Abstract
Rationale
GABAB receptors (GABABR) play a critical role in GABAergic neurotransmission in the brain and are thought to be one of the most promising targets for the treatment of drug addiction. GABABR positive allosteric modulators (PAMs) have shown promise as potential anti-addictive therapies, as they lack the sedative and muscle relaxant properties of full GABAB receptor agonists such as baclofen.
Objectives
The present study was aimed at developing novel, selective, and potent GABABR PAMs with efficacy on abuse-related effects of nicotine.
Results
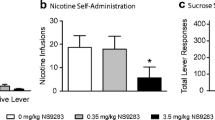
We synthetized ~100 analogs of BHF177, a GABABR PAM that has been shown to inhibit nicotine taking and seeking, and tested their activity in multiple cell-based functional assays. Among these compounds, KK-92A displayed superior PAM properties at the GABABR. Interestingly, our results revealed the existence of pathway-selective differential modulation of GABABR signaling by the structurally related GABABR allosteric modulators BHF177 and KK-92A. In vivo, similarly to BHF177, KK-92A inhibited intravenous nicotine self-administration under both fixed- and progressive-ratio schedules of reinforcement in rats. In contrast to BHF177, KK-92A had no effect on food self-administration. Furthermore, KK-92A decreased cue-induced nicotine-seeking behavior without affecting food seeking.
Conclusions
These results indicate that KK-92A is a selective GABABR PAM with efficacy in inhibition of the primary reinforcing and incentive motivational effects of nicotine, and attenuation of nicotine seeking, further confirming that GABABR PAMs may be useful antismoking medications.





Similar content being viewed by others
References
American Psychiatric Association (1994) Diagnostic and statistical manual of mental disorders, 4th edn. American Psychiatric Press, Washington
Besnard J, Ruda GF, Setola V, Abecassis K, Rodriguiz RM, Huang XP, Norval S, Sassano MF, Shin AI, Webster LA, Simeons FR, Stojanovski L, Prat A, Seidah NG, Constam DB, Bickerton GR, Read KD, Wetsel WC, Gilbert IH, Roth BL, Hopkins AL (2012) Automated design of ligands to polypharmacological profiles. Nature 492:215–220
Bettler B, Tiao JY (2006) Molecular diversity, trafficking and subcellular localization of GABAB receptors. Pharmacol Ther 110:533–543
Beveridge TJ, Smith HR, Porrino LJ (2013) Differential development of tolerance to the functional and behavioral effects of repeated baclofen treatment in rats. Pharmacol Biochem Behav 106:27–32
Chiamulera C (2005) Cue reactivity in nicotine and tobacco dependence: a "multiple-action" model of nicotine as a primary reinforcement and as an enhancer of the effects of smoking-associated stimuli. Brain Res Brain Res Rev 48:74–97
Christopoulos A (2002) Allosteric binding sites on cell-surface receptors: novel targets for drug discovery. Nat Rev Drug Discov 1:198–210
Ciambrone GJ, Liu VF, Lin DC, McGuinness RP, Leung GK, Pitchford S (2004) Cellular dielectric spectroscopy: a powerful new approach to label-free cellular analysis. J Biomol Screen 9:467–480
Corrigall WA, Coen KM, Adamson KL, Chow BL, Zhang J (2000) Response of nicotine self-administration in the rat to manipulations of mu-opioid and gamma-aminobutyric acid receptors in the ventral tegmental area. Psychopharmacology 149:107–114
Corrigall WA, Coen KM, Zhang J, Adamson KL (2001) GABA mechanisms in the pedunculopontine tegmental nucleus influence particular aspects of nicotine self-administration selectively in the rat. Psychopharmacology 158:190–197
Cousins MS, Stamat HM, de Wit H (2001) Effects of a single dose of baclofen on self-reported subjective effects and tobacco smoking. Nicotine Tob Res 3:123–129
