Abstract
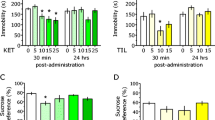
Ketamine is emerging as a new hope against depression, but ketamine-associated psychotomimetic effects limit its clinical use. An adjunct therapy along with ketamine to alleviate its adverse effects and even potentiate the antidepressant effects might be an alternative strategy. Betaine, a methyl derivative of glycine and a dietary supplement, has been shown to have antidepressant-like effects and to act like a partial agonist at the glycine site of N-methyl-D-aspartate receptors (NMDARs). Accordingly, betaine might have potential to be an adjunct to ketamine treatment for depression. The antidepressant-like effects of ketamine and betaine were evaluated by forced swimming test and novelty suppressed feeding test in mice. Both betaine and ketamine produced antidepressant-like effects. Furthermore, we determined the effects of betaine on ketamine-induced antidepressant-like and psychotomimetic behaviors, motor incoordination, hyperlocomotor activity, and anesthesia. The antidepressant-like responses to betaine combined with ketamine were stronger than their individual effects. In contrast, ketamine-induced impairments in prepulse inhibition, novel object recognition test, social interaction, and rotarod test were remarkably attenuated, whereas ketamine-induced hyperlocomotion and loss of righting reflex were not affected by betaine. These findings revealed that betaine could enhance the antidepressant-like effects, yet block the psychotomimetic effects of ketamine, suggesting that betaine can be considered as an add-on therapy to ketamine for treatment-resistant depression and suitable for the treatment of depressive symptoms in patients with schizophrenia.







Similar content being viewed by others
References
Aligeti S, Quinones M, Salazar R (2014) Rapid resolution of suicidal behavior and depression with single low-dose ketamine intravenous push even after 6 months of follow-up. J Clin Psychopharmacol 34:533–535
Autry AE, Adachi M, Nosyreva E, Na ES, Los MF, Cheng PF, Kavalali ET, Monteggia LM (2011) NMDA receptor blockade at rest triggers rapid behavioural antidepressant responses. Nature 475:91–95
Browne CA, Lucki I (2013) Antidepressant effects of ketamine: mechanisms underlying fast-acting novel antidepressants. Front Pharmacol 4:161
Burgdorf J, Zhang XL, Nicholson KL, Balster RL, Leander JD, Stanton PK, Gross AL, Kroes RA, Moskal JR (2013) GLYX-13, a NMDA receptor glycine-site functional partial agonist, induces antidepressant-like effects without ketamine-like side effects. Neuropsychopharmacology 38:729–742
Chai GS, Jiang X, Ni ZF, Ma ZW, Xie AJ, Cheng XS, Wang Q, Wang JZ, Liu GP (2013) Betaine attenuates Alzheimer-like pathological changes and memory deficits induced by homocysteine. J Neurochem 124:388–396
Chan MH, Chiu PH, Sou JH, Chen HH (2008) Attenuation of ketamine-evoked behavioral responses by mGluR5 positive modulators in mice. Psychopharmacology (Berl) 198:141–148
Chan MH, Chiu PH, Lin CY, Chen HH (2012) Inhibition of glycogen synthase kinase-3 attenuates psychotomimetic effects of ketamine. Schizophr Res 136:96–103
Chen X, Shu S, Bayliss DA (2009) HCN1 channel subunits are a molecular substrate for hypnotic actions of ketamine. J Neurosci 29:600–609
Chen HH, Stoker A, Markou A (2010) The glutamatergic compounds sarcosine and N-acetylcysteine ameliorate prepulse inhibition deficits in metabotropic glutamate 5 receptor knockout mice. Psychopharmacology (Berl) 209:343–350
Chen KT, Tsai MH, Wu CH, Jou MJ, Wei IH, Huang CC (2015) AMPA receptor-mTOR activation is required for the antidepressant-like effects of sarcosine during the forced swim test in rats: insertion of AMPA receptor may play a role. Front Behav Neurosci 9:162
Chiu CT, Scheuing L, Liu G, Liao HM, Linares GR, Lin D, Chuang DM (2015) The mood stabilizer lithium potentiates the antidepressant-like effects and ameliorates oxidative stress induced by acute ketamine in a mouse model of stress. Int J Neuropsychopharmacol 18. doi: 10.1093/ijnp/pyu102
