Abstract
Rationale
Anxiety is one of the most comorbid conditions with major depressive disorder (MDD). Depression-associated anxiety often stems from the dysfunctional hypothalamic-pituitary-adrenal (HPA) axis and its altered regulation by the amygdala. Furthermore, MDD is associated with altered glutamatergic processing leading to anxiety and impaired regulation of the HPA axis. Recent studies have demonstrated that N-acetyl cysteine (NAC), a pleiotropic drug, exerts antidepressant-like effect by modulation of hippocampal functions, periterminal release of glutamate, and/or redox systems. However, the effects of NAC on depression-associated anxiety, HPA axis hyperactivity, and amygdalar dysfunctions are relatively unknown.
Objectives
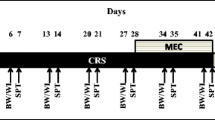
Accordingly, we evaluated the effect of NAC on neonatal clomipramine (CLI)-induced adulthood anxiety and accompanying changes in plasma corticosterone levels, amygdalar volumes, neuronal/glial densities, levels of monoamines, and their metabolites in the amygdalar complex.
Results
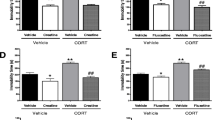
We found that chronic treatment with NAC reverses CLI-induced anhedonia and enhanced anxiety. Interestingly, attenuation of CLI-associated anxiety in NAC-treated rats were accompanied by a reversal of adrenal and spleen hypertrophy, and normalization of enhanced plasma corticosterone levels, indicating improved HPA axis functioning. Furthermore, NAC treatment was sufficient to reverse volumetric hypertrophy of basolateral amygdala (BLA), and altered noradrenaline (NA) metabolism in the amygdalar complex. The effects of NAC in the reversal of CLI-induced impairments were similar to that of fluoxetine (FLX).
Conclusions
We suggest that beneficial effects of NAC on antidepressive- and antianxiety-like behaviors are at least in part mediated via restoration of amygdalar and HPA axis functioning. Our results support the hypothesis that NAC might be evolved as a therapeutic strategy for reversal of amygdalar dysfunction in depression.







Similar content being viewed by others
Data accessibility
This study includes original data. Authors have full access to all data and take responsibility for the integrity of the data and the accuracy of the data analysis. The data that support the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
- 5-HIAA:
-
5-Hydroxyindoleacetic acid
- 5-HT:
-
5-Hydroxytryptamine (serotonin)
- 5-HTT:
-
Serotonin transporter
- BLA:
-
Basolateral nucleus of amygdala
- CeA:
-
Central nucleus of amygdala
- CLI:
-
Clomipramine
- DA:
-
Dopamine
- DOPAC:
-
3,4-Dihydroxyphenylacetic acid
- FLX:
-
Fluoxetine
- FSL:
-
Flinders sensitive line
- HPA axis:
-
Hypothalamic-pituitary-adrenal axis
- HVA:
-
Homovanillic acid
- LA:
-
Lateral nucleus of amygdala
- LC:
-
Locus coeruleus
- MDD:
-
Major depressive disorder
- MHPG:
-
3-Methoxy-4-hydroxyphenylglycol
- NA:
-
Noradrenaline
- NAC:
-
N-acetyl cysteine
- NSFT:
-
Novelty-suppressed feeding test
- PFC:
-
Prefrontal cortex
- PVN:
-
Paraventricular nucleus
- SPT:
-
Sucrose preference test
- VCT:
-
Vogel’s conflict test
References
Abhijit S, Tripathi SJ, Shankaranarayana Rao BS, Asha Devi S (2019) Grape seed proanthocyanidin extract and swimming training enhances neuronal number in dorso-medial prefrontal cortex in middle-aged male rats by alleviating oxidative stress. J Funct Foods 64:103693. https://doi.org/10.1016/j.jff.2019.103693
