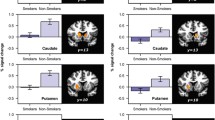
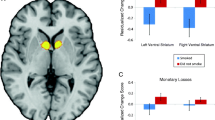
Abstract
Rationale
We have shown that differences in the level of neural activation to stimuli associated with smoking vs. natural rewards, a biomarker related to reward sensitivity, predict treatment outcome.
Objectives
This paper examined whether this biomarker moderates the impact of bupropion or varenicline on smoking cessation.
Methods
Prior to treatment randomization, smokers (N = 180) in a placebo-controlled trial using bupropion and varenicline completed event-related potential recording (late positive potential, LPP) while viewing pleasant (P), cigarette (C)-related, and other pictures. We used Bayesian models to estimate the probability of interaction between treatment and the LPP for both efficacy and comparative effectiveness analyses.
Results
Efficacy analysis showed that smokers with more neural activation to pleasant vs. cigarette-related stimuli (P > C) had a 98–99% chance of achieving greater abstinence than placebo (OR >1.00), using either medication from the end of treatment (EOT, primary outcome) through the 3-month follow-up. Relative to placebo, smokers with higher activation to cigarette-related vs. pleasant stimuli (C > P) had a 99% chance of increased benefit from varenicline at both time points (OR >1), but only 67 and 43% with bupropion at the EOT and 3-month follow-up, respectively. Comparative effectiveness analysis found that smokers with the C > P activation pattern had a 95–98% chance of benefit from varenicline vs. bupropion, while P > C smokers had a 50–58% chance of similar improvement with varenicline at the EOT and 3 months.
Conclusions
Varenicline appears to be the treatment of choice for smokers with the C > P pattern of neural activation, while for those showing P > C, varenicline and bupropion have similar efficacy.

Similar content being viewed by others
References
Anthenelli RM, Benowitz NL, West R, St AL, McRae T, Lawrence D, Ascher J, Russ C, Krishen A, Evins AE (2016) Neuropsychiatric safety and efficacy of varenicline, bupropion, and nicotine patch in smokers with and without psychiatric disorders (EAGLES): a double-blind, randomised, placebo-controlled clinical trial. Lancet 387(10037):2507–2520. doi:10.1016/S0140-6736(16)30272-0
Berry DA (2005) Introduction to Bayesian methods III: use and interpretation of Bayesian tools in design and analysis. Clinical Trials 2(4):295–300. doi:10.1191/1740774505cn100oa
Brandon TH, Drobes DJ, Unrod M, Heckman BW, Oliver JA, Roetzheim RC, Karver SB, Small BJ (2011) Varenicline effects on craving, cue reactivity, and smoking reward. Psychopharmacology 218(2):391–403. doi:10.1007/s00213-011-2327-z
Brookes ST, Whitely E, Egger M, Smith GD, Mulheran PA, Peters TJ (2004) Subgroup analyses in randomized trials: risks of subgroup-specific analyses; power and sample size for the interaction test. J Clin Epidemiol 57(3):229–236. doi:10.1016/j.jclinepi.2003.08.009
Brown RA, Burgess ES, Sales SD, Whiteley JA, Evans DM, Miller IW (1998) Reliability and validity of a smoking timeline follow-back interview. Psychol Addict Behav 12(2):101–112. doi:10.1037/0893-164X.12.2.101
Caggiula AR, Donny EC, Palmatier MI, Liu X, Chaudhri N, Sved AF (2009) The role of nicotine in smoking: a dual-reinforcement model. In: Bevins R, Caggiula A (eds) The motivational impact of nicotine and its role in tobacco use, Nebraska symposium on motivation, vol 55. Springer-Verlag, New York, pp 91–109
