Abstract
Rationale
The co-abuse of ethanol (EtOH) and nicotine (NIC) increases the likelihood that an individual will relapse to drug use while attempting to maintain abstinence. There is limited research examining the consequences of long-term EtOH and NIC co-abuse.
Objectives
The current experiments determined the enduring effects of chronic EtOH, NIC, or EtOH + NIC intake on the reinforcing properties of NIC and glutamate (GLU) activity within the mesocorticolimbic (MCL) system.
Methods
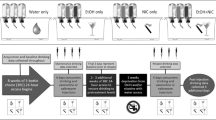
Alcohol-preferring (P) rats self-administered EtOH, Sacc + NIC, or EtOH + NIC combined for 10 weeks. The reinforcing properties of 0.1–3.0 μM NIC within the nucleus accumbens shell (AcbSh) were assessed following a 2–3-week drug-free period using intracranial self-administration (ICSA) procedures. The effects of EtOH, Sacc, Sacc + NIC, or EtOH + NIC intake on extracellular levels and clearance of glutamate (GLU) in the medial prefrontal cortex (mPFC) were also determined.
Results
Binge intake of EtOH (96–100 mg%) and NIC (21–27 mg/mL) were attained. All groups of P rats self-infused 3.0 μM NIC directly into the AcbSh, whereas only animals in the EtOH + NIC co-abuse group self-infused the 0.3 and 1.0 μM NIC concentrations. Additionally, self-administration of EtOH + NIC, but not EtOH, Sacc or Sacc + NIC, resulted in enduring increases in basal extracellular GLU levels in the mPFC.
Conclusions
Overall, the co-abuse of EtOH + NIC produced enduring neuronal alterations within the MCL which enhanced the rewarding properties of NIC in the AcbSh and elevated extracellular GLU levels within the mPFC.






Similar content being viewed by others
References
Benowitz NL (1997) Systemic absorption and effects of nicotine from smokeless tobacco. Adv Dent Res 11:336–341
Clark A, Little HJ (2004) Interaction between low concentrations of ethanol and nicotine on firing rate of ventral tegmental dopamine neurons. Drug Alcohol Depend 75:199–206
DiFranza JR, Guerrera MP (1990) Alcoholism and smoking. J Stud Alcohol 51:130–135
Ding ZM, Rodd ZA, Engleman EA, Bailey JA, Lahiri DK, McBride WJ (2013) Alcohol drinking and deprivation alter basal extracellular glutamate concentrations and clearance in the mesolimbic system of alcohol-preferring (P) rats. Addict Biol 18:297–306
Engleman EA, Ding ZM, Oster SM, Toalston JE, Bell RL, Murphy JM, McBride WJ, Rodd ZA (2009) Ethanol is self-administered into the nucleus accumbens shell, but not the core: evidence of genetic sensitivity. Alcohol Clin Exp Res 12:2162–71
Hauser SR, Katner SN, Deehan GA Jr, Ding ZM, Toalston JE, Scott BJ, Bell RL, McBride WJ, Rodd ZA (2012) Development of an oral operant nicotine/ethanol co-use model in alcohol-preferring (p) rats. Alcohol Clin Exp Res 36:1963–72
Hauser SR, Bracken AL, Deehan GA Jr, Toalston JE, Ding ZM, Truitt WA, Bell RL, McBride WJ, Rodd ZA (2014) Selective breeding for high alcohol preference increases the sensitivity of the posterior VTA to the reinforcing effects of nicotine. Addict Biol 19:800–11
Holmes A, Fitzgerald PJ, MacPherson KP, DeBrouse L, Colacicco G, Flynn SM, Masneuf S et al (2012) Chronic alcohol remodels prefrontal neurons and disrupts NMDAR-mediated fear extinction encoding. Nat Neurosci 15:1359–61
Hoover WB, Vertes RP (2007) Anatomical analysis of afferent projections to the medial prefrontal cortex in the rat. Brain Struct Funct 212:149–79
Innocent N, Linvingstone PD, Hone A, Kimura A, Young T, Whiteaker P, McIntosh JM, Wonnacott S (2008) alphaConotoxin ArlB[V11L, V16D] is a potent and selective antagonist at rat and human native alpha 7 nicotinic acetycholine receptors. J Pharmacol Exp Ther 327:529–37
