Abstract
Rationale
Patients diagnosed with schizophrenia typically receive life-long treatments with antipsychotic drugs (APDs). However, the impact of chronic APDs treatment on neuroplastic mechanisms in the brain remains largely elusive.
Objective
Here, we focused on blonanserin, a second-generation antipsychotic (SGA) that acts as an antagonist at dopamine D2, D3, and serotonin 5-HT2A receptors, and represents an important tool for the treatment of schizophrenia.
Methods
We used rats to investigate the ability of chronic treatment blonanserin to modulate the activity of brain structures relevant for schizophrenia, under baseline conditions or in response to an acute forced swim session (FSS). We measured the expression of different immediate early genes (IEGs), including c-Fos, Arc/Arg 3.1, Zif268 and Npas4.
Results
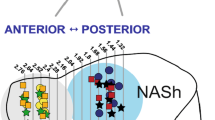
Blonanserin per se produced limited changes in the expression of these genes under basal conditions, while, as expected, FSS produced a significant elevation of IEGs transcription in different brain regions. The response of blonanserin-treated rats to FSS show anatomical and gene-selective differences. Indeed, the upregulation of IEGs was greatly reduced in the striatum, a brain structure enriched in dopamine receptors, whereas the upregulation of some genes (Zif268, Npas4) was largely preserved in other regions, such as the prefrontal cortex and the ventral hippocampus.
Conclusions
Taken together, our findings show that chronic exposure to blonanserin modulates selective IEGs with a specific anatomical profile. Moreover, the differential activation of specific brain regions under challenging conditions may contribute to specific clinical features of the drug.






Similar content being viewed by others
Bibliography
Aringhieri S, Carli M, Kolachalam S, Verdesca V, Cini E, Rossi M et al (2018) Molecular targets of atypical antipsychotics: from mechanism of action to clinical differences. Pharmacol Ther. https://doi.org/10.1016/j.pharmthera.2018.06.012
Benito E, Barco A (2015) The neuronal activity-driven Transcriptome. Mol Neurobiol. https://doi.org/10.1007/s12035-014-8772-z
Bersten DC, Bruning JB, Peet DJ, Whitelaw ML (2014) Human variants in the neuronal basic helix-loop-helix/per-ARNT-Sim (bHLH/PAS) transcription factor complex NPAS4/ARNT2 disrupt function. PLoS One. https://doi.org/10.1371/journal.pone.0085768
Bramham CR, Worley PF, Moore MJ, Guzowski JF (2008) The immediate early gene arc/Arg3.1: regulation, mechanisms, and function. In Journal of Neuroscience. https://doi.org/10.1523/JNEUROSCI.3864-08.2008
Brenner K, Liu A, Laplante DP, Lupien S, Pruessner JC, Ciampi A et al (2009) Cortisol response to a psychosocial stressor in schizophrenia: blunted, delayed, or normal? Psychoneuroendocrinology. https://doi.org/10.1016/j.psyneuen.2009.01.002
Brivio P, Corsini G, Riva MA, Calabrese F (2019) Chronic vortioxetine treatment improves the responsiveness to an acute stress acting through the ventral hippocampus in a glucocorticoid-dependent way. Pharmacol Res. https://doi.org/10.1016/j.phrs.2019.02.006
Buijs RM, Van Eden CG (2000) The integration of stress by the hypothalamus, amygdala and prefrontal cortex: balance between the autonomic nervous system and the neuroendocrine system. In Progress in Brain Research. https://doi.org/10.1016/S0079-6123(00)26011-1
Cai HL, Jiang P, Tan QY, Dang RL, Tang MM, Xue Y, Deng Y, Zhang BK, Fang PF, Xu P, Xiang DX, Li HD, Yao JK (2017) Therapeutic efficacy of atypical antipsychotic drugs by targeting multiple stress-related metabolic pathways. Transl Psychiatry 7. https://doi.org/10.1038/tp.2017.94
