Abstract
Background
Current diagnosis of drug addiction like other mental disorders is based on clinical symptoms not on neural pathophysiology and consequently, does not provide useful information on the underlying pathophysiology and may impede the efforts to identify the underlying mechanisms. Identifying the functional deficits that are relevant to addiction and can be traced to the neural systems will greatly facilitate our understanding of the heterogeneity of the condition and improve future diagnosis and treatment. Cocaine addiction is characterized by the continued use despite the dire consequences, and the deficit in inhibitory control may play a key role in this process. This study aimed to develop a paradigm to measure the punishment-induced inhibitory regulation of reward-seeking behavior.
Methods
Rats were first trained to self-administer sucrose pellets under a chained schedule and then the breaking points (BPs) under the progressive-ratio schedule, and the intensity-response effects of footshock punishment on sucrose SA were measured. Subsequently, the rats went on to self-administer intravenous cocaine, and the BPs and the punishment intensity-response effects were similarly determined.
Results
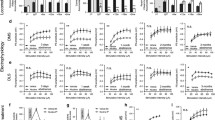
The areas under the punishment intensity-response curves (AUCs) were calculated and used as an indicator of the sensitivity of the inhibitory system. The BPs for cocaine were not correlated with the AUCs. Furthermore, the change in the BPs for cocaine induced by changing cocaine dose did not predict the change in the AUCs.
Conclusion
The intensity-response effects of punishment can be used to measure the function or sensitivity of the inhibitory system independent of the motivational state.








Similar content being viewed by others
References
American Psychiatric Association (2013) Diagnostic and Statistical Manual of Mental Disorders, 5th edn. American Psychiatric Publishing, Incorporated, Washington, DC
Bedford JA, Bailey LP, Wilson MC (1978) Cocaine reinforced progressive ratio performance in the rhesus monkey. Pharmacol Biochem Behav 9(5):631–638. https://doi.org/10.1016/0091-3057(78)90214-9
Belin D, Balado E, Piazza PV, Deroche-Gamonet V (2009) Pattern of intake and drug craving predict the development of cocaine addiction-like behavior in rats. Biol Psychiatry 65(10):863–868. https://doi.org/10.1016/j.biopsych.2008.05.031
Belin D, Belin-Rauscent A, Murray JE, Everitt BJ (2013) Addiction: failure of control over maladaptive incentive habits. Curr Opin Neurobiol 23(4):564–572. https://doi.org/10.1016/j.conb.2013.01.025
Belin D, Mar AC, Dalley JW, Robbins TW, Everitt BJ (2008) High impulsivity predicts the switch to compulsive cocaine-taking. Science 320(5881):1352–1355. https://doi.org/10.1126/science.1158136
Bentzley BS, Jhou TC, Aston-Jones G (2014) Economic demand predicts addiction-like behavior and therapeutic efficacy of oxytocin in the rat. Proc Natl Acad Sci U S A
Bradshaw CM, Killeen PR (2012) A theory of behaviour on progressive ratio schedules, with applications in behavioural pharmacology. Psychopharmacology 222(4):549–564. https://doi.org/10.1007/s00213-012-2771-4
Calu DJ, Stalnaker TA, Franz TM, Singh T, Shaham Y, Schoenbaum G (2007) Withdrawal from cocaine self-administration produces long-lasting deficits in orbitofrontal-dependent reversal learning in rats. Learn Mem 14(5):325–328. https://doi.org/10.1101/lm.534807
Chen BT, Yau HJ, Hatch C, Kusumoto-Yoshida I, Cho SL, Hopf FW, Bonci A (2013) Rescuing cocaine-induced prefrontal cortex hypoactivity prevents compulsive cocaine seeking. Nature 496(7445):359–362. https://doi.org/10.1038/nature12024
Cuthbert BN, Insel TR (2010) Toward new approaches to psychotic disorders: the NIMH research domain criteria project. Schizophr Bull 36(6):1061–1062. https://doi.org/10.1093/schbul/sbq108
Deroche-Gamonet V, Belin D, Piazza PV (2004) Evidence for addiction-like behavior in the rat. Science 305(5686):1014–1017. https://doi.org/10.1126/science.1099020
Dickinson A, Pearce JM (1976) Preference and response suppression under different correlations between shock and a positive reinforcer in rats. Learn Motiv 7(1):66–85. https://doi.org/10.1016/0023-9690(76)90018-7