Cahill K, Stevens S, Perera R, Lancaster T (2013) Pharmacological interventions for smoking cessation: an overview and network meta-analysis. Cochrane Database Syst Rev 5:CD009329–CD009329. doi:10.1002/14651858.CD009329.pub2
Carter BL, Robinson JD, Lam CY, Wetter DW, Day SX, Tsan JY, Cinciripini PM (2006) A psychometric evaluation of cigarette stimuli used in a cue reactivity study. Nicotine Tob Res 8(3):361–369. doi:10.1080/14622200600670215
Cinciripini PM, Robinson JD, Karam-Hage M, Minnix JA, Lam CY, Versace F, Brown VL, Engelmann JM, Wetter DW (2013) Effects of varenicline and bupropion sustained-release use plus intensive smoking cessation counseling on prolonged abstinence from smoking and on depression, negative affect, and other symptoms of nicotine withdrawal. JAMA Psychiatry 70(5):522–533. doi:10.1001/jamapsychiatry.2013.678
Coe JW, Brooks PR, Vetelino MG, Wirtz MC, Arnold EP, Huang J, Sands SB, Davis TI, Lebel LA, Fox CB, Shrikhande A, Heym JH, Schaeffer E, Rollema H, Lu Y, Mansbach RS, Chambers LK, Rovetti CC, Schulz DW, Tingley FDI, O’Neill BT (2005) Varenicline: an alpha4beta2 nicotinic receptor partial agonist for smoking cessation. J Med Chem 48(10):3474–3477. doi:10.1021/jm050069n
Dixon DO, Simon R (1991) Bayesian subset analysis. Biometrics 47(3):871–881
Gelman A (2011) Induction and deduction in Bayesian data analysis. Rationality, Markets and Morals 2:67–78
Gilbert DG, Rabinovich NE (1999) The international smoking image series (with neutral counterparts), v. 1.2. Department of Psychology, Southern Illinois University, Carbondale
Goodman SN (2005) Introduction to Bayesian methods I: measuring the strength of evidence. Clinical Trials 2(4):282–290. doi:10.1191/1740774505cn098oa
Green CE, Moeller FG, Schmitz JM, Lucke JF, Lane SD, Swann AC, Lasky RE, Carbonari JP (2009) Evaluation of heterogeneity in pharmacotherapy trials for drug dependence: a Bayesian approach. Am J Drug Alcohol Abuse 35(2):95–102. doi:10.1080/00952990802647503
Hajcak G, MacNamara A, Olvet DM (2010) Event-related potentials, emotion, and emotion regulation: an integrative review. Dev Neuropsychol 35(2):129–155. doi:10.1080/87565640903526504
Heatherton TF, Kozlowski LT, Frecker RC, Fagerström KO (1991) The Fagerström test for nicotine dependence: a revision of the Fagerström Tolerance Questionnaire. Br J Addiction 86(9):1119–1127. doi:10.1111/j.1360-0443.1991.tb01879.x
Hughes JR, Keely JP, Niaura RS, Ossip-Klein DJ, Richmond RL, Swan GE (2003) Measures of abstinence in clinical trials: issues and recommendations. Nicotine Tob Res 5(1):13–25. doi:10.1080/1462220031000070552
Hughes JR, Stead LF, Hartmann-Boyce J, Cahill K, Lancaster T (2014) Antidepressants for smoking cessation. Cochrane Database Syst Rev 1:CD000031
Koob GF, Volkow ND (2010) Neurocircuitry of addiction. Neuropsychopharmacol 35(1):217–238. doi:10.1038/npp.2009.110
Lang PJ, Bradley MM (2009) Emotion and the motivational brain. Biol Psychol 84(3):437–450. doi:10.1016/j.biopsycho.2009.10.007
Lang PJ, Bradley MM, Cuthbert BN (2005) International Affective Picture System (IAPS): affective ratings of pictures and instruction manual. Technical report no. A-6. University of Florida, Gainesville
Law KL, Stroud LR, LaGasse LL, Niaura R, Liu J, Lester BM (2003) Smoking during pregnancy and newborn neurobehavior. Pediatrics 111(6):1318–1323
Lipscomb B, Ma G, Berry DA (2005) Bayesian predictions of final outcomes: regulatory approval of a spinal implant. Clinical Trials 2(4):325–333. doi:10.1191/1740774505cn104oa