Cattaneo A, Cattane N, Begni V, Pariante CM, Riva MA (2016) The human BDNF gene: peripheral gene expression and protein levels as biomarkers for psychiatric disorders. Transl Psychiatry 6. https://doi.org/10.1038/tp.2016.214
Chawla MK, Guzowski JF, Ramirez-Amaya V, Lipa P, Hoffman KL, Marriott LK et al (2005) Sparse, environmentally selective expression of arc RNA in the upper blade of the rodent fascia dentata by brief spatial experience. Hippocampus. https://doi.org/10.1002/hipo.20091
Coutellier L, Beraki S, Ardestani PM, Saw NL, Shamloo M (2012) Npas4: a neuronal transcription factor with a key role in social and cognitive functions relevant to developmental disorders. PLoS One. https://doi.org/10.1371/journal.pone.0046604
de Bartolomeis A, Buonaguro EF, Latte G, Rossi R, Marmo F, Iasevoli F, Tomasetti C (2017) Immediate-early genes modulation by Antipsychotics: Translational Implications for a Putative Gateway to Drug-Induced Long-Term Brain Changes. Front Behav Neurosci. https://doi.org/10.3389/fnbeh.2017.00240
de Kloet ER, Joëls M, Holsboer F (2005) Stress and the brain: from adaptation to disease. Nat Rev Neurosci. https://doi.org/10.1038/nrn1683
Fanselow MS, Dong HW (2010) Are the dorsal and ventral Hippocampus functionally distinct structures? Neuron. https://doi.org/10.1016/j.neuron.2009.11.031
Fumagalli F, Bedogni F, Frasca A, Di Pasquale L, Racagni G, Riva MA (2006) Corticostriatal up-regulation of activity-regulated cytoskeletal-associated protein expression after repeated exposure to cocaine. Mol Pharmacol. https://doi.org/10.1124/mol.106.026302
Fumagalli F, Calabrese F, Luoni A, Bolis F, Racagni G, Riva MA (2012) Modulation of BDNF expression by repeated treatment with the novel antipsychotic lurasidone under basal condition and in response to acute stress. Int J Neuropsychopharmacol. https://doi.org/10.1017/S1461145711000150
Gallo FT, Katche C, Morici JF, Medina JH, Weisstaub NV (2018) Immediate early genes, memory and psychiatric disorders: focus on c-Fos, Egr1 and arc. Front Behav Neurosci. https://doi.org/10.3389/fnbeh.2018.00079
Guilloux JP, Seney M, Edgar N, Sibille E (2011) Integrated behavioral z-scoring increases the sensitivity and reliability of behavioral phenotyping in mice: relevance to emotionality and sex. J Neurosci Methods. https://doi.org/10.1016/j.jneumeth.2011.01.019
Hermans EJ, Henckens MJAG, Joëls M, Fernández G (2014) Dynamic adaptation of large-scale brain networks in response to acute stressors. Trends Neurosci. https://doi.org/10.1016/j.tins.2014.03.006
Homberg JR, Kozicz T, Fernández G (2017) Large-scale network balances in the transition from adaptive to maladaptive stress responses. Curr Opin Behav Sci. https://doi.org/10.1016/j.cobeha.2016.11.003
Kishi T, Matsuda Y, Nakamura H, Iwata N (2013) Blonanserin for schizophrenia: systematic review and meta-analysis of double-blind, randomized, controlled trials. J Psychiatr Res 47(2):149–154. https://doi.org/10.1016/j.jpsychires.2012.10.011
Kishi T, Matsui Y, Matsuda Y, Katsuki A, Hori H, Yanagimoto H et al (2019) Efficacy, tolerability, and safety of Blonanserin in schizophrenia: an updated and extended systematic review and meta-analysis of randomized controlled trials. Pharmacopsychiatry 52(2):52–62. https://doi.org/10.1055/a-0574-0088
Korb E, Finkbeiner S (2011) Arc in synaptic plasticity: from gene to behavior. Trends Neurosci. https://doi.org/10.1016/j.tins.2011.08.007