Cryan JF, Kaupmann K (2005) Don't worry 'B' happy!: a role for GABA(B) receptors in anxiety and depression. Trends Pharmacol Sci 26:36–43
Dewey SL, Brodie JD, Gerasimov M, Horan B, Gardner EL, Ashby CR, Jr. (1999) A pharmacologic strategy for the treatment of nicotine addiction. Synapse (New York, NY) 31: 76–86
Ehrman RN, Robbins SJ, Childress AR, Goehl L, Hole AV, O'Brien CP (1998) Laboratory exposure to cocaine cues does not increase cocaine use by outpatient subjects. J Subst Abus Treat 15:431–435
Epstein DH, Preston KL, Stewart J, Shaham Y (2006) Toward a model of drug relapse: an assessment of the validity of the reinstatement procedure. Psychopharmacology 189:1–16
Fadda P, Scherma M, Fresu A, Collu M, Fratta W (2003) Baclofen antagonizes nicotine-, cocaine-, and morphine-induced dopamine release in the nucleus accumbens of rat. Synapse (New York, NY) 50: 1–6
Ferguson SG, Shiffman S (2009) The relevance and treatment of cue-induced cravings in tobacco dependence. J Subst Abus Treat 36:235–243
Filip M, Frankowska M (2007) Effects of GABA(B) receptor agents on cocaine priming, discrete contextual cue and food induced relapses. Eur J Pharmacol 571:166–173
Filip M, Frankowska M, Przegalinski E (2007) Effects of GABA(B) receptor antagonist, agonists and allosteric positive modulator on the cocaine-induced self-administration and drug discrimination. Eur J Pharmacol 574:148–157
Filip M, Frankowska M, Sadakierska-Chudy A, Suder A, Szumiec L, Mierzejewski P, Bienkowski P, Przegalinski E, Cryan JF (2014) GABA receptors as a therapeutic strategy in substance use disorders: focus on positive allosteric modulators. Neuropharmacology
Franklin TR, Harper D, Kampman K, Kildea-McCrea S, Jens W, Lynch KG, O'Brien CP, Childress AR (2009) The GABA B agonist baclofen reduces cigarette consumption in a preliminary double-blind placebocontrolled smoking reduction study, Drug Alcohol Depend. 103(1-2): 30-6
Guery S, Floersheim P, Kaupmann K, Froestl W (2007) Syntheses and optimization of new GS39783 analogues as positive allosteric modulators of GABA B receptors. Bioorg Med Chem Lett 17:6206–6211
Im BH, Rhim H (2012) GABA(B) receptor-mediated ERK1/2 phosphorylation via a direct interaction with Ca(V)1.3 channels. Neurosci Lett 513:89–94
Jensen AA, Spalding TA (2004) Allosteric modulation of G-protein coupled receptors. European journal of pharmaceutical sciences : official journal of the European Federation for Pharmaceutical Sciences 21:407–420
Kalivas PW (1993) Neurotransmitter regulation of dopamine neurons in the ventral tegmental area. Brain Res Brain Res Rev 18:75–113
Keiser MJ, Setola V, Irwin JJ, Laggner C, Abbas AI, Hufeisen SJ, Jensen NH, Kuijer MB, Matos RC, Tran TB, Whaley R, Glennon RA, Hert J, Thomas KL, Edwards DD, Shoichet BK, Roth BL (2009) Predicting new molecular targets for known drugs. Nature 462:175–181
Kenakin T (2007) Allosteric theory: taking therapeutic advantage of the malleable nature of GPCRs. Curr Neuropharmacol 5:149–156
Laviolette SR, van der Kooy D (2004) The neurobiology of nicotine addiction: bridging the gap from molecules to behaviour. Nat Rev Neurosci 5:55–65
Li X, Semenova S, D'Souza MS, Stoker AK, Markou A (2014) Involvement of glutamatergic and GABAergic systems in nicotine dependence: implications for novel pharmacotherapies for smoking cessation. Neuropharmacology 76(Pt B):554–565
Li X, Kaczanowska K, Finn MG, Markou A, Risbrough VB (2015) The GABA(B) receptor positive modulator BHF177 attenuated anxiety, but not conditioned fear, in rats. Neuropharmacology 97:357–364
