Abstract
Rationale
Over-the-counter drugs containing Hypericum perforatum (H. perforatum) have been argued to improve memory and sustained attention. So far, these claims have not been supported in human studies. However, previous studies used rather high dosages, and little is known about the acute effect of small dosages.
Objective
We evaluated whether an acute treatment with Remotiv 500 and Remotiv 250 (500 or 250 mg of H. perforatum quantified to either 1 or 0.5 mg of hypericin) improved memory and sustained attention, as well as mood and state anxiety in healthy adults.
Method
A single dosage, randomized, double-blind, placebo-controlled trial was conducted with 82 student participants (33 women). Each participant received placebo in one session and one of two dosages in the other session. Order of the sessions and dosage conditions were randomized between subjects. Participants completed a battery of tasks assessing short-term memory capacity and sustained attention.
Results
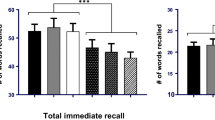
A significant positive effect of Remotiv 250 on digit span (mean Cohen’s d = 0.58; p = .01) was observed. By contrast, Remotiv 500 had a negative effect on digit span (mean d = − 0.48, p = 0.04). A similar effect emerged when factoring across tests of short-term memory. Both dosages improved mood (d = 0.60, p = .03).
Conclusions
The results indicate that acute treatment with small (250 mg) dosages of H. perforatum has a positive effect on the capacity of short-term verbal memory, and stress the importance of maintaining small dosages in nootropic applications.
Trial registration
www.clinicaltrials.gov NCT02862236.



Similar content being viewed by others
Notes
Those with sexual dysfunctions and sleeping disorders were allowed to participate.
One participant scored above cutoff on the BDI. Yet as his score was very close to the cutoff level it was decided to include him. As described below, all analyses were re-rerun controlling for BDI scores.
Additionally, colors were re-randomized after about half the subjects had been run to prevent an effect of color on the experimental results.
In all of these analyses, there was no significant main effect of dosage (p’s > 0.30), confirming the absence of a difference between the two dosage groups independently of the administration of the drug.
References
Agay N, Yechiam E, Carmel Z, Levkovitz Y (2010) Non-specific effects of methylphenidate (Ritalin) on cognitive ability and decision-making of ADHD and healthy adults. Psychopharmacology 210:511–519
Amado-Boccara I, Gougoulis N, Littre MFP, Galinowski A, Loo H (1995) Effects of antidepressants on cognitive functions: a review. Neurosci Biobehav Rev 19:479–493
Austin MP, Tully L, Parker G (2007) Examining the relationship between antenatal anxiety and postnatal depression. J Affect Disord 101:169–174
Bartholomeusz CF, Box G, Rooy CV, Nathan PJ (2003) The modulatory effects of dopamine D1 and D2 receptor function on object working memory in humans. J Psychopharmacol 17:9–15
Beck AT, Ward C, Mendelson M (1961) Beck depression inventory (BDI). Arch Gen Psychiatry 4:561–571
Ben-Eliezer D, Yechiam E (2016) Hypericum perforatum as a cognitive enhancer in rodents: a meta-analysis. Sci Rep UK 6:35700
Brozoski TJ, Brown R, Rosvold HE, Goldman PS (1979) Cognitive deficit caused by regional depletion of dopamine in the prefrontal cortex of rhesus monkeys. Science 205:929–931
Cala S, Crismon ML, Baumgartner J (2003) A survey of herbal use in children with attention-deficit/hyperactivity-disorder or depression. Pharmacotherapy 23:222–230
Carvalho AF, Köhler CA, Cruz EP, Stürmer PL, Reichman BP, Barea BM, Izquierdo I, Chaves MLF (2006) Acute treatment with the antidepressants bupropion and sertraline do not influence memory retrieval in man. Eur Arch Psychiatry Clin Neurosci 256:320–325