Lin Y, Bloodgood BL, Hauser JL, Lapan AD, Koon AC, Kim TK et al (2008) Activity-dependent regulation of inhibitory synapse development by Npas4. Nature. https://doi.org/10.1038/nature07319
Luoni A, Berry A, Calabrese F, Capoccia S, Bellisario V, Gass P et al (2014) Delayed BDNF alterations in the prefrontal cortex of rats exposed to prenatal stress: preventive effect of lurasidone treatment during adolescence. Eur Neuropsychopharmacol 24(6):986–995. https://doi.org/10.1016/j.euroneuro.2013.12.010
Luoni A, Fumagalli F, Racagni G, Riva MA (2014b) Repeated aripiprazole treatment regulates Bdnf, arc and Npas4 expression under basal condition as well as after an acute swim stress in the rat brain. Pharmacol Res. https://doi.org/10.1016/j.phrs.2013.11.008
MacDowell KS, Caso JR, Martín-Hernández D, Moreno BM, Madrigal JLM, Micó JA, Leza JC, García-Bueno B (2016) The atypical antipsychotic paliperidone regulates endogenous antioxidant/anti-inflammatory pathways in rat models of acute and chronic restraint stress. Neurotherapeutics. 13:833–843. https://doi.org/10.1007/s13311-016-0438-2
McEwen BS, Nasca C, Gray JD (2016) Stress effects on neuronal Structure: Hippocampus, Amygdala, and Prefrontal Cortex. Neuropsychopharmacology. https://doi.org/10.1038/npp.2015.171
Molendijk ML, de Kloet ER (2019) Coping with the forced swim stressor: current state-of-the-art. Behav Brain Res. https://doi.org/10.1016/j.bbr.2019.02.005
Molteni R, Calabrese F, Cattaneo A, Mancini M, Gennarelli M, Racagni G, Riva MA (2009) Acute stress responsiveness of the neurotrophin bdnf in the rat hippocampus is modulated by chronic treatment with the antidepressant duloxetine. Neuropsychopharmacology. https://doi.org/10.1038/npp.2008.208
Ohno Y, Okano M, Imaki J, Tatara A, Okumura T, Shimizu S (2010) Atypical antipsychotic properties of blonanserin, a novel dopamine D2 and 5-HT2A antagonist. Pharmacol Biochem Behav 96(2):175–180. https://doi.org/10.1016/j.pbb.2010.04.027
Page CE, Alexander J, Shepard R, Coutellier L (2018) Npas4 deficiency interacts with adolescent stress to disrupt prefrontal GABAergic maturation and adult cognitive flexibility. Genes Brain Behav. https://doi.org/10.1111/gbb.12459
Paxinos G, Watson C (2007) The rat brain in stereotaxic coordinates sixth edition. Elsevier Academic Press
Pérez-Cadahía B, Drobic B, Davie JR (2011) Activation and function of immediate-early genes in the nervous system. Biochem Cell Biol. https://doi.org/10.1139/O10-138
Pintchovski SA, Peebles CL, Hong JK, Verdin E, Finkbeiner S (2009) The serum response factor and a putative novel transcription factor regulate expression of the immediate-early gene arc/Arg3.1 in neurons. J Neurosci. https://doi.org/10.1523/JNEUROSCI.5575-08.2009
Qin S, Hermans EJ, van Marle HJF, Luo J, Fernández G (2009) Acute psychological stress reduces working memory-related activity in the dorsolateral prefrontal cortex. Biol Psychiatry. https://doi.org/10.1016/j.biopsych.2009.03.006
Raio CM, Phelps EA (2015) The influence of acute stress on the regulation of conditioned fear. Neurobiol Stress. https://doi.org/10.1016/j.ynstr.2014.11.004
Sakuma K, Komatsu H, Maruyama M, Imaichi S, Habata Y, Mori M (2015) Temporal and spatial transcriptional fingerprints by antipsychotic or propsychotic drugs in mouse brain. PLoS One. https://doi.org/10.1371/journal.pone.0118510