Adhikari A, Lerner TN, Finkelstein J, Pak S, Jennings JH, Davidson TJ, Ferenczi E, Gunaydin LA, Mirzabekov JJ, Ye L, Kim SY, Lei A, Deisseroth K (2015) Basomedial amygdala mediates top-down control of anxiety and fear. Nature 527:179–185. https://doi.org/10.1038/nature15698
Al-Samhari MM, Al-Rasheed NM, Al-Rejaie S, Al-Rasheed NM, Hasan IH, Mahmoud AM, Dzimiri N (2016) Possible involvement of the JAK/STAT signaling pathway in N-acetylcysteine-mediated antidepressant-like effects. Exp Biol Med (Maywood) 241:509–518. https://doi.org/10.1177/1535370215619707
Berry A, Bellisario V, Panetta P, Raggi C, Magnifico MC, Arese M, Cirulli F (2018) Administration of the antioxidant N-acetyl-cysteine in pregnant mice has long-term positive effects on metabolic and behavioral endpoints of male and female offspring prenatally exposed to a high-fat diet. Front Behav Neurosci 12:48. https://doi.org/10.3389/fnbeh.2018.00048
Bonilla-Jaime H, Retana-Marquez S, Arteaga-Silva M, Hernandez-Gonzalez M, Vazquez-Palacios G (2010) Circadian activity of corticosterone in an animal model of depression: response to muscarinic cholinergic stimulation. Physiol Behav 100:311–315. https://doi.org/10.1016/j.physbeh.2010.03.002
Caspi A, Sugden K, Moffitt TE, Taylor A, Craig IW, Harrington H, McClay J, Mill J, Martin J, Braithwaite A, Poulton R (2003) Influence of life stress on depression: moderation by a polymorphism in the 5-HTT gene. Science 301:386–389. https://doi.org/10.1126/science.1083968
Chakraborty S, Tripathi SJ, Srikumar BN, Raju TR, Shankaranarayana Rao BS (2019) Chronic brain stimulation rewarding experience ameliorates depression-induced cognitive deficits and restores aberrant plasticity in the prefrontal cortex. Brain Stimul 12:752–766. https://doi.org/10.1016/j.brs.2019.01.020
Chakraborty S, Tripathi SJ, Srikumar BN, Raju TR, Shankaranarayana Rao BS (2020) N-acetyl cysteine ameliorates depression-induced cognitive deficits by restoring the volumes of hippocampal subfields and associated neurochemical changes. Neurochem Int 132:104605. https://doi.org/10.1016/j.neuint.2019.104605
de Winter RF, van Hemert AM, DeRijk RH, Zwinderman KH, Frankhuijzen-Sierevogel AC, Wiegant VM, Goekoop JG (2003) Anxious-retarded depression: relation with plasma vasopressin and cortisol. Neuropsychopharmacology 28:140–147. https://doi.org/10.1038/sj.npp.1300002
Dean O, Giorlando F, Berk M (2011) N-Acetylcysteine in psychiatry: current therapeutic evidence and potential mechanisms of action. J Psychiatry Neurosci 36:78–86. https://doi.org/10.1503/jpn.100057
Dienes KA, Hazel NA, Hammen CL (2013) Cortisol secretion in depressed, and at-risk adults. Psychoneuroendocrinology 38:927–940. https://doi.org/10.1016/j.psyneuen.2012.09.019
Drevets WC, Price JL, Bardgett ME, Reich T, Todd RD, Raichle ME (2002) Glucose metabolism in the amygdala in depression: relationship to diagnostic subtype and plasma cortisol levels. Pharmacol Biochem Behav 71:431–447. https://doi.org/10.1016/s0091-3057(01)00687-6
Fava M, Rush AJ, Alpert JE, Balasubramani GK, Wisniewski SR, Carmin CN, Biggs MM, Zisook S, Leuchter A, Howland R, Warden D, Trivedi MH (2008) Difference in treatment outcome in outpatients with anxious versus nonanxious depression: a STAR*D report. Am J Psychiatry 165:342–351. https://doi.org/10.1176/appi.ajp.2007.06111868