Chevassus H, Farret A, Gagnol JP, Ponçon C-A, Costa F, Roux C, Galtier F, Petit P (2013) Psychological and physiological effects of bupropion compared to methylphenidate after prolonged administration in healthy volunteers (NCT00285155). Eur J Clin Pharmacol 69:779–787
Concerto C, Boo H, Hu C, Sandilya P, Krish A, Chusid E, Coira D, Aguglia E, Battaglia F (2018) Hypericum perforatum extract modulates cortical plasticity in humans. Psychopharmacology in press 235:145–153
Cools R, D’Esposito M (2011) Inverted-U-shaped dopamine actions on human working memory and cognitive control. Biol Psychiatry 69:e113–e125
Cousineau D (2005) Confidence intervals in within-subject designs: a simpler solution to Loftus and Masson’s method. Lect Quant Psych 1:4–45
Ellis KA, Stough C, Vitetta L, Heinrich K, Nathan PJ (2001) An investigation into the acute nootropic effects of Hypericum perforatum L. (St. John's Wort) in healthy human volunteers. Behav Pharmacol 12:173–182
First MB, Spitzer RL, Gibbon M, Williams JBW (2002) Structured clinical interview for DSM-IV-TR axis I disorders, research version, non-patient edition (SCID-I/NP). Biometrics Research, New York State Psychiatric Institute, New York
Foster JL, Shipstead Z, Harrison TL, Hicks KL, Redick TS, Engle RW (2015) Shortened complex span tasks can reliably measure working memory capacity. Mem Cogn 43:226–236
Franklin M, Chi J, McGavin C, Hockney R, Reed A, Campling G, Whale RWR, Cowen PJ (1999) Neuroendocrine evidence for dopaminergic actions of hypericum extract (LI 160) in healthy volunteers. Biol Psychiatry 46:581–584
Franklin M, Chi D, Mannel M, Cowen PJ (2000) Acute effects of LI 160 (extract of Hypericum perforatum, St John’s wort) and two of its constituents on neuroendocrine responses in the rat. J Psychpharmacol 14:360–363
Greenberg LM, Kindschi CL (1996) TOVA clinical guide. Universal Attention Disorders, Los Alamitos
Griffith TN, Varela-Nallar L, Dinamarca MC, Inestrosa NC (2010) Neurobiological effects of Hyperforin and its potential in Alzheimer’s disease therapy. Curr Med Chem 17:391–406
Harmer CJ, Bhagwagar Z, Cowen PJ, Goodwin GM (2002) Acute administration of citalopram facilitates memory consolidation in healthy volunteers. Psychopharmacology 163:106–110
Hasanein P, Shahidi S (2011) Effects of Hypericum perforatum extract on diabetes-induced learning and memory impairment in rats. Phytother Res 25:544–549
Horesh N (1998) Theorerical models for understanding the relation between stressful life events, copying style, and affective disorders [dissertation]. Tel Aviv University, Tel Aviv
Ilieva IP, Hook CJ, Farah MJ (2015) Prescription stimulants’ effects on healthy inhibitory control, working memory, and episodic memory: a meta-analysis. J Cogn Neurosci 27:1069–1089
Istikoglou CI, Mavreas V, Geroulanos G (2010) History and therapeutic properties of Hypericum Perforatum from antiquity until today. Psychiatriki 21:332–338
Karim J, Weisz R, Rehman SU (2011) International positive and negative affect schedule short-form (I-PANAS-SF): testing for factorial invariance across cultures. Procd Soc Behav 15:2016–2022
Khalifa AE (2001) Hypericum perforatum as a nootropic drug: enhancement of retrieval memory of a passive avoidance conditioning paradigm in mice. J Ehnopharmacol 76:49–57
Klusa V, Germane S, Nöldner M, Chatterjee SS (2001) Hypericum extract and hyperforin: memory-enhancing properties in rodents. Pharmacopsychiatry 34:61–69
Kumar V, Khanna VK, Seth PK, Singh PN, Bhattacharya SK (2002) Brain neurotransmitter receptor binding and nootropic studies on Indian Hypericum perforatum Linn. Phytother Res 16:210–216
Linde K, Berner M, Egger M, Mulrow C (2005) St. John’s wort for depression: meta-analysis of randomised controlled trials. Br J Psychiat 186:99–107
Lovibond SH, Lovibond PF (1995) Manual for the depression anxiety stress scales, 2nd edn. Psychology Foundation, Sydney
