Abstract
Rationale
Human motivation and decision-making is influenced by the interaction of Pavlovian and instrumental systems. The neurotransmitters dopamine and serotonin have been suggested to play a major role in motivation and decision-making, but how they affect this interaction in humans is largely unknown.
Objective
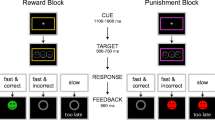
We investigated the effect of these neurotransmitters in a general Pavlovian-to-instrumental transfer (PIT) task which measured the nonspecific effect of appetitive and aversive Pavlovian cues on instrumental responses.
Methods
For that purpose, we used selective dietary depletion of the amino acid precursors of serotonin and dopamine: tryptophan (n = 34) and tyrosine/phenylalanine (n = 35), respectively, and compared the performance of these groups to a control group (n = 34) receiving a nondepleted (balanced) amino acid drink.
Results
We found that PIT differed between groups: Relative to the control group that exhibited only appetitive PIT, we found reduced appetitive PIT in the tyrosine/phenylalanine-depleted group and enhanced aversive PIT in the tryptophan-depleted group.
Conclusions
These results demonstrate a differential involvement of serotonin and dopamine in motivated behavior. They suggest that reductions in serotonin enhance the motivational influence of aversive stimuli on instrumental behavior and do not affect the influence of appetitive stimuli, while reductions in dopamine diminish the influence of appetitive stimuli. No conclusions could be drawn about how dopamine affects the influence of aversive stimuli. The interplay of both neurotransmitter systems allows for flexible and adaptive responses depending on the behavioral context.



Similar content being viewed by others
References
Allman MJ, DeLeon IG, Cataldo MF et al (2010) Learning processes affecting human decision making: an assessment of reinforcer-selective Pavlovian-to-instrumental transfer following reinforcer devaluation. J Exp Psychol Anim Behav Process 36:402–408
Ardis TC, Cahir M, Elliott JJ et al (2009) Effect of acute tryptophan depletion on noradrenaline and dopamine in the rat brain. J Psychopharmacol (Oxf) 23:51–55
Biskup CS, Sánchez CL, Arrant A et al (2012) Effects of acute tryptophan depletion on brain serotonin function and concentrations of dopamine and norepinephrine in C57BL/6J and BALB/cJ mice. PLoS One 7:e35916
Booij L, Van der Does W, Benkelfat C et al (2002) Predictors of mood response to acute tryptophan depletion: a reanalysis. Neuropsychopharmacology 27:852–861
Borkenau P, Ostendorf F (1993) NEO-Fünf-Faktoren-Inventar (NEO-FFI) nach Costa und McCrae. Hogrefe, Göttingen
Boureau Y-L, Dayan P (2011) Opponency revisited: competition and cooperation between dopamine and serotonin. Neuropsychopharmacology 36:74–97
Braver TS, Krug MK, Chiew KS et al (2014) Mechanisms of motivation–cognition interaction: challenges and opportunities. Cogn Affect Behav Neurosci 1–30
Bray S, Rangel A, Shimojo S et al (2008) The neural mechanisms underlying the influence of Pavlovian cues on human decision making. J Neurosci 28:5861–5866
Bromberg-Martin ES, Matsumoto M, Hikosaka O (2010) Dopamine in motivational control: rewarding, aversive, and alerting. Neuron 68:815–834
Carpenter LL, Anderson GM, Pelton GH et al (1998) Tryptophan depletion during continuous CSF sampling in healthy human subjects. Neuropsychopharmacology 19:26–35