Frodl T, Meisenzahl E, Zetzsche T, Bottlender R, Born C, Groll C, Jager M, Leinsinger G, Hahn K, Moller HJ (2002) Enlargement of the amygdala in patients with a first episode of major depression. Biol Psychiatry 51:708–714. https://doi.org/10.1016/s0006-3223(01)01359-2
Gaffrey MS, Barch DM, Bogdan R, Farris K, Petersen SE, Luby JL (2018) Amygdala reward reactivity mediates the association between preschool stress response and depression severity. Biol Psychiatry 83:128–136. https://doi.org/10.1016/j.biopsych.2017.08.020
Gillespie CF, Nemeroff CB (2005) Hypercortisolemia and depression. Psychosom Med 67(Suppl 1):S26–S28. https://doi.org/10.1097/01.psy.0000163456.22154.d2
Gold PW, Chrousos GP (2002) Organization of the stress system and its dysregulation in melancholic and atypical depression: high vs low CRH/NE states. Mol Psychiatry 7:254–275. https://doi.org/10.1038/sj.mp.4001032
Gotlib IH, Joormann J, Minor KL, Hallmayer J (2008) HPA axis reactivity: a mechanism underlying the associations among 5-HTTLPR, stress, and depression. Biol Psychiatry 63:847–851. https://doi.org/10.1016/j.biopsych.2007.10.008
Gundersen HJ, Jensen EB (1987) The efficiency of systematic sampling in stereology and its prediction. J Microsc 147:229–263. https://doi.org/10.1111/j.1365-2818.1987.tb02837.x
Gundersen HJ, Jensen EB, Kieu K, Nielsen J (1999) The efficiency of systematic sampling in stereology-reconsidered. J Microsc 193:199–211. https://doi.org/10.1046/j.1365-2818.1999.00457.x
Hariri AR, Mattay VS, Tessitore A, Kolachana B, Fera F, Goldman D, Egan MF, Weinberger DR (2002) Serotonin transporter genetic variation and the response of the human amygdala. Science 297:400–403. https://doi.org/10.1126/science.1071829
Holmes A, Murphy DL, Crawley JN (2003) Abnormal behavioral phenotypes of serotonin transporter knockout mice: parallels with human anxiety and depression. Biol Psychiatry 54:953–959. https://doi.org/10.1016/j.biopsych.2003.09.003
Holsboer F (2000) The corticosteroid receptor hypothesis of depression. Neuropsychopharmacology 23:477–501. https://doi.org/10.1016/S0893-133X (00)00159-7
Keller J, Gomez R, Williams G, Lembke A, Lazzeroni L, Murphy GM Jr, Schatzberg AF (2017) HPA axis in major depression: cortisol, clinical symptomatology and genetic variation predict cognition. Mol Psychiatry 22:527–536. https://doi.org/10.1038/mp.2016.120
Kobiella A, Reimold M, Ulshöfer DE, Ikonomidou VN, Vollmert C, Vollstädt-Klein S, Rietschel M, Reischl G, Heinz A, Smolka MN (2011) How the serotonin transporter 5-HTTLPR polymorphism influences amygdala function: the roles of in vivo serotonin transporter expression and amygdala structure. Transl Psychiatry 1:e37–e37. https://doi.org/10.1038/tp.2011.29
Korou LM, Agrogiannis G, Koros C, Kitraki E, Vlachos IS, Tzanetakou I, Karatzas T, Pergialiotis V, Dimitroulis D, Perrea DN (2014) Impact of N-acetylcysteine and sesame oil on lipid metabolism and hypothalamic-pituitary-adrenal axis homeostasis in middle-aged hypercholesterolemic mice. Sci Rep 4:6806. https://doi.org/10.1038/srep06806
Krystal JH, Sanacora G, Duman RS (2013) Rapid-acting glutamatergic antidepressants: the path to ketamine and beyond. Biol Psychiatry 73:1133–1141. https://doi.org/10.1016/j.biopsych.2013.03.026