Liang JH, Chen F, Krstew E, Cowen MS, Carroll FY, Crawford D, Beart PM, Lawrence AJ (2006) The GABA(B) receptor allosteric modulator CGP7930, like baclofen, reduces operant self-administration of ethanol in alcohol-preferring rats. Neuropharmacology 50:632–639
Liechti ME, Lhuillier L, Kaupmann K, Markou A (2007) Metabotropic glutamate 2/3 receptors in the ventral tegmental area and the nucleus accumbens shell are involved in behaviors relating to nicotine dependence. The Journal of neuroscience : the official journal of the Society for Neuroscience 27:9077–9085
Loi B, Maccioni P, Lobina C, Carai MA, Gessa GL, Thomas AW, Malherbe P, Colombo G (2013) Reduction of alcohol intake by the positive allosteric modulator of the GABA(B) receptor, rac-BHFF, in alcohol-preferring rats. Alcohol (Fayetteville, NY) 47: 69–73
Maccioni P, Pes D, Orru A, Froestl W, Gessa GL, Carai MA, Colombo G (2007) Reducing effect of the positive allosteric modulator of the GABA(B) receptor, GS39,783, on alcohol self-administration in alcohol-preferring rats. Psychopharmacology 193:171–178
Maccioni P, Fantini N, Froestl W, Carai MA, Gessa GL, Colombo G (2008) Specific reduction of alcohol's motivational properties by the positive allosteric modulator of the GABAB receptor, GS39783—comparison with the effect of the GABAB receptor direct agonist, baclofen. Alcohol Clin Exp Res 32:1558–1564
Maccioni P, Carai MA, Kaupmann K, Guery S, Froestl W, Leite-Morris KA, Gessa GL, Colombo G (2009) Reduction of alcohol's reinforcing and motivational properties by the positive allosteric modulator of the GABA(B) receptor, BHF177, in alcohol-preferring rats. Alcohol Clin Exp Res 33:1749–1756
Markou A, Paterson NE (2009) Multiple motivational forces contribute to nicotine dependence. Neb Symp Motiv 55:65–89
Markou A, Paterson NE, Semenova S (2004) Role of gamma-aminobutyric acid (GABA) and metabotropic glutamate receptors in nicotine reinforcement: potential pharmacotherapies for smoking cessation. Ann N Y Acad Sci 1025:491–503
Meier SD, Kafitz KW, Rose CR (2008) Developmental profile and mechanisms of GABA-induced calcium signaling in hippocampal astrocytes. Glia 56:1127–1137
New DC, An H, Ip NY, Wong YH (2006) GABAB heterodimeric receptors promote Ca2+ influx via store-operated channels in rat cortical neurons and transfected Chinese hamster ovary cells. Neuroscience 137:1347–1358
O'Brien CP, Childress AR, Ehrman R, Robbins SJ (1998) Conditioning factors in drug abuse: can they explain compulsion? J Psychopharmacol 12:15–22
Ong J, Kerr DI (2005) Clinical potential of GABAB receptor modulators. CNS Drug Rev 11:317–334
Park HW, Jung H, Choi KH, Baik JH, Rhim H (2010) Direct interaction and functional coupling between voltage-gated CaV1.3 Ca2+ channel and GABAB receptor subunit 2. FEBS Lett 584:3317–3322
Paterson NE, Markou A (2002) Increased GABA neurotransmission via administration of gamma-vinyl GABA decreased nicotine self-administration in the rat. Synapse (New York, NY) 44: 252–3
Paterson NE, Froestl W, Markou A (2004) The GABAB receptor agonists baclofen and CGP44532 decreased nicotine self-administration in the rat. Psychopharmacology 172:179–186
Paterson NE, Froestl W, Markou A (2005) Repeated administration of the GABAB receptor agonist CGP44532 decreased nicotine self-administration, and acute administration decreased cue-induced reinstatement of nicotine-seeking in rats. Neuropsychopharmacology : official publication of the American College of Neuropsychopharmacology 30:119–128
Paterson NE, Vlachou S, Guery S, Kaupmann K, Froestl W, Markou A (2008) Positive modulation of GABA(B) receptors decreased nicotine self-administration and counteracted nicotine-induced enhancement of brain reward function in rats. J Pharmacol Exp Ther 326:306–314