Ducret E, Puaud M, Lacoste J, Belin-Rauscent A, Fouyssac M, Dugast E, Murray JE, Everitt BJ, Houeto JL, Belin D (2016) N-acetylcysteine facilitates self-imposed abstinence after escalation of cocaine intake. Biol Psychiatry 80(3):226–234. https://doi.org/10.1016/j.biopsych.2015.09.019
Dworkin SI, Bimle C, Miyauchi T (1989) Differential effects of pentobarbital and cocaine on punished and nonpunished responding. J Exp Anal Behav 51(2):173–184. https://doi.org/10.1901/jeab.1989.51-173
Everitt BJ (2014) Neural and psychological mechanisms underlying compulsive drug seeking habits and drug memories—indications for novel treatments of addiction. Eur J Neurosci 40(1):2163–2182. https://doi.org/10.1111/ejn.12644
Everitt BJ, Robbins TW (2016) Drug addiction: updating actions to habits to compulsions ten years on. Annu Rev Psychol 67(1):23–50. https://doi.org/10.1146/annurev-psych-122414-033457
Grove RN, Schuster CR (1974) Suppression of cocaine self-administration by extinction and punishment. Pharmacol Biochem Behav 2(2):199–208. https://doi.org/10.1016/0091-3057(74)90053-7
Hao Y, Martin-Fardon R, Weiss F (2010) Behavioral and functional evidence of metabotropic glutamate receptor 2/3 and metabotropic glutamate receptor 5 dysregulation in cocaine-escalated rats: factor in the transition to dependence. Biol Psychiatry 68(3):240–248. https://doi.org/10.1016/j.biopsych.2010.02.011
Hursh SR, Silberberg A (2008) Economic demand and essential value. Psychol Rev 115(1):186–198. https://doi.org/10.1037/0033-295X.115.1.186
Hyman SE (2005) Addiction: a disease of learning and memory. Am J Psychiatry 162(8):1414–1422. https://doi.org/10.1176/appi.ajp.162.8.1414
Insel T, Cuthbert B, Garvey M, Heinssen R, Pine DS, Quinn K, Sanislow C, Wang P (2010) Research domain criteria (RDoC): toward a new classification framework for research on mental disorders. Am J Psychiatry 167(7):748–751. https://doi.org/10.1176/appi.ajp.2010.09091379
Jentsch JD, Olausson P, De La Garza R 2nd, Taylor JR (2002) Impairments of reversal learning and response perseveration after repeated, intermittent cocaine administrations to monkeys. Neuropsychopharmacology 26(2):183–190. https://doi.org/10.1016/S0893-133X(01)00355-4
Jentsch JD, Taylor JR (1999) Impulsivity resulting from frontostriatal dysfunction in drug abuse: implications for the control of behavior by reward-related stimuli. Psychopharmacology 146(4):373–390. https://doi.org/10.1007/PL00005483
Johanson CE (1977) The effects of electric shock on responding maintained by cocaine injections in a choice procedure in the rhesus monkey. Psychopharmacology 53(3):277–282. https://doi.org/10.1007/BF00492364
Johanson CE, Schuster CR (1975) A choice procedure for drug reinforcers: cocaine and methylphenidate in the rhesus monkey. J Pharmacol Exp Ther 193(2):676–688
Kasanetz F, Lafourcade M, Deroche-Gamonet V, Revest JM, Berson N, Balado E, Fiancette JF, Renault P, Piazza PV, Manzoni OJ (2013) Prefrontal synaptic markers of cocaine addiction-like behavior in rats. Mol Psychiatry 18(6):729–737. https://doi.org/10.1038/mp.2012.59
Koob GF, Mason BJ (2016) Existing and future drugs for the treatment of the dark side of addiction. Annu Rev Pharmacol Toxicol 56(1):299–322. https://doi.org/10.1146/annurev-pharmtox-010715-103143
Krueger DD, Howell JL, Oo H, Olausson P, Taylor JR, Nairn AC (2009) Prior chronic cocaine exposure in mice induces persistent alterations in cognitive function. Behav Pharmacol 20(8):695–704. https://doi.org/10.1097/FBP.0b013e328333a2bb
Kwako LE, Momenan R, Grodin EN, Litten RZ, Koob GF, Goldman D (2017) Addictions neuroclinical assessment: a reverse translational approach. Neuropharmacology
Kwako LE, Momenan R, Litten RZ, Koob GF, Goldman D (2016) Addictions neuroclinical assessment: a neuroscience-based framework for addictive disorders. Biol Psychiatry 80(3):179–189. https://doi.org/10.1016/j.biopsych.2015.10.024
Litten RZ, Ryan ML, Falk DE, Reilly M, Fertig JB, Koob GF (2015) Heterogeneity of alcohol use disorder: understanding mechanisms to advance personalized treatment. Alcoholism-Clinical and Experimental Research 39(4):579–584. https://doi.org/10.1111/acer.12669
McMillan DE (1975) Determinants of drug effects on punished responding. Fed Proc 34(9):1870–1879
Morgan D, Smith MA, Roberts DC (2005) Binge self-administration and deprivation produces sensitization to the reinforcing effects of cocaine in rats. Psychopharmacology 178(2–3):309–316. https://doi.org/10.1007/s00213-004-1992-6