Lamers F, van Oppen P, Comijs HC, Smit JH, Spinhoven P, van Balkom AJ, Nolen WA, Zitman FG, Beekman AT, Penninx BW (2011) Comorbidity patterns of anxiety and depressive disorders in a large cohort study: the Netherlands study of depression and anxiety (NESDA). J Clin Psychiatry 72:341–348. https://doi.org/10.4088/JCP.10m06176blu
Li Q, Wichems C, Heils A, Van De Kar LD, Lesch KP, Murphy DL (1999) Reduction of 5-hydroxytryptamine (5-HT)(1A)-mediated temperature and neuroendocrine responses and 5-HT(1A) binding sites in 5-HT transporter knockout mice. J Pharmacol Exp Ther 291:999–1007
Limon-Morales O, Soria-Fregozo C, Arteaga-Silva M, Vazquez-Palacios G, Bonilla-Jaime H (2014) Altered expression of 5-HT1A receptors in adult rats induced by neonatal treatment with clomipramine. Physiol Behav 124:37–44. https://doi.org/10.1016/j.physbeh.2013.10.026
Mahati K, Bhagya V, Christofer T, Sneha A, Shankaranarayana Rao BS (2016) Enriched environment ameliorates depression-induced cognitive deficits and restores abnormal hippocampal synaptic plasticity. Neurobiol Learn Mem 134(Pt B):379–391. https://doi.org/10.1016/j.nlm.2016.08.017
McCall JG, Siuda ER, Bhatti DL, Lawson LA, McElligott ZA, Stuber GD, Bruchas MR (2017) Locus coeruleus to basolateral amygdala noradrenergic projections promote anxiety-like behavior. Elife 6:e18247. https://doi.org/10.7554/eLife.18247
Millan MJ (2006) Multi-target strategies for the improved treatment of depressive states: conceptual foundations and neuronal substrates, drug discovery and therapeutic application. Pharmacol Ther 110:135–370. https://doi.org/10.1016/j.pharmthera.2005.11.006
Mitra R, Jadhav S, McEwen BS, Vyas A, Chattarji S (2005) Stress duration modulates the spatiotemporal patterns of spine formation in the basolateral amygdala. Proc Natl Acad Sci U S A 102:9371–9376. https://doi.org/10.1073/pnas.0504011102
Mocelin R, Marcon M, D'Ambros S, Mattos J, Sachett A, Siebel AM, Herrmann AP, Piato A (2018) N-Acetylcysteine reverses anxiety and oxidative damage induced by unpredictable chronic stress in zebrafish. Mol Neurobiol 56:1188–1195. https://doi.org/10.1007/s12035-018-1165-y
Molina-Jimenez T, Limon-Morales O, Bonilla-Jaime H (2018) Early postnatal treatment with clomipramine induces female sexual behavior and estrous cycle impairment. Pharmacol Biochem Behav 166:27–34. https://doi.org/10.1016/j.pbb.2018.01.004
Nasca C, Bigio B, Zelli D, de Angelis P, Lau T, Okamoto M, Soya H, Ni J, Brichta L, Greengard P, Neve RL, Lee FS, McEwen BS (2017) Role of the astroglial glutamate exchanger xCT in ventral hippocampus in resilience to stress. Neuron 96:402–413.e5. https://doi.org/10.1016/j.neuron.2017.09.020
Nierenberg AA, Husain MM, Trivedi MH, Fava M, Warden D, Wisniewski SR, Miyahara S, Rush AJ (2010) Residual symptoms after remission of major depressive disorder with citalopram and risk of relapse: a STAR*D report. Psychol Med 40:41–50. https://doi.org/10.1017/S0033291709006011
Nietzer SL, Bonn M, Jansen F, Heiming RS, Lewejohann L, Sachser N, Asan ES, Lesch KP, Schmitt AG (2011) Serotonin transporter knockout and repeated social defeat stress: impact on neuronal morphology and plasticity in limbic brain areas. Behav Brain Res 220:42–54. https://doi.org/10.1016/j.bbr.2011.01.011