Mills EJ, Wu P, Lockhart I, Thorlund K, Puhan M, Ebbert JO (2012) Comparisons of high-dose and combination nicotine replacement therapy, varenicline, and bupropion for smoking cessation: a systematic review and multiple treatment meta-analysis. Ann Med 44(6):588–597. doi:10.3109/07853890.2012.705016
O’Neill RT (2006) FDA’s critical path initiative: a perspective on contributions of biostatistics. Biom J 48(4):559–564. doi:10.1002/bimj.200510237
Parmar MKB, Ungerleider RS, Simon R (1996) Assessing whether to perform a confirmatory randomized clinical trial. J Natl Cancer Inst 88(22):1645–1651. doi:10.1093/jnci/88.22.1645
Piper ME, Smith SS, Schlam TR, Fiore MC, Jorenby DE, Fraser D, Baker TB (2009) A randomized placebo-controlled clinical trial of 5 smoking cessation pharmacotherapies. Arch Gen Psychiatry 66(11):1253–1262. doi:10.1001/archgenpsychiatry.2009.142
Robinson JD, Versace F, Lam CY, Minnix JA, Engelmann JM, Cui Y, Karam-Hage M, Shete S, Tomlinson GE, Chen TT-L, Wetter DW, Green CE, Cinciripini PM (2013) The CHRNA3 rs578776 variant is associated with an intrinsic reward sensitivity deficit in smokers. Front Psychiatry 4(114):1–11. doi:10.3389/fpsyt.2013.00114
Rukstalis M, Jepson C, Strasser A, Lynch KG, Perkins K, Patterson F, Lerman C (2005) Naltrexone reduces the relative reinforcing value of nicotine in a cigarette smoking choice paradigm. Psychopharmacology 180(1):41–48. doi:10.1007/s00213-004-2136-8
Sescousse G, Caldu X, Segura B, Dreher JC (2013) Processing of primary and secondary rewards: a quantitative meta-analysis and review of human functional neuroimaging studies. Neurosci Biobehav Rev 37(4):681–696. doi:10.1016/j.neubiorev.2013.02.002
Simon R (2002) Bayesian subset analysis: application to studying treatment-by-gender interactions. Stat Med 21(19):2909–2916. doi:10.1002/sim.1295
Simon R, Freedman LS (1997) Bayesian design and analysis of two x two factorial clinical trials. Biometrics 53:456–464. doi:10.2307/2533949
Simon R, Dixon DO, Freidlin B (1996) Bayesian subset analysis of a clinical trial for the treatment of HIV infections. Statist Textbooks Monogr 151:555–576
Spiegelhalter DJ, Abrams KR, Myles JP (2004) Bayesian approaches to clinical trials and health-care evaluation, vol 13. Wiley, New York
Stead LF, Perera R, Bullen C, Mant D, Hartmann-Boyce J, Cahill K, Lancaster T (2012) Nicotine replacement therapy for smoking cessation. Cochrane Database Syst Rev 11:CD000146–CD000146. doi:10.1002/14651858.CD000146.pub4
Stritzke WG, Breiner MJ, Curtin JJ, Lang AR (2004) Assessment of substance cue reactivity: advances in reliability, specificity, and validity. Psychol Addict Behav 18(2):148–159. doi:10.1037/0893-164X.18.2.148
Temple R (2005) How FDA currently makes decisions on clinical studies. Clinical Trials 2(4):276–281. doi:10.1191/1740774505cn097oa
Thanos PK, Taintor NB, Rivera SN, Umegaki H, Ikari H, Roth G, Ingram DK, Hitzemann R, Fowler JS, Gatley SJ, Wang GJ, Volkow ND (2004) DRD2 gene transfer into the nucleus accumbens core of the alcohol preferring and nonpreferring rats attenuates alcohol drinking. Alcohol Clin ExpRes 28(5):720–728. doi:10.1097/01.ALC.0000125270.30501.08
Thanos PK, Michaelides M, Umegaki H, Volkow ND (2008) D2R DNA transfer into the nucleus accumbens attenuates cocaine self-administration in rats. Synapse 62(7):481–486. doi:10.1002/syn.20523
US Food and Drug Administration (2004) Inovation/stagnation: challenge and opportunity on the critical path to new medical products. http://www.fda.gov/downloads/ScienceResearch/SpecialTopics/CriticalPathInitiative/CriticalPathOpportunitiesReports/UCM113411.pdf. Accessed 27 Jan 2017