National Research Council (2011) Guide for the care and use of laboratory animals, eighth edn. The National Academies, Washington, DC
Pascoli V, Terrier J, Hiver A, Luscher C (2015) Sufficiency of mesolimbic dopamine neuron stimulation for the progression to addiction. Neuron 88(5):1054–1066. https://doi.org/10.1016/j.neuron.2015.10.017
Pearce JM, Dickinson A (1975) Pavlovian counterconditioning: changing the suppressive properties of shock by association with food. J Exp Psychol Anim Behav Process 1(2):170–177. https://doi.org/10.1037/0097-7403.1.2.170
Pelloux Y, Dilleen R, Economidou D, Theobald D, Everitt BJ (2012) Reduced forebrain serotonin transmission is causally involved in the development of compulsive cocaine seeking in rats. Neuropsychopharmacology 37(11):2505–2514. https://doi.org/10.1038/npp.2012.111
Pelloux Y, Everitt BJ, Dickinson A (2007) Compulsive drug seeking by rats under punishment: effects of drug taking history. Psychopharmacology 194(1):127–137. https://doi.org/10.1007/s00213-007-0805-0
Pelloux Y, Murray JE, Everitt BJ (2015) Differential vulnerability to the punishment of cocaine related behaviours: effects of locus of punishment, cocaine taking history and alternative reinforcer availability. Psychopharmacology 232(1):125–134. https://doi.org/10.1007/s00213-014-3648-5
Roberts DC, Bennett SA, Vickers GJ (1989a) The estrous cycle affects cocaine self-administration on a progressive ratio schedule in rats. Psychopharmacology 98(3):408–411. https://doi.org/10.1007/BF00451696
Roberts DC, Gabriele A, Zimmer BA (2013) Conflation of cocaine seeking and cocaine taking responses in IV self-administration experiments in rats: methodological and interpretational considerations. Neurosci Biobehav Rev 37(9):2026–2036. https://doi.org/10.1016/j.neubiorev.2013.04.017
Roberts DC, Loh EA, Vickers G (1989b) Self-administration of cocaine on a progressive ratio schedule in rats: dose-response relationship and effect of haloperidol pretreatment. Psychopharmacology 97(4):535–538. https://doi.org/10.1007/BF00439560
Robinson TE, Berridge KC (1993) The neural basis of drug craving: an incentive-sensitization theory of addiction. Brain Res Brain Res Rev 18(3):247–291. https://doi.org/10.1016/0165-0173(93)90013-P
Robinson TE, Berridge KC (2008) The incentive sensitization theory of addiction: some current issues. Philos Trans R Soc Lond Ser B Biol Sci 363(1507):3137–3146. https://doi.org/10.1098/rstb.2008.0093
Schoenbaum G, Saddoris MP, Ramus SJ, Shaham Y, Setlow B (2004) Cocaine-experienced rats exhibit learning deficits in a task sensitive to orbitofrontal cortex lesions. Eur J Neurosci 19(7):1997–2002. https://doi.org/10.1111/j.1460-9568.2004.03274.x
Smith MA, Ward SJ, Roberts DC (2008) Lesions of the dorsomedial frontal cortex block sensitization to the positive-reinforcing effects of cocaine. Pharmacol Biochem Behav 88(3):238–246. https://doi.org/10.1016/j.pbb.2007.08.007
Spealman RD (1979) Comparison of drug effects on responding punished by pressurized air or electric shock delivery in squirrel monkeys: pentobarbital, chlordiazepoxide, d-amphetamine and cocaine. J Pharmacol Exp Ther 209(2):309–315
Vanderschuren LJ, Everitt BJ (2004) Drug seeking becomes compulsive after prolonged cocaine self-administration. Science 305(5686):1017–1019. https://doi.org/10.1126/science.1098975
Xue Y, Steketee JD, Sun W (2012) Inactivation of the central nucleus of the amygdala reduces the effect of punishment on cocaine self-administration in rats. Eur J Neurosci 35(5):775–783. https://doi.org/10.1111/j.1460-9568.2012.08000.x
Funding
The project was supported by Grant Number DA034776 (WLS) from the National Institute on Drug Abuse, and its contents are solely the responsibility of the authors and do not necessarily represent the official views of NIDA or NIH.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics
All procedures followed the National Institute of Health Guidelines for the Care and Use of Laboratory Animals.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Datta, U., Martini, M. & Sun, W. Different functional domains measured by cocaine self-administration under the progressive-ratio and punishment schedules in male Wistar rats. Psychopharmacology 235, 897–907 (2018). https://doi.org/10.1007/s00213-017-4808-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-017-4808-1