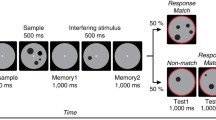
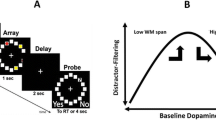
Abstract
Dopamine has long been implicated in the online maintenance of information across short delays. Specifically, dopamine has been proposed to modulate the strength of working memory representations in the face of intervening distracters. This hypothesis has not been tested in humans. We fill this gap using pharmacological neuroimaging. Healthy young subjects were scanned after intake of the dopamine receptor agonist bromocriptine or placebo (in a within-subject, counterbalanced, and double-blind design). During scanning, subjects performed a delayed match-to-sample task with face stimuli. A face or scene distracter was presented during the delay period (between the cue and the probe). Bromocriptine altered distracter-resistance, such that it impaired performance after face relative to scene distraction. Individual differences in the drug effect on distracter-resistance correlated negatively with drug effects on delay period signal in the prefrontal cortex, as well as on functional connectivity between the prefrontal cortex and the fusiform face area. These results provide evidence for the hypothesis that dopaminergic modulation of the prefrontal cortex alters resistance of working memory representations to distraction. Moreover, we show that the effects of dopamine on the distracter-resistance of these representations are accompanied by modulation of the functional strength of connections between the prefrontal cortex and stimulus-specific posterior cortex.





Similar content being viewed by others
References
Barde LHF, Thompson-Schill SL (2002) Models of functional organization of the lateral prefrontal cortex in verbal working memory: evidence in favor of the process model. J Cogn Neurosci 14:1054–1063. doi:10.1162/089892902320474508
Brozoski T, Brown R, Rosvold H, Goldman P (1979) Cognitive deficit caused by regional depletion of dopamine in prefrontal cortex of rhesus monkey. Science 205(80):929–932
Chao LL, Knight RT (1995) Human prefrontal lesions increase distractibility to irrelevant sensory inputs. Neuroreport 6:1605–1610
Clapp WC, Rubens MT, Gazzaley A (2010) Mechanisms of working memory disruption by external interference. Cereb Cortex 20:859–872. doi:10.1093/cercor/bhp150
Collins P, Wilkinson LS, Everitt BJ et al (2000) The effect of dopamine depletion from the caudate nucleus of the common marmoset (Callithrix jacchus) on tests of prefrontal cognitive function. Behav Neurosci 114:3–17
Cools R, D’Esposito M (2011) Inverted-U-shaped dopamine actions on human working memory and cognitive control. Biol Psychiatry 69:e113–e125. doi:10.1016/j.biopsych.2011.03.028
Cools R, Frank MJ, Gibbs SE et al (2009) Striatal dopamine predicts outcome-specific reversal learning and its sensitivity to dopaminergic drug administration. J Neurosci 29:1538–1543. doi:10.1523/JNEUROSCI.4467–08.2009
Cools R, Sheridan M, Jacobs E, D’Esposito M (2007) Impulsive personality predicts dopamine-dependent changes in frontostriatal activity during component processes of working memory. J Neurosci 27:5506–5514. doi:10.1523/JNEUROSCI.0601-07.2007
Cools R, Stefanova E, Barker RA et al (2002) Dopaminergic modulation of high-level cognition in Parkinson’s disease: the role of the prefrontal cortex revealed by PET. Brain 125:584–594. doi:10.1093/brain/awf052
Curtis CE, Rao VY, D’Esposito M (2004) Maintenance of spatial and motor codes during oculomotor delayed response tasks. J Neurosci 24:3944–3952. doi:10.1523/JNEUROSCI.5640–03.2004
Diamond A (2007) Consequences of variations in genes that affect dopamine in prefrontal cortex. Cereb Cortex 17(Suppl 1):i161–i170. doi:10.1093/cercor/bhm082
Druzgal TJ, D’Esposito M (2003) Dissecting contributions of prefrontal cortex and fusiform face area to face working memory. J Cogn Neurosci 15:771–784. doi:10.1162/089892903322370708
Durstewitz D, Kelc M, Güntürkün O (1999) A neurocomputational theory of the dopaminergic modulation of working memory functions. J Neurosci 19:2807–2822
Durstewitz D, Seamans JK (2008) The dual-state theory of prefrontal cortex dopamine function with relevance to catechol-o-methyltransferase genotypes and schizophrenia. Biol Psychiatry 64:739–749. doi:10.1016/j.biopsych.2008.05.015
Durstewitz D, Seamans JK, Sejnowski TJ (2000) Dopamine-mediated stabilization of delay-period activity in a network model of prefrontal cortex. J Neurophysiol 83:1733–1750
Feredoes E, Heinen K, Weiskopf N, et al. (2011) Causal evidence for frontal involvement in memory target maintenance by posterior brain areas during distracter interference of visual working memory. PNAS. doi: 10.1073/pnas.1106439108
Floresco SB (2013) Prefrontal dopamine and behavioral flexibility: shifting from an “inverted-U” toward a family of functions. Front Neurosci 7:62. doi:10.3389/fnins.2013.00062
Fuster JM, Alexander G. (1971) Neuron activity related to short-term memory. Science (80) 652–654.