Pacak K, Armando I, Fukuhara K, Kvetnansky R, Palkovits M, Kopin IJ, Goldstein DS (1992) Noradrenergic activation in the paraventricular nucleus during acute and chronic immobilization stress in rats: an in vivo microdialysis study. Brain Res 589:91–96. https://doi.org/10.1016/0006-8993(92)91165-b
Pagliaccio D, Luby JL, Bogdan R, Agrawal A, Gaffrey MS, Belden AC, Botteron KN, Harms MP, Barch DM (2015) Amygdala functional connectivity, HPA axis genetic variation, and life stress in children and relations to anxiety and emotion regulation. J Abnorm Psychol 124:817–833. https://doi.org/10.1037/abn0000094
Park J, Moghaddam B (2017) Impact of anxiety on prefrontal cortex encoding of cognitive flexibility. Neuroscience 345:193–202. https://doi.org/10.1016/j.neuroscience.2016.06.013
Pawlak R, Magarinos AM, Melchor J, McEwen B, Strickland S (2003) Tissue plasminogen activator in the amygdala is critical for stress-induced anxiety-like behavior. Nat Neurosci 6:168–174. https://doi.org/10.1038/nn998
Pawlak R, Shankaranarayana Rao BS, Melchor JP, Chattarji S, McEwen B, Strickland S (2005) Tissue plasminogen activator and plasminogen mediate stress-induced decline of neuronal and cognitive functions in the mouse hippocampus. Proc Natl Acad Sci U S A 102:18201–18206. https://doi.org/10.1073/pnas.0509232102
Paxinos G, Watson C (2006) The rat brain in stereotaxic coordinates. Academic Press, Amsterdam
Piwowarska J, Chimiak A, Matsumoto H, Dziklinska A, Radziwon-Zaleska M, Szelenberger W, Pachecka J (2012) Serum cortisol concentration in patients with major depression after treatment with fluoxetine. Psychiatry Res 198:407–411. https://doi.org/10.1016/j.psychres.2012.01.029
Pizzagalli DA (2014) Depression, stress, and anhedonia: toward a synthesis and integrated model. Annu Rev Clin Psychol 10:393–423. https://doi.org/10.1146/annurev-clinpsy-050212-185606
Prathiba J, Kumar KB, Karanth KS (1998) Hyperactivity of hypothalamic pituitary axis in neonatal clomipramine model of depression. J Neural Transm (Vienna) 105:1335–1339. https://doi.org/10.1007/s007020050135
Rei D, Mason X, Seo J, Graff J, Rudenko A, Wang J, Rueda R, Siegert S, Cho S, Canter RG, Mungenast AE, Deisseroth K, Tsai LH (2015) Basolateral amygdala bidirectionally modulates stress-induced hippocampal learning and memory deficits through a p25/Cdk5-dependent pathway. Proc Natl Acad Sci U S A 112:7291–7296. https://doi.org/10.1073/pnas.1415845112
Roozendaal B (2003) Systems mediating acute glucocorticoid effects on memory consolidation and retrieval. Prog Neuro-Psychopharmacol Biol Psychiatry 27:1213–1223. https://doi.org/10.1016/j.pnpbp.2003.09.015
Sheline YI, Barch DM, Donnelly JM, Ollinger JM, Snyder AZ, Mintun MA (2001) Increased amygdala response to masked emotional faces in depressed subjects resolves with antidepressant treatment: an fMRI study. Biol Psychiatry 50:651–658. https://doi.org/10.1016/S0006-3223(01)01263-X
Siuda ER, Al-Hasani R, McCall JG, Bhatti DL, Bruchas MR (2016) Chemogenetic and optogenetic activation of gas signaling in the basolateral amygdala induces acute and social anxiety-like states. Neuropsychopharmacology 41:2011–2023. https://doi.org/10.1038/npp.2015.371
Strawn JR, Saldana SN (2012) Treatment with adjunctive N-acetylcysteine in an adolescent with selective serotonin reuptake inhibitor-resistant anxiety. J Child Adolesc Psychopharmacol 22:472–473. https://doi.org/10.1089/cap.2012.0066