John U, Hill A, Rumpf H, Meyer C (2003a) Alcohol high risk drinking, abuse, and dependence among tobacco smoking medical care patients and the general population. Drug Alcohol Depend 64:233–241
John U, Meyer C, Rumpf HJ, Schumann A, Thyrian JR, Hapke U (2003b) Strength of the relationship between tobacco smoking, nicotine dependence and the severity of alcohol dependence syndrome criteria in a population-based sample. Alcohol Alcohol 38:606–612
Kalivas PW (2009) The glutamate homeostasis hypothesis of addiction. Nat Rev Neurosci 10:561–72
Kalivas PW, O’Brien C (2008) Drug addiction as a pathology of staged neuroplasticity. Neuropsychopharmacology 33:166–80
Katner SN, Oster SM, Ding ZM, Deehan GA Jr, Toalston JE, Hauser SR, McBride WJ, Rodd ZA (2011) Alcohol-preferring (P) rats are more sensitive than Wistar rats to the reinforcing effects of cocaine self-administered directly into the nucleus accumbens shell. Pharmacol Biochem Behav 99:688–95
Lajtha A, Sershen H (2010) Nicotine: alcohol reward interactions. Neurochem Res 35:1248–58
Lallemand F, Ward RJ, De Witte P, Verbanck P (2011) Binge drinking +/− chronic nicotine administration alters extracellular glutamate and arginine levels in the nucleus accumbens of adult male and female Wistar rats. Alcohol Alcohol 46:373–82
Livingstone PD, Wonnacott S (2009) Nicotinic acetylcholine receptors and the ascending dopamine pathways. Biochem Pharmacol 78:744–55
Ma YY, Lee BR, Wang X, Guo C, Liu L, Cui R, Lan Y et al (2014) Bidirectional modulation of incubation of cocaine craving by silent synapse-base remodeling of prefrontal cortex to accumbens projections. Neuron 83:1453–67
Marshall DL, Redfern PH, Wonnacott S (1997) Presynaptic nicotinic modulation of dopamine release in the three ascending pathways studied by in vivo microdialysis: comparison of naive and chronic nicotine-treated rats. J Neurochem 68:1511–9
Melendez RI, Rodd-Henricks ZA, Engleman EA, Li TK, McBride WJ, Murphy JM (2002) Microdialysis of dopamine in the nucleus accumbens of alcohol-preferring (P) rats during anticipation and operant self-administration of ethanol. Alcohol Clin Exp Res 26:318–25
Melendez RI, Rodd ZA, McBride WJ, Murphy JM (2004) Involvement of the mesopallidal dopamine system in ethanol reinforcement. Alcohol 32:137–44
Meyer EL, Yoshikami D, McIntosh JM (2008) The neuronal nicotinic acetylcholine receptors α4* and α6* differentially modulate dopamine release in mouse striatal slices. J Neurochem 105:1761–69
Miller CA, Marshall JF (2004) Altered prelimbic cortex output during cue-elicited drug seeking. J Neurosci 4:6889–97
Mokdad AH, Marks JS, Stroup DF, Gerberding JL (2004) Actual causes of death in the United States, 2000. JAMA 291:1238–1245
National Institute on Drug Abuse (2014) The science of drug addiction: the basics. Available via http: http://www.drugabuse.gov/publications/media-guide/science-drug-abuse-addiction-basics. Accessed 10 Aug 2015
Nelson ME, Kuryatov A, Choi CH, Zhou Y, Lindstrom J (2003) Alternate stoichiometries of alpha4beta2 nicotinic acetylcholine receptors. Mol Pharmacol 63:332–41
Paterson NE, Semenova S, Gasparini F, Markou A (2003) The mGluR5 antagonist MPEP decreased nicotine self-administration in rats and mice. Psychopharmacology (Berl) 167:257–64
Paxinos G, Watson C (2005) The rat brain in stereotaxic coordinates, 5th edn. Academic Press, New York
Peters J, LaLumiere RT, Kalivas PW (2008) Infralimbic prefrontal cortex is responsible for inhibiting cocaine seeking in extinguished rats. J Neuorsci 28:6046–53
Research Institute for Laboratory Animal Research (2011) Guide for the Care and Use of Laboratory Animals, 8th edn. The National Academies Press, Washington, D.C