Cloninger CR, Przybeck TR, Svrakic DM (1994) The Temperament and Character Inventory (TCI): a guide to its development and use. Center for Psychobiology of Personality. Washington University, St. Louis
Colwill RM, Rescorla RA (1988) Associations between the discriminative stimulus and the reinforcer in instrumental learning. J Exp Psychol Anim Behav Process 14:155–164
Cools R, Blackwell A, Clark L et al (2005) Tryptophan depletion disrupts the motivational guidance of goal-directed behavior as a function of trait impulsivity. Neuropsychopharmacology 30:1362–1373
Cools R, Roberts AC, Robbins TW (2008) Serotoninergic regulation of emotional and behavioural control processes. Trends Cogn Sci 12:31–40
Cools R, Nakamura K, Daw ND (2011) Serotonin and dopamine: unifying affective, activational, and decision functions. Neuropsychopharmacology 36:98–113
Corbit LH, Balleine BW (2005) Double dissociation of basolateral and central amygdala lesions on the general and outcome-specific forms of Pavlovian-instrumental transfer. J Neurosci 25:962–970
Corbit LH, Janak PH (2007) Inactivation of the lateral but not medial dorsal striatum eliminates the excitatory impact of Pavlovian stimuli on instrumental responding. J Neurosci 27:13977–13981
Corbit LH, Muir JL, Balleine BW (2001) The role of the nucleus accumbens in instrumental conditioning: evidence of a functional dissociation between accumbens core and shell. J Neurosci 21:3251–3260
Corbit LH, Janak PH, Balleine BW (2007) General and outcome-specific forms of Pavlovian-instrumental transfer: the effect of shifts in motivational state and inactivation of the ventral tegmental area. Eur J Neurosci 26:3141–3149
Costa PT, McCrae RR (1992) Revised NEO Personality Inventory (NEO PI-R) and NEO Five-Factor Inventory (NEO-FFI). Psychological Assessment Resources, Odessa
Crockett MJ, Clark L, Robbins TW (2009) Reconciling the role of serotonin in behavioral inhibition and aversion: acute tryptophan depletion abolishes punishment-induced inhibition in humans. J Neurosci 29:11993–11999
Crockett MJ, Clark L, Apergis-Schoute AM et al (2012) Serotonin modulates the effects of Pavlovian aversive predictions on response vigor. Neuropsychopharmacology 37:2244–2252
Dayan P, Huys QJ (2008) Serotonin, inhibition, and negative mood. PLoS Comput Biol 4:e4
Dayan P, Huys QJ (2009) Serotonin in affective control. Annu Rev Neurosci 32:95–126
Dayan P, Niv Y, Seymour B, Daw ND (2006) The misbehavior of value and the discipline of the will. Neural Netw 19:1153–1160
Deakin JW, Graeff FG (1991) 5-HT and mechanisms of defence. J Psychopharmacol (Oxf) 5:305–315
Delamater AR, Holland PC (2008) The influence of CS-US interval on several different indices of learning in appetitive conditioning. J Exp Psychol Anim Behav Process 34:202
Dickinson A, Balleine B (2002) The role of learning in the operation of motivational systems. In: Gallistel CR (ed) Stevens’ handbook of experimental psychology: learning, motivation, and emotion. John Wiley & Sons, New York, pp 497–534
Estes WK (1943) Discriminative conditioning. I. A discriminative property of conditioned anticipation. J Exp Psychol 32:150–155
Fernstrom JD (1978) Diet-induced changes in plasma amino acid pattern: effects on the brain uptake of large neutral amino acids, and on brain serotonin synthesis. J Neural Transm Suppl 55–67
Geurts DE, Huys QJ, den Ouden HE, Cools R (2013a) Aversive Pavlovian control of instrumental behavior in humans. J Cogn Neurosci 25:1428–1441
Geurts DE, Huys QJ, den Ouden HE, Cools R (2013b) Serotonin and aversive Pavlovian control of instrumental behavior in humans. J Neurosci 33:18932–18939
