freedman R (2011) Effects of an alpha 7-nicotinic agonist on default network activity in schizophrenia. Biol Psychiatry 69:7–11. https://doi.org/10.1016/j.biopsych.2010.07.004
Whitfield-Gabrieli S, Ford JM (2012) Default mode network activity and connectivity in psychopathology. Annu Rev Clin Psychol 8:49–76. https://doi.org/10.1146/annurev-clinpsy-032511-143049
Young JM, Shytle RD, Sanberg PR, George TP (2001) Mecamylamine: new therapeutic uses and toxicity/risk profile. Clin Ther 23:532–565
Funding
This work was funded by the National Institutes of Health grant R21 DA027894 to B. Hahn, and the National Institute on Drug Abuse Intramural Research Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hahn, B., Harvey, A.N., Concheiro-Guisan, M. et al. Nicotinic receptor modulation of the default mode network. Psychopharmacology 238, 589–597 (2021). https://doi.org/10.1007/s00213-020-05711-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-020-05711-9