Di Pierro F, Orsi R, Settembre R (2015) Role of betaine in improving the antidepressant effect of S-adenosyl-methionine in patients with mild-to-moderate depression. J Multidiscip Healthc 8:39–45
Dwyer JM, Duman RS (2013) Activation of mammalian target of rapamycin and synaptogenesis: role in the actions of rapid-acting antidepressants. Biol Psychiatry 73:1189–1198
Freed WJ (1984) N, N-dimethylglycine, betaine, and seizures. Arch Neurol 41:1129–1130
Freed WJ (1985) Prevention of strychnine-induced seizures and death by the N-methylated glycine derivatives betaine, dimethylglycine and sarcosine. Pharmacol Biochem Behav 22:641–643
Garcia LS, Comim CM, Valvassori SS, Reus GZ, Barbosa LM, Andreazza AC, Stertz L, Fries GR, Gavioli EC, Kapczinski F, Quevedo J (2008) Acute administration of ketamine induces antidepressant-like effects in the forced swimming test and increases BDNF levels in the rat hippocampus. Prog Neuropsychopharmacol Biol Psychiatry 32:140–144
Huang CC, Wei IH, Huang CL, Chen KT, Tsai MH, Tsai P, Tun R, Huang KH, Chang YC, Lane HY, Tsai GE (2013) Inhibition of glycine transporter-I as a novel mechanism for the treatment of depression. Biol Psychiatry 74:734–741
Ibrahim L, Diazgranados N, Franco-Chaves J, Brutsche N, Henter ID, Kronstein P, Moaddel R, Wainer I, Luckenbaugh DA, Manji HK, Zarate CA Jr (2012) Course of improvement in depressive symptoms to a single intravenous infusion of ketamine vs add-on riluzole: results from a 4-week, double-blind, placebo-controlled study. Neuropsychopharmacology 37:1526–1533
Ionescu DF, Luckenbaugh DA, Niciu MJ, Richards EM, Zarate CA Jr (2015) A single infusion of ketamine improves depression scores in patients with anxious bipolar depression. Bipolar Disord 17:438–443
Kanahara N, Shimizu E, Ohgake S, Fujita Y, Kohno M, Hashimoto T, Matsuzawa D, Shirayama Y, Hashimoto K, Iyo M (2008) Glycine and D-serine, but not D-cycloserine, attenuate prepulse inhibition deficits induced by NMDA receptor antagonist MK-801. Psychopharmacology (Berl) 198:363–374
Katsuki H, Watanabe Y, Fujimoto S, Kume T, Akaike A (2007) Contribution of endogenous glycine and d-serine to excitotoxic and ischemic cell death in rat cerebrocortical slice cultures. Life Sci 81:740–749
Kawaura K, Koike H, Kinoshita K, Kambe D, Kaku A, Karasawa J, Chaki S, Hikichi H (2015) Effects of a glycine transporter-1 inhibitor and D-serine on MK-801-induced immobility in the forced swimming test in rats. Behav Brain Res 278:186–192
Kim SJ, Lee L, Kim JH, Lee TH, Shim I (2013) Antidepressant-like effects of lycii radicis cortex and betaine in the forced swimming test in rats. Biomol Ther (Seoul) 21:79–83
Krystal JH, Karper LP, Seibyl JP, Freeman GK, Delaney R, Bremner JD, Heninger GR, Bowers MB Jr, Charney DS (1994) Subanesthetic effects of the noncompetitive NMDA antagonist, ketamine, in humans. Psychotomimetic, perceptual, cognitive, and neuroendocrine responses. Arch Gen Psychiatry 51:199–214
Krystal JH, Abi-Saab W, Perry E, D’Souza DC, Liu N, Gueorguieva R, McDougall L, Hunsberger T, Belger A, Levine L, Breier A (2005) Preliminary evidence of attenuation of the disruptive effects of the NMDA glutamate receptor antagonist, ketamine, on working memory by pretreatment with the group II metabotropic glutamate receptor agonist, LY354740, in healthy human subjects. Psychopharmacology (Berl) 179:303–309
Lee M-Y, Chen H-H (2014) Modulatory role for sarcosine, N, N-dimethylglycine and betaine in NMDA receptor activation. Basic Clin Pharmacol Toxicol 115:203 (Abstract)
Li N, Lee B, Liu RJ, Banasr M, Dwyer JM, Iwata M, Li XY, Aghajanian G, Duman RS (2010) mTOR-dependent synapse formation underlies the rapid antidepressant effects of NMDA antagonists. Science 329:959–964
Lima-Ojeda JM, Vogt MA, Pfeiffer N, Dormann C, Kohr G, Sprengel R, Gass P, Inta D (2013) Pharmacological blockade of GluN2B-containing NMDA receptors induces antidepressant-like effects lacking psychotomimetic action and neurotoxicity in the perinatal and adult rodent brain. Prog Neuropsychopharmacol Biol Psychiatry 45:28–33