Surget A, Tanti A, Leonardo ED, Laugeray A, Rainer Q, Touma C, Palme R, Griebel G, Ibarguen-Vargas Y, Hen R, Belzung C (2011) Antidepressants recruit new neurons to improve stress response regulation. Mol Psychiatry 16:1177–1188. https://doi.org/10.1038/mp.2011.48
Tripathi SJ, Chakraborty S, Srikumar BN, Raju TR, Shankaranarayana Rao BS (2017) Inactivation of basolateral amygdala prevents chronic immobilization stress-induced memory impairment and associated changes in corticosterone levels. Neurobiol Learn Mem 142:218–229. https://doi.org/10.1016/j.nlm.2017.05.005
Tripathi SJ, Chakraborty S, Srikumar BN, Raju TR, Shankaranarayana Rao BS (2019a) Basolateral amygdalar inactivation blocks chronic stress-induced lamina-specific reduction in prefrontal cortex volume and associated anxiety-like behavior. Prog Neuro-Psychopharmacol Biol Psychiatry 88:194–207. https://doi.org/10.1016/j.pnpbp.2018.07.016
Tripathi SJ, Chakraborty S, Srikumar BN, Raju TR, Shankaranarayana Rao BS (2019b) Inactivation of basolateral amygdala prevents stress-induced astroglial loss in the prefrontal cortex. Mol Neurobiol 56:350–366. https://doi.org/10.1007/s12035-018-1057-1
Tripathi SJ, Chakraborty S, Srikumar BN, Raju TR, Shankaranarayana Rao BS (2019c) Prevention of chronic immobilization stress-induced enhanced expression of glucocorticoid receptors in the prefrontal cortex by inactivation of basolateral amygdala. J Chem Neuroanat 95:134–145. https://doi.org/10.1016/j.jchemneu.2017.12.006
Ulrich-Lai YM, Herman JP (2009) Neural regulation of endocrine and autonomic stress responses. Nat Rev Neurosci 10:397–409. https://doi.org/10.1038/nrn2647
van der Doelen RH, Deschamps W, D'Annibale C, Peeters D, Wevers RA, Zelena D, Homberg JR, Kozicz T (2014) Early life adversity and serotonin transporter gene variation interact at the level of the adrenal gland to affect the adult hypothalamo-pituitary-adrenal axis. Transl Psychiatry 4:e409. https://doi.org/10.1038/tp.2014.57
Vogel G, Neill D, Hagler M, Kors D (1990) A new animal model of endogenous depression: a summary of present findings. Neurosci Biobehav Rev 14:85–91. https://doi.org/10.1016/s0149-7634(05)80164-2
Vreeburg SA, Hoogendijk WJ, van Pelt J, Derijk RH, Verhagen JC, van Dyck R, Smit JH, Zitman FG, Penninx BW (2009) Major depressive disorder and hypothalamic-pituitary-adrenal axis activity: results from a large cohort study. Arch Gen Psychiatry 66:617–626. https://doi.org/10.1001/archgenpsychiatry.2009.50
Vyas A, Mitra R, Shankaranarayana Rao BS, Chattarji S (2002) Chronic stress induces contrasting patterns of dendritic remodeling in hippocampal and amygdaloid neurons. J Neurosci 22:6810–6818 20026655
Wang B, Wang Y, Wu Q, Huang HP, Li S (2017) Effects of alpha2A adrenoceptors on norepinephrine secretion from the locus coeruleus during chronic stress-induced depression. Front Neurosci 11:243. https://doi.org/10.3389/fnins.2017.00243
Warden D, Rush AJ, Trivedi MH, Fava M, Wisniewski SR (2007) The STAR*D Project results: a comprehensive review of findings. Curr Psychiatry Rep 9:449–459. https://doi.org/10.1007/s11920-007-0061-3
Wei J, Zhong P, Qin L, Tan T, Yan Z (2017) Chemicogenetic restoration of the prefrontal cortex to amygdala pathway ameliorates stress-induced deficits. Cereb Cortex 1:1–11. https://doi.org/10.1093/cercor/bhx104