Sarantis K, Antoniou K, Matsokis N, Angelatou F (2012) Exposure to novel environment is characterized by an interaction of D1/NMDA receptors underlined by phosphorylation of the NMDA and AMPA receptor subunits and activation of ERK1/2 signaling, leading to epigenetic changes and gene expression in rat hippoca. Neurochem Int. https://doi.org/10.1016/j.neuint.2011.10.018
Sawaguchi T (2000) The role of D1-dopamine receptors in working memory-guided movements mediated by frontal cortical areas. Parkinsonism and Related Disorders. https://doi.org/10.1016/S1353-8020(00)00044-4
Shepard R, Heslin K, Hagerdorn P, Coutellier L (2019) Downregulation of Npas4 in parvalbumin interneurons and cognitive deficits after neonatal NMDA receptor blockade: relevance for schizophrenia. Transl Psychiatry 9:1–11. https://doi.org/10.1038/s41398-019-0436-3
Sinha R, Lacadie CM, Constable RT, Seo D (2016) Dynamic neural activity during stress signals resilient coping. Proc Natl Acad Sci U S A. https://doi.org/10.1073/pnas.1600965113
Tateno A, Sakayori T, Kim, W. chan, Honjo, K., Nakayama, H., Arakawa, R., & Okubo, Y. (2018) Comparison of dopamine D 3 and D 2 receptor occupancies by a single dose of blonanserin in healthy subjects: a positron emission tomography study with [ 11 C]-(+)-PHNO. Int J Neuropsychopharmacol. https://doi.org/10.1093/ijnp/pyy004
Tenjin T, Miyamoto S, Ninomiya Y, Kitajima R, Ogino S, Miyake N, Yamaguchi N (2013) Profile of blonanserin for the treatment of schizophrenia. Neuropsychiatr Dis Treat. https://doi.org/10.2147/NDT.S34433
Van Os J, Kenis G, Rutten BPF (2010) The environment and schizophrenia. Nature. https://doi.org/10.1038/nature09563
Weinberger DR, Berman KF, Daniel DG (1992) Mesoprefrontal cortical dopaminergic activity and prefrontal hypofunction in schizophrenia. Clin Neuropharmacol
Yang B, Ren Q, Zhang JC, Chen QX, Hashimoto K (2017) Altered expression of BDNF, BDNF pro-peptide and their precursor proBDNF in brain and liver tissues from psychiatric disorders: Rethinking the brain-liver axis. Translational Psychiatry 7. https://doi.org/10.1038/tp.2017.95
Ying SW, Futter M, Rosenblum K, Webber MJ, Hunt SP, Bliss TVP, Bramham CR (2002) Brain-derived neurotrophic factor induces long-term potentiation in intact adult hippocampus: requirement for ERK activation coupled to CREB and upregulation of arc synthesis. J Neurosci
Yuen EY, Liu W, Karatsoreos IN, Feng J, McEwen BS, Yan Z (2009) Acute stress enhances glutamatergic transmission in prefrontal cortex and facilitates working memory. Proc Natl Acad Sci U S A. https://doi.org/10.1073/pnas.0906791106
Funding
This research has been supported by grants from the Italian Ministry of University and Research (PRIN 2017AY8BP4 and PON “Ricerca e Innovazione” PerMedNet project ARS01_01226) and by Sumitomo Dainippon Pharma Co. Ltd., although none of them had a role in study design, data collection, and analysis as well as in manuscript writing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M.A.R. has received compensation as speaker/consultant from Lundbeck, Otzuka, Sumitomo Dainippon Pharma and Sunovion, and he has received research grants from Lundbeck, Sumitomo Dainippon Pharma and Sunovion. All the other authors declare no financial interests or potential conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Marchisella, F., Paladini, M.S., Guidi, A. et al. Chronic treatment with the antipsychotic drug blonanserin modulates the responsiveness to acute stress with anatomical selectivity. Psychopharmacology 237, 1783–1793 (2020). https://doi.org/10.1007/s00213-020-05498-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-020-05498-9