Luciana M, Collins PF (2008) Dopaminergic modulation of working memory for spatial but not object cues in normal humans. J Cog Neurosci 9:330–347
Maher B (2008) Poll results: look who’s doping. Nature 452(7188):674–675
Mehta MA, Swainson R, Ogilvie AD, Sahakian BJ, Robbins TW (2001) Improved short-term spatial memory but impaired reversal learning following the dopamine D2 agonist bromocriptine in human volunteers. Psychopharmacology 159:10–20
Mehta MA, Manes FF, Magnolfi G, Sahakian BJ, Robbins TW (2004) Impaired set-shifting and dissociable effects on tests of spatial working memory following the dopamine D2 receptor antagonist sulpiride in human volunteers. Psychopharmacology 176:331–342
Niculescu A, Cao Y, Nijholt A (2010) Manipulating stress and cognitive load in conversational interactions with a multimodal system for crisis management support. In: Esposito A, Campbell N, Vogel C, Hussain A, Nijholt A (eds) Development of multimodal interfaces: active listening and synchrony. Springer, Berlin Heidelberg, pp 134–147
Orth HCJ, Schmidt PC (2000) Stability and stabilization of hyperforin. Pharm Ind 62:60–63
Park SB, Coull JT, McShane RH, Young AH, Sahakian BJ, Robbins TW et al (1994) Tryptophan depletion in normal volunteers produces selective impairments in learning and memory. Neuropharmacology 33:575–588
Pharmeuropa (2004) St John’s wort dry extract, quantified 16:97–98
Pietrzak RH, Maruff P, Mayes LC, Roman SA, Sosa JA, Snyder PJ (2008) An examination of the construct validity and factor structure of the Groton Maze Learning Test, a new measure of spatial working memory, learning efficiency, and error monitoring. Arch Clin Neuropsych 23:433–445
Preacher KJ, Hayes AF (2004) SPSS and SAS procedures for estimating indirect effects in simple mediation models. Behav Res Methods 36:717–731
Quartermain D, Judge ME, Leo P (1988) Attenuation of forgetting by pharmacological stimulation of aminergic neurotransmitter systems. Pharmacol Biochem Behav 30:77–78
Rahimi R, Nikfar S, Abdollahi M (2009) Efficacy and tolerability of Hypericum perforatum in major depressive disorder in comparison with selective serotonin reuptake inhibitors: a meta-analysis. Prog Neuro-Psychopharmacol Biol Psychiatry 33:118–127
Redick TS, Broadway JM, Meier ME, Kuriakose PS, Unsworth N, Kane MJ, Engle RW (2012) Measuring working memory capacity with automated complex span tasks. Eur J Psychol Assess 28:164–171
Repantis D, Laisney O, Heuser I (2010a) Acetylcholinesterase inhibitors and memantine for neuroenhancement in healthy individuals: a systematic review. Pharmacol Res 61:473–481
Repantis D, Schlattmann P, Laisney O, Heuser I (2010b) Modafinil and methylphenidate for neuroenhancement in healthy individuals: a systematic review. Pharmacol Res 62:187–206
Riedel WJ, Klaassen T, Deutz NE, van Someren A, van Praag HM (1999) Tryptophan depletion in normal volunteers produces selective impairment in memory consolidation. Psychopharmacology 141:362–369
Rodriguez-Landa JF, Contreras CM (2003) A review of clinical and experimental observations about antidepressant actions and side effects produced by Hypericum perforatum extracts. Phytomedicine 10:688–699
Rommelspacher H, Siemanowitz B, Mannel M (2001) Acute and chronic actions of a dry methanolic extract of hypericum perforatum and a hyperforin-rich extract on dopaminergic and serotonergic neurones in rat nucleus accumbens. Pharmacopsychiatry 34:119–126
Rose EJ, Simonotto E, Spencer EP, Ebmeier KP (2006) The effects of escitalopram on working memory and brain activity in healthy adults during performance of the n-back task. Psychopharmacology 185:339–347
Sawaguchi T, Matsumura M, Kubota K (1988) Dopamine enhances the neuronal activity of spatial short-term memory task in the primate prefrontal cortex. Neurosci Res 5:465–473