Guitart-Masip M, Duzel E, Dolan R, Dayan P (2014) Action versus valence in decision making. Trends Cogn Sci 18:194–202
Habib M (2004) Athymhormia and disorders of motivation in basal ganglia disease. J Neuropsychiatry Clin Neurosci 16:509–524
Hall J, Parkinson JA, Connor TM et al (2001) Involvement of the central nucleus of the amygdala and nucleus accumbens core in mediating Pavlovian influences on instrumental behaviour. Eur J Neurosci 13:1984–1992
Hautzinger M, Bailer M, Hofmeister D, Keller F (1993) Allgemeine Depressionsskala (ADS). Psychiatr Prax 39:302–304
Hindi Attar C, Finckh B, Büchel C (2012) The influence of serotonin on fear learning. PLoS ONE 7:e42397
Hogarth L, Dickinson A, Wright A et al (2007) The role of drug expectancy in the control of human drug seeking. J Exp Psychol Anim Behav Process 33:484–496
Holland PC (2004) Relations between Pavlovian-instrumental transfer and reinforcer devaluation. J Exp Psychol Anim Behav Process 30:104–117
Holland PC, Gallagher M (2003) Double dissociation of the effects of lesions of basolateral and central amygdala on conditioned stimulus-potentiated feeding and Pavlovian-instrumental transfer. Eur J Neurosci 17:1680–1694
Holmes NM, Marchand AR, Coutureau E (2010) Pavlovian to instrumental transfer: a neurobehavioural perspective. Neurosci Biobehav Rev 34:1277–1295
Hood SD, Bell CJ, Nutt DJ (2005) Acute tryptophan depletion. Part I: rationale and methodology. Aust N Z J Psychiatr 39:558–564
Huys QJ, Cools R, Golzer M et al (2011) Disentangling the roles of approach, activation and valence in instrumental and Pavlovian responding. PLoS Comput Biol 7:e1002028
Le Masurier M, Zetterström T, Cowen P, Sharp T (2013) Tyrosine-free amino acid mixtures reduce physiologically-evoked release of dopamine in a selective and activity-dependent manner. J Psychopharmacol (Oxf) 28:561–569
Lewis AH, Niznikiewicz MA, Delamater AR, Delgado MR (2013) Avoidance-based human Pavlovian-to-instrumental transfer. Eur J Neurosci 38:3740–3748
Lex A, Hauber W (2008) Dopamine D1 and D2 receptors in the nucleus accumbens core and shell mediate Pavlovian-instrumental transfer. Learn Mem 15:483–491
Loftus GR, Masson ME (1994) Using confidence intervals in within-subject designs. Psychon Bull Rev 1:476–490
Lovibond PF (1983) Facilitation of instrumental behavior by a Pavlovian appetitive conditioned stimulus. J Exp Psychol Anim Behav Process 9:225
McTavish SF, Cowen PJ, Sharp T (1999) Effect of a tyrosine-free amino acid mixture on regional brain catecholamine synthesis and release. Psychopharmacology (Berlin) 141:182–188
Mendelsohn D, Riedel WJ, Sambeth A (2009) Effects of acute tryptophan depletion on memory, attention and executive functions: a systematic review. Neurosci Biobehav Rev 33:926–952
Moja EA, Cipolla P, Castoldi D, Tofanetti O (1989) Dose-response decrease in plasma tryptophan and in brain tryptophan and serotonin after tryptophan-free amino acid mixtures in rats. Life Sci 44:971–976
Moja EA, Lucini V, Benedetti F, Lucca A (1996) Decrease in plasma phenylalanine and tyrosine after phenylalanine-tyrosine free amino acid solutions in man. Life Sci 58:2389–2395
Murschall A, Hauber W (2006) Inactivation of the ventral tegmental area abolished the general excitatory influence of Pavlovian cues on instrumental performance. Learn Mem 13:123–126
Nadler N, Delgado MR, Delamater AR (2011) Pavlovian to instrumental transfer of control in a human learning task. Emotion 11:1112–1123