Willner P, Mitchell PJ (2002) The validity of animal models of predisposition to depression. Behav Pharmacol 13:169–188. https://doi.org/10.1097/00008877-200205000-00001
Wong ML, Kling MA, Munson PJ, Listwak S, Licinio J, Prolo P, Karp B, McCutcheon IE, Geracioti TD Jr, DeBellis MD, Rice KC, Goldstein DS, Veldhuis JD, Chrousos GP, Oldfield EH, McCann SM, Gold PW (2000) Pronounced and sustained central hypernoradrenergic function in major depression with melancholic features: relation to hypercortisolism and corticotropin-releasing hormone. Proc Natl Acad Sci U S A 97:325–330. https://doi.org/10.1073/pnas.97.1.325
Wright DJ, Renoir T, Smith ZM, Frazier AE, Francis PS, Thorburn DR, McGee SL, Hannan AJ, Gray LJ (2015) N-Acetylcysteine improves mitochondrial function and ameliorates behavioral deficits in the R6/1 mouse model of Huntington’s disease. Transl Psychiatry 5:e492. https://doi.org/10.1038/tp.2014.131
Wright DJ, Gray LJ, Finkelstein DI, Crouch PJ, Pow D, Pang TY, Li S, Smith ZM, Francis PS, Renoir T, Hannan AJ (2016) N-Acetylcysteine modulates glutamatergic dysfunction and depressive behavior in Huntington’s disease. Hum Mol Genet 25:2923–2933. https://doi.org/10.1093/hmg/ddw144
Yavari P, Vogel GW, Neill DB (1993) Decreased raphe unit activity in a rat model of endogenous depression. Brain Res 611:31–36. https://doi.org/10.1016/0006-8993(93)91773-L
Young EA, Abelson JL, Cameron OG (2004) Effect of comorbid anxiety disorders on the hypothalamic-pituitary-adrenal axis response to a social stressor in major depression. Biol Psychiatry 56:113–120. https://doi.org/10.1016/j.biopsych.2004.03.017
Zhu XH, Yan HC, Zhang J, Qu HD, Qiu XS, Chen L, Li SJ, Cao X, Bean JC, Chen LH, Qin XH, Liu JH, Bai XC, Mei L, Gao TM (2010) Intermittent hypoxia promotes hippocampal neurogenesis and produces antidepressant-like effects in adult rats. J Neurosci 30:12653–12663. https://doi.org/10.1523/JNEUROSCI.6414-09.2010
Funding
Suwarna Chakraborty was supported by a Senior Research Fellowship from Indian Council of Medical Research (ICMR; 3/1/2/74/Neuro/2018-NCD-I), New Delhi, India. Sunil Jamuna Tripathi was supported by Research Associate Fellowship (45/04/2018-ANA-BMS) from the ICMR, New Delhi, India. We received financial support from the Department of Biotechnology (DBT), Government of India, New Delhi, and the National Institute of Mental Health and Neuro Sciences (NIMHANS), Bengaluru, India. Funding agencies have no role in study design, collection, analysis, and interpretation of data and writing the reports.
Author information
Authors and Affiliations
Contributions
SC, TRR, and BSSR conceptualized and designed the experiments; SC and SJT performed the experiments and analyzed the data; SC, SJT, TRR, and BSSR wrote the manuscript.
Corresponding author
Ethics declarations
All experiments comply with the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals, Government of India.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chakraborty, S., Tripathi, S.J., Raju, T.R. et al. Mechanisms underlying remediation of depression-associated anxiety by chronic N-acetyl cysteine treatment. Psychopharmacology 237, 2967–2981 (2020). https://doi.org/10.1007/s00213-020-05585-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-020-05585-x