Peters MF, Knappenberger KS, Wilkins D, Sygowski LA, Lazor La, Liu J, Scott CW (2007) Evaluation of Cellular Dielectric Spectroscopy, a Whole-Cell, Label-Free Technology for Drug Discovery on Gi-Coupled GPCRs, Journal of Biomolecular Screening: 12(3):312-9
Picciotto MR, Corrigall WA (2002) Neuronal systems underlying behaviors related to nicotine addiction: neural circuits and molecular genetics. The Journal of neuroscience : the official journal of the Society for Neuroscience 22:3338–3341
Richardson NR, Roberts DC (1996) Progressive ratio schedules in drug self-administration studies in rats: a method to evaluate reinforcing efficacy. J Neurosci Methods 66:1–11
Shiffman S, Brockwell SE, Pillitteri JL, Gitchell JG (2008) Use of smoking-cessation treatments in the United States. Am J Prev Med 34:102–111
Smith MA, Yancey DL, Morgan D, Liu Y, Froestl W, Roberts DC (2004) Effects of positive allosteric modulators of the GABAB receptor on cocaine self-administration in rats. Psychopharmacology 173:105–111
Sturchler E, Li X, de Lourdes LM, Kaczanowska K, Cameron M, Griffin FPG, Markou A, Patricia M (2017) GABAB receptor allosteric modulators exhibit pathway-dependent and species-selective activity. Pharmacol Res Perspect. doi:10.1002/prp2.288
Tu H, Rondard P, Xu C, Bertaso F, Cao F, Zhang X, Pin JP, Liu J (2007) Dominant role of GABAB2 and Gbetagamma for GABAB receptor-mediated-ERK1/2/CREB pathway in cerebellar neurons. Cell Signal 19:1996–2002
Tyacke RJ, Lingford-Hughes A, Reed LJ, Nutt DJ (2010) GABAB receptors in addiction and its treatment. Advances in pharmacology (San Diego, Calif) 58: 373–96
Urwyler S, Mosbacher J, Lingenhoehl K, Heid J, Hofstetter K, Froestl W, Bettler B, Kaupmann K (2001) Positive allosteric modulation of native and recombinant gamma-aminobutyric acid(B) receptors by 2,6-di-tert-butyl-4-(3-hydroxy-2,2-dimethyl-propyl)-phenol (CGP7930) and its aldehyde analog CGP13501. Mol Pharmacol 60:963–971
Urwyler S, Pozza MF, Lingenhoehl K, Mosbacher J, Lampert C, Froestl W, Koller M, Kaupmann K (2003) N,N'-Dicyclopentyl-2-methylsulfanyl-5-nitro-pyrimidine-4,6-diamine (GS39783) and structurally related compounds: novel allosteric enhancers of gamma-aminobutyric acidB receptor function. J Pharmacol Exp Ther 307:322–330
Vlachou S, Markou A (2010) GABAB receptors in reward processes. Advances in pharmacology (San Diego, Calif) 58: 315–71
Vlachou S, Guery S, Froestl W, Banerjee D, Benedict J, Finn MG, Markou A (2011) Repeated administration of the GABAB receptor positive modulator BHF177 decreased nicotine self-administration, and acute administration decreased cue-induced reinstatement of nicotine seeking in rats. Psychopharmacology 215:117–128
Acknowledgements
This work was funded by NIH grant U19 DA026838 to AM, PG, and MGF.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Athina Markou had received contract research support from Astra-Zeneca, Forest Laboratories, and Bristol-Myers Squibb, and honorarium from AbbVie, Germany, during the last 3 years. The remaining authors report no financial conflicts of interests.
Additional information
Xia Li and Emmanuel Sturchler are equal contributors.
Electronic supplementary material
.
ESM 1
(DOCX 527 kb)
Rights and permissions
About this article
Cite this article
Li, X., Sturchler, E., Kaczanowska, K. et al. KK-92A, a novel GABAB receptor positive allosteric modulator, attenuates nicotine self-administration and cue-induced nicotine seeking in rats. Psychopharmacology 234, 1633–1644 (2017). https://doi.org/10.1007/s00213-017-4594-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-017-4594-9