Lin BF, Ou MC, Chung SS, Pang CY, Chen HH (2010) Adolescent toluene exposure produces enduring social and cognitive deficits in mice: an animal model of solvent-induced psychosis. World J Biol Psychiatry 11:792–802
Lipina T, Labrie V, Weiner I, Roder J (2005) Modulators of the glycine site on NMDA receptors, D-serine and ALX 5407, display similar beneficial effects to clozapine in mouse models of schizophrenia. Psychopharmacology (Berl) 179:54–67
Maeng S, Zarate CA Jr, Du J, Schloesser RJ, McCammon J, Chen G, Manji HK (2008) Cellular mechanisms underlying the antidepressant effects of ketamine: role of alpha-amino-3-hydroxy-5-methylisoxazole-4-propionic acid receptors. Biol Psychiatry 63:349–352
Malkesman O, Austin DR, Tragon T, Wang G, Rompala G, Hamidi AB, Cui Z, Young WS, Nakazawa K, Zarate CA, Manji HK, Chen G (2012) Acute D-serine treatment produces antidepressant-like effects in rodents. Int J Neuropsychopharmacol 15:1135–1148
Miwa M, Tsuboi M, Noguchi Y, Enokishima A, Nabeshima T, Hiramatsu M (2011) Effects of betaine on lipopolysaccharide-induced memory impairment in mice and the involvement of GABA transporter 2. J Neuroinflammation 8:153
Moskal JR, Burch R, Burgdorf JS, Kroes RA, Stanton PK, Disterhoft JF, Leander JD (2014) GLYX-13, an NMDA receptor glycine site functional partial agonist enhances cognition and produces antidepressant effects without the psychotomimetic side effects of NMDA receptor antagonists. Expert Opin Investig Drugs 23:243–254
Murrough JW, Perez AM, Pillemer S, Stern J, Parides MK, aan het Rot M, Collins KA, Mathew SJ, Charney DS, Iosifescu DV (2013) Rapid and longer-term antidepressant effects of repeated ketamine infusions in treatment-resistant major depression. Biol Psychiatry 74:250–256
Nakao S, Nagata A, Masuzawa M, Miyamoto E, Yamada M, Nishizawa N, Shingu K (2003) NMDA receptor antagonist neurotoxicity and psychotomimetic activity. Masui 52:594–602
Neymotin SA, Lazarewicz MT, Sherif M, Contreras D, Finkel LH, Lytton WW (2011) Ketamine disrupts theta modulation of gamma in a computer model of hippocampus. J Neurosci 31:11733–11743
Nugent AC, Diazgranados N, Carlson PJ, Ibrahim L, Luckenbaugh DA, Brutsche N, Herscovitch P, Drevets WC, Zarate CA Jr (2014) Neural correlates of rapid antidepressant response to ketamine in bipolar disorder. Bipolar Disord 16:119–128
Obeid R (2013) The metabolic burden of methyl donor deficiency with focus on the betaine homocysteine methyltransferase pathway. Nutrients 5:3481–3495
Papp M, Moryl E (1994) Antidepressant activity of non-competitive and competitive NMDA receptor antagonists in a chronic mild stress model of depression. Eur J Pharmacol 263:1–7
Papp M, Moryl E (1996) Antidepressant-like effects of 1-aminocyclopropanecarboxylic acid and D-cycloserine in an animal model of depression. Eur J Pharmacol 316:145–151
Park MJ, SRK HH, JHJ YCK (1994) Betaine attenuates glutamate-induced neurotoxicity in primary cultured brain cells. Arch Pharm Res 17:343–347
Percy AK, Lane JB (2005) Rett syndrome: model of neurodevelopmental disorders. J Child Neurol 20:718–721
Perry EB Jr, Cramer JA, Cho HS, Petrakis IL, Karper LP, Genovese A, O’Donnell E, Krystal JH, D’Souza DC, Yale Ketamine Study G (2007) Psychiatric safety of ketamine in psychopharmacology research. Psychopharmacology (Berl) 192:253–260
Qiao H, Noda Y, Kamei H, Nagai T, Furukawa H, Miura H, Kayukawa Y, Ohta T, Nabeshima T (2001) Clozapine, but not haloperidol, reverses social behavior deficit in mice during withdrawal from chronic phencyclidine treatment. Neuroreport 12:11–15
Roberts BM, Shaffer CL, Seymour PA, Schmidt CJ, Williams GV, Castner SA (2010) Glycine transporter inhibition reverses ketamine-induced working memory deficits. Neuroreport 21:390–394
Rybakowski JK, Permoda-Osip A, Skibinska M, Adamski R, Bartkowska-Sniatkowska A (2013) Single ketamine infusion in bipolar depression resistant to antidepressants: are neurotrophins involved? Hum Psychopharmacol 28:87–90