Rodd ZA, Melendez RI, Bell RL, Kuc KA, Zhang Y, Murphy JM, McBride WJ (2004) Intracranial self-administration of ethanol within the ventral tegmental area of male Wistar rats: evidence for involvement of dopamine neurons. J Neurosci 24:1050–7
Rodd ZA, Bell RL, Zhang Y, Murphy JM, Goldstein A, Zaffaroni A, Li TK, McBride (2005) Regional heterogeneity for the intracranial self-administration of ethanol and acetaldehyde within the ventral tegmental area of alcohol-preferring (P) rats: involvement of dopamine and serotonin. Neuropsychopharmacology 30:330–8
Rodd ZA, McKinzie DL, Bell RL, McQueen VK, Murphy JM, Schoepp DD, McBride WJ (2006) The metabotropic glutamate 2/3 receptor agonist LY404039 reduces alcohol-seeking but not alcohol self-administration in alcohol-preferring (P) rats. Behav Brain Res 171:207–15
Rodd-Henricks ZA, McKinzie DL, Li TK, Murphy JM, McBride WJ (2002) Cocaine is self-administered into the shell but not the core of the nucleus accumbens of Wistar rats. J Pharmacol Exp Ther 303:1216–26
Saab BJ, Maclean AJ, Kanisek M, Zurek AA, Martin LJ, Roder JC, Orser BA (2010) Short-term memory impairment after isoflurane in mice is prevented by the α5 γ-aminobutyric acid type A receptor inverse agonist L-655,708. Anethesiology 113:1061–71
Stefanik MT, Moussawi K, Kupchik YM, Smith KC, Miller RL, Huff ML, Deisseroth K, Kalivas PW, LaLumiere RT (2013) Optogenetic inhibition of cocaine seeking in rats. Addict Biol 18:50–53
Steketee JD (2003) Neurotransmitter systems of the medial prefrontal cortex: potential role in sensitization to psychostimulants. Brain Res Rev 41:203–28
Sun N, Laviolette SR, Addiction Research Group (2014) Dopamine receptor blockade modulates the rewarding and aversive properties of nicotine via dissociable neuronal activity patterns in the nucleus accumbens. Neuropsychopharmacology 12:2799–815
Toth E, Sershen H, Hashim A, Vizi ES, Lajtha A (1992) Effect of nicotine on extracellular levels of neurotransmitters assessed by microdialysis in various brain regions: role of glutamic acid. Neurochem Res 17:265–271
Truitt WA, Hauser SR, Deehan GA Jr, Toalston JE, Wilden JA, Bell RL, McBride WJ, Rodd ZA (2015) Ethanol and nicotine interaction within the posterior ventral tegmental area in male and female alcohol-preferring rats: evidence of synergy and differential gene activation in the nucleus accumbens shell. Psychopharmacology 232:639–49
Wang F, Chen H, Steketee JD, Sharp BM (2007) Upregulation of ionotropic glutamate receptor subunits within specific mesocorticolimbic regions during chronic nicotine self-administration. Neuropsychopharm 32:103–9
Wang F, Chen H, Sharp BM (2008) Neuroadaptive changes in the mesocortical glutamatergic system during chronic nicotine self-administration and after extinction in rats. J Neurochem 106:943–56
Zurek AA, Bridgwater EM, Orser BA (2012) Inhibition of α5 γ-aminobutyric acid type A receptors restores recognition memory after general anesthesia. Anesth Analg 114:845–55
Acknowledgments
The research in this manuscript was supported by grants from the National Institute on Alcohol Abuse and Alcoholism: AA019366, AA07611, AA07462, AA012262.
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Deehan, G.A., Hauser, S.R., Waeiss, R.A. et al. Co-administration of ethanol and nicotine: the enduring alterations in the rewarding properties of nicotine and glutamate activity within the mesocorticolimbic system of female alcohol-preferring (P) rats. Psychopharmacology 232, 4293–4302 (2015). https://doi.org/10.1007/s00213-015-4056-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-015-4056-1