Niv Y, Daw ND, Joel D, Dayan P (2007) Tonic dopamine: opportunity costs and the control of response vigor. Psychopharmacology (Berlin) 191:507–520
Paredes-Olay C, Abad MJ, Gámez M, Rosas JM (2002) Transfer of control between causal predictive judgments and instrumental responding. Anim Learn Behav 30:239–248
Prévost C, Liljeholm M, Tyszka JM, O’Doherty JP (2012) Neural correlates of specific and general Pavlovian-to-instrumental transfer within human amygdalar subregions: a high-resolution fMRI study. J Neurosci 32:8383–8390
Radloff LS (1977) The CES-D scale a self-report depression scale for research in the general population. Appl Psychol Meas 1:385–401
Reilly JG, McTavish SFB, Young AH (1997) Rapid depletion of plasma tryptophan: a review of studies and experimental methodology. J Psychopharmacol (Oxf) 11:381–392
Rescorla RA, Solomon RL (1967) Two-process learning theory: relationships between Pavlovian conditioning and instrumental learning. Psychol Rev 74:151–182
Richter J, Eisemann M, Richter G, Cloninger CR (1999) Das Temperament-und Charakter-Inventar (TCI). Swets Test Services, Frankfurt/Main, Germany
Robbins TW, Everitt BJ (2007) A role for mesencephalic dopamine in activation: commentary on Berridge (2006). Psychopharmacology (Berlin) 191:433–437
Roiser JP, Blackwell AD, Cools R et al (2006) Serotonin transporter polymorphism mediates vulnerability to loss of incentive motivation following acute tryptophan depletion. Neuropsychopharmacology 31:2264–2272
Roth M, Hammelstein P (2003) Sensation Seeking - Konzeption, Diagnostik und Anwendung. Hogrefe, Göttingen
Salamone JD, Correa M (2002) Motivational views of reinforcement: implications for understanding the behavioral functions of nucleus accumbens dopamine. Behav Brain Res 137:3–25
Salamone JD, Correa M (2012) The mysterious motivational functions of mesolimbic dopamine. Neuron 76:470–485
Salamone JD, Correa M, Mingote SM et al (2006) Nucleus accumbens dopamine and the forebrain circuitry involved in behavioral activation and effort-related decision making: implications for understanding anergia and psychomotor slowing in depression. Curr Psychiatr Rev 2:267–280
Salamone JD, Correa M, Farrar A, Mingote SM (2007) Effort-related functions of nucleus accumbens dopamine and associated forebrain circuits. Psychopharmacology (Berlin) 191:461–482
Sanders AC, Hussain AJ, Hen R, Zhuang X (2007) Chronic blockade or constitutive deletion of the serotonin transporter reduces operant responding for food reward. Neuropsychopharmacology 32:2321–2329
Schultz W (2007a) Behavioral dopamine signals. Trends Neurosci 30:203–210
Schultz W (2007b) Multiple dopamine functions at different time courses. Annu Rev Neurosci 30:259–288
Sheehan BD, Tharyan P, McTavish SFB et al (1996) Use of a dietary manipulation to deplete plasma tyrosine and phenylalanine in healthy subjects. J Psychopharmacol (Oxf) 10:231–234
Soubrié P (1986) Reconciling the role of central serotonin neurons in human and animal behavior. Behav Brain Sci 9:319–363
Stancampiano R, Melis F, Sarais L et al (1997) Acute administration of a tryptophan-free amino acid mixture decreases 5-HT release in rat hippocampus in vivo. Am J Physiol Regul Integr Comp Physiol 272:R991–R994
Steyer R (1997) Der Mehrdimensionale Befindlichkeitsfragebogen. Hogrefe, Göttingen
Talmi D, Seymour B, Dayan P, Dolan RJ (2008) Human Pavlovian-instrumental transfer. J Neurosci 28:360–368
Tops M, Russo S, Boksem MA, Tucker DM (2009) Serotonin: modulator of a drive to withdraw. Brain Cogn 71