Gazzaley A, Rissman J, D’Esposito M (2004) Functional connectivity during working memory maintenance. Cogn Affect Behav Neurosci 4:580–599
Gibbs SEB, D’Esposito M (2005) A functional MRI study of the effects of bromocriptine, a dopamine receptor agonist, on component processes of working memory. Psychopharmacology (Berl) 180:644–653. doi:10.1007/s00213-005-0077-5
Jacobsen CF (1936) Studies of Cerebral Function in Primates. Psychol, Comp
Jha AP, Fabian SA, Aguirre GK (2004) The role of prefrontal cortex in resolving distractor interference. Cogn Affect Behav Neurosci 4:517–527
Jha AP, McCarthy G (2000) The influence of memory load upon delay-interval activity in a working-memory task: an event-related functional MRI study. J Cogn Neurosci 12(Suppl 2):90–105. doi:10.1162/089892900564091
Kanwisher N, McDermott J, Chun MM (1997) The fusiform face area: a module in human extrastriate cortex specialized for face perception. J Neurosci 17:4302–4311
Kvernmo T, Härtter S, Burger E (2006) A review of the receptor-binding and pharmacokinetic properties of dopamine agonists. Clin Ther 28:1065–1078. doi:10.1016/j.clinthera.2006.08.004
Luciana M, Collins P (1992) Dopaminergic modulation of working n memory for spatial but not object cues in normal humans. J Cogn Neurosci 330–347
Malmo RB (1942) Interference factors in delayed response in monkeys after removal of frontal lobes. J Neurophysiol 5:295–308
Mattay V, Tessitore A (2002) Dopaminergic modulation of cortical function in patients with Parkinson’s disease. Ann Neurol 51:156–164. doi:10.1002/ana.10078
Mehta MA, Riedel WJ (2006) Dopaminergic enhancement of cognitive function. Curr Pharm Des 12:2487–2500
Miller EK, Cohen JD (2001) An integrative theory of prefrontal cortex function. Annu Rev Neurosci 24:167–202. doi:10.1146/annurev.neuro.24.1.167
Miller EK, Erickson CA, Desimone R (1996) Neural mechanisms of visual working memory in prefrontal cortex of the macaque. J Neurosci 16:5154–5167
Patton JH, Stanford MS, Barratt ES (1995) Factor structure of the Barratt impulsiveness scale. J Clin Psychol 51:768–774
Pessoa L, Gutierrez E, Bandettini P, Ungerleider L (2002) Neural correlates of visual working memory: fMRI amplitude predicts task performance. Neuron 35:975–987
Postle BR, Zarahn E, D’Esposito M (2000) Using event-related fMRI to assess delay-period activity during performance of spatial and nonspatial working memory tasks. Brain Res Brain Res Protoc 5:57–66
Rabbit P (1966) Errors and error correction in choice-response tasks. J Exp Psychol 71:264–272
Ranganath C, Cohen MX, Dam C, D’Esposito M (2004) Inferior temporal, prefrontal, and hippocampal contributions to visual working memory maintenance and associative memory retrieval. J Neurosci 24:3917–3925. doi:10.1523/JNEUROSCI.5053-03.2004
Sawaguchi T, Goldman-Rakic PS (1991) D1 dopamine receptors in prefrontal cortex: involvement in working memory. Science 251(80):947–950
Seamans JK, Yang CR (2004) The principal features and mechanisms of dopamine modulation in the prefrontal cortex. Prog Neurobiol 74:1–58. doi:10.1016/j.pneurobio.2004.05.006
Seeman P, van Tol HM (1994) Dopamine receptor pharmacology. TiPS 15:264–270. doi:10.1016/S0072–9752(07)83004–1
Servan-Schreiber D, Printz H, Cohen JD (1990) A network model of catecholamine effects: gain, signal-to-noise ratio, and behavior. Science 249(80):892–895
Sreenivasan KK, Curtis CE, D’Esposito M (2014) Revisiting the role of persistent neural activity during working memory. Trends Cogn Sci 18:82–89. doi:10.1016/j.tics.2013.12.001
Wang M, Vijayraghavan S, Goldman-Rakic PS (2004) Selective D2 receptor actions on the functional circuitry of working memory. Science 303(80):853–856. doi:10.1126/science.1091162
Yoon JH, Curtis CE, D’Esposito M (2006) Differential effects of distraction during working memory on delay-period activity in the prefrontal cortex and the visual association cortex. Neuroimage 29:1117–1126. doi:10.1016/j.neuroimage.2005.08.024
Zarahn E, Aguirre G, D’Esposito M (1997) A trial-based experimental design for fMRI. Neuroimage 6:122–138. doi:10.1006/nimg.1997.0279
Author information
Authors and Affiliations
Corresponding author
Additional information
Mirjam Bloemendaal and Martine R. van Schouwenburg contributed equally to the manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 1197 kb)
Rights and permissions
About this article
Cite this article
Bloemendaal, M., van Schouwenburg, M.R., Miyakawa, A. et al. Dopaminergic modulation of distracter-resistance and prefrontal delay period signal. Psychopharmacology 232, 1061–1070 (2015). https://doi.org/10.1007/s00213-014-3741-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-014-3741-9