Versace F, Robinson JD, Lam CY, Minnix JA, Brown VL, Carter BL, Wetter DW, Cinciripini PM (2010) Cigarette cues capture smokers’ attention: evidence from event-related potentials. Psychophysiology 47(3):435–441. doi:10.1111/j.1469-8986.2009.00946.x
Versace F, Engelmann JM, Jackson EF, Costa VD, Robinson JD, Lam CY, Minnix JA, Brown VL, Wetter DW, Cinciripini PM (2011a) Do brain responses to emotional images and cigarette cues differ? An fMRI study in smokers. Eur J Neurosci 34(12):2054–2063. doi:10.1111/j.1460-9568.2011.07915.x
Versace F, Minnix JA, Robinson JD, Lam CY, Brown VL, Cinciripini PM (2011b) Brain reactivity to emotional, neutral and cigarette-related stimuli in smokers. Addict Biol 16(2):296–307. doi:10.1111/j.1369-1600.2010.00273.x
Versace F, Lam CY, Engelmann JM, Robinson JD, Minnix JA, Brown VL, Cinciripini PM (2012) Beyond cue reactivity: blunted brain responses to pleasant stimuli predict long term smoking abstinence. Addict Biol 17(6):991–1000. doi:10.1111/j.1369-1600.2011.00372.x
Versace F, Engelmann JM, Robinson JD, Jackson EF, Green CE, Lam CY, Minnix JA, Karam-Hage M, Brown VL, Wetter DW, Cinciripini PM (2014) Pre-quit fMRI responses to pleasant and cigarette cues predict cessation outcome. Nicotine Tob Res 16(6):697–708. doi:10.1093/ntr/ntt214
Volkow ND, Wang GJ, Fowler JS, Tomasi D, Telang F, Baler R (2010) Addiction: decreased reward sensitivity and increased expectation sensitivity conspire to overwhelm the brain’s control circuit. BioEssays 32(9):748–755. doi:10.1002/bies.201000042
Wijeysundera DN, Austin PC, Hux JE, Beattie WS, Laupacis A (2009) Bayesian statistical inference enhances the interpretation of contemporary randomized controlled trials. J Clin Epidemiol 62(1):13–21. doi:10.1016/j.jclinepi.2008.07.006
Acknowledgements
The authors wish to thank Krystle Bartley, Dr. Victoria L. Brown, Jennifer Canul, Janeene Frerking, Christine Jeria, Paul Longoria, Samuel W. Miller, Kevin Mulpur, Cissette Muster, Tiffany Rattler, and Susana Torres for their help in the data collection. This study was conducted in accordance with the ethical standards of the University of Texas MD Anderson Cancer Center and applicable US Department of Health and Human Services regulations.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The University of Texas MD Anderson Cancer Center Institutional Review Board approved the protocol and informed consent.
Funding
Support for this research was provided by grants from the National Institute on Drug Abuse (grant number R01DA017073) to Paul M. Cinciripini and the Cancer Center Support Grant from the National Cancer Institute (grant number P50CA70907) to MD Anderson Cancer Center. Pfizer provided the varenicline and matching placebo.
Conflict of interest
Dr. Cinciripini served on the scientific advisory board of Pfizer Pharmaceuticals and conducted educational talks sponsored by Pfizer on smoking cessation (2006–2008) and has received grant support from Pfizer. The other authors declare that they have no conflict of interest.
Electronic supplementary material
ESM 1
(PDF 316 kb)
Rights and permissions
About this article
Cite this article
Cinciripini, P.M., Green, C.E., Robinson, J.D. et al. Benefits of varenicline vs. bupropion for smoking cessation: a Bayesian analysis of the interaction of reward sensitivity and treatment. Psychopharmacology 234, 1769–1779 (2017). https://doi.org/10.1007/s00213-017-4580-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-017-4580-2