Schellenberg R, Sauer S, Dimpfel W (1998) Pharmacodynamic effects of two different hypericum extracts in healthy volunteers measured by quantitative EEG. Pharmacopsychiatry 31:44–53
Schmitt JAJ, Jorissen BL, Sobczak S, van Boxtel MPJ, Hogervorst E, Deutz EP et al (2000) Tryptophan depletion impairs memory consolidation but improves focussed attention in healthy young volunteers. J Psychopharmacol 14:21–29
Siepmann M, Krause S, Joraschky P, Mück-Weymann M, Kirch W (2002) The effects of St John’s wort extract on heart rate variability, cognitive function and quantitative EEG: a comparison with amitriptyline and placebo in healthy men. Br J Clin Pharmacol 54:277–282
Spielberger CD, Gorsuch RL, Lushene R, Vagg PR, Jacobs GA (1983) Manual for the state-trait anxiety inventory. Consulting Psychologists Press, Palo Alto
Teichman Y, Melnick H (1984) Manual for the Hebrew Slate-Trait Anxiety Inventory, 2nd edition. Ramot, Tel Aviv
Terfehr K, Wolf OT, Schlosser N, Fernando SC, Otte C, Muhtz C, Beblo T, Driessen M, Spitzer C, Löwe B, Wingenfeld K (2011) Effects of acute hydrocortisone administration on declarative memory in patients with major depressive disorder: a placebo controlled, double-blind crossover study. J Clin Psychiatry 72:1644–1650
Timoshanko A, Stough C, Vitetta L, Nathan PJ (2001) A preliminary investigation on the acute pharmacodynamic effects of hypericum on cognitive and psychomotor performance. Behav Pharmacol 12:635–640
Trofimiuk E, Holownia A, Braszko JJ (2010) Activation of CREB by St. John’s wort may diminish deletorious effects of aging on spatial memory. Arch Pharm Res 33:469–477
Turner ML, Engle RW (1989) Is working memory capacity task dependent? J Mem Lang 28:127–154
Unsworth N, Heitz RP, Schrock JC, Engle RW (2005) An automated version of the operation span task. Behav Res Methods 37:498–505
Vissiennon C, Nieber K, Kelber O, Butterweck V (2012) Route of administration determines the anxiolytic activity of the flavonols kaempferol, quercetin and myricetin—are they prodrugs? J Nutr Biochem 23:733–740
Votruba KL, Langenecker SA (2013) Factor structure, construct validity, and age-and education-based normative data for the parametric go/no-go test. J Clin Exp Neuropsychol 35:132–146
Watson D, Clark LA, Tellegen A (1988) Development and validation of brief measures of positive and negative affect: the PANAS scales. J Pers Soc Psychol 54:1063–1070
Weber W, Stoep AB, McCarty RL, Weiss NS, Biederman J, McClellan J (2008) Hypericum perforatum (St John’s Wort) for attention-deficit/hyperactivity disorder in children and adolescents: a randomized controlled trial. JAMA 299:2633–2641
Wechsler D (1981) Manual for the Wechsler adult intelligence scale (Rev.). Psychological Corporation, New York
Widy-Tyszkiewicz E, Piechal A, Joniec I, Blecharz-Klin K (2002) Long term administration of Hypericum perforatum improves spatial learning and memory in the water maze. Biol Pharm Bull 25:1289–1294
Wood S, Sage JR, Shuman T, Anagnostaras SG (2013) Psychostimulants and cognition: a continuum of behavioral and cognitive activation. Pharmacol Rev 66:193–221
Woods D, Kishiyama MM, Yund EW, Herron TJ, Edwards B, Poliva O et al (2011) Improving digit span assessment of short-term verbal memory. J Clin Exp Neuropsychol 33:101–111
Funding
This work was supported by the I-CORE program of the Planning and Budgeting Committee and the Israel Science Foundation (1821/12).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(DOCX 20 kb)
Rights and permissions
About this article
Cite this article
Yechiam, E., Ben-Eliezer, D., Ashby, N.J.S. et al. The acute effect of Hypericum perforatum on short-term memory in healthy adults. Psychopharmacology 236, 613–623 (2019). https://doi.org/10.1007/s00213-018-5088-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-018-5088-0