Santini AC, Pierantoni GM, Gerlini R, Iorio R, Olabinjo Y, Giovane A, Di Domenico M, Sogos C (2014) Glix 13, a new drug acting on glutamatergic pathways in children and animal models of autism spectrum disorders. BioMed research international 2014:234295
Thakurta RG, Das R, Bhattacharya AK, Saha D, Sen S, Singh OP, Bisui B (2012) Rapid response with ketamine on suicidal cognition in resistant depression. Indian J Psychol Med 34:170–175
Thomson AM, West DC, Lodge D (1985) An N-methylaspartate receptor-mediated synapse in rat cerebral cortex: a site of action of ketamine? Nature 313:479–481
van der Westhuyzen J, Metz J (1984) Betaine delays the onset of neurological impairment in nitrous oxide-induced vitamin B-12 deficiency in fruit bats. J Nutr 114:1106–1111
Vollenweider FX, Geyer MA (2001) A systems model of altered consciousness: integrating natural and drug-induced psychoses. Brain Res Bull 56:495–507
Wan LB, Levitch CF, Perez AM, Brallier JW, Iosifescu DV, Chang LC, Foulkes A, Mathew SJ, Charney DS, Murrough JW (2014) Ketamine safety and tolerability in clinical trials for treatment-resistant depression. J Clin Psychiatry
Yang SY, Hong CJ, Huang YH, Tsai SJ (2010) The effects of glycine transporter I inhibitor, N-methylglycine (sarcosine), on ketamine-induced alterations in sensorimotor gating and regional brain c-Fos expression in rats. Neurosci Lett 469:127–130
Zhou W, Wang N, Yang C, Li XM, Zhou ZQ, Yang JJ (2013) Ketamine-induced antidepressant effects are associated with AMPA receptors-mediated upregulation of mTOR and BDNF in rat hippocampus and prefrontal cortex. Eur Psychiatry
Zhu WL, Wang SJ, Liu MM, Shi HS, Zhang RX, Liu JF, Ding ZB, Lu L (2013) Glycine site N-methyl-D-aspartate receptor antagonist 7-CTKA produces rapid antidepressant-like effects in male rats. J Psychiatry Neurosci 38:306–316
Zigman D, Blier P (2013) Urgent ketamine infusion rapidly eliminated suicidal ideation for a patient with major depressive disorder: a case report. J Clin Psychopharmacol 33:270–272
Acknowledgments
This work was supported by the National Health Research Institutes (NP-103-PP-02) and the Ministry of Science and Technology (MOST-104-2314-B-400-007-MY22).
Authors’ contributions
MHC, YCC, and HHC were responsible for the study concept and design. JCL and MYL contributed to the acquisition and analysis of the animal data and interpretation of the findings. JCL drafted the manuscript. YCC and HHC provided critical revision of the manuscript for important intellectual content. MHC contributed to reviewing and editing of the manuscript. All of the authors critically reviewed the content and approved the final version for publication.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Fig. S1
NMDAR glycine binding site antagonist 7-chlorokynurenic acid (7-CTKA) abolished the reversing effect of betaine on ketamine-induced cognitive deficits in the novel object recognition test. 7-CTKA (0, 0.3 and 1 mg/kg, i.p.) was given 30 min prior to betaine (30 mg/kg, i.p.) or saline, followed by administration of ketamine (30 mg/kg) or saline 30 min later. The training session was conducted 5 min after ketamine administration. The retention session was performed 24 h later. All values are expressed as the mean ± SEM. A mixed-design ANOVA revealed significant main effects of treatment (F5, 43 = 4.361, p < 0.01) and session (F1, 43 = 47.944, p < 0.001) and a significant treatment x session interaction (F5, 43 = 8.677, p < 0.001). 7-CTKA (1 mg/kg, i.p.) affected the novel object preferences and disrupted the reversing effect of betaine on ketamine-induced impairment in the retention session. ***p < 0.001 vs. Saline/Saline/Saline, ## p < 0.01, ### p < 0.001 vs. Saline/Saline /Ketamine, $$$ p < 0.001 vs. Saline /Betaine/Ketamine. (PPTX 110 kb)
Rights and permissions
About this article
Cite this article
Lin, JC., Lee, MY., Chan, MH. et al. Betaine enhances antidepressant-like, but blocks psychotomimetic effects of ketamine in mice. Psychopharmacology 233, 3223–3235 (2016). https://doi.org/10.1007/s00213-016-4359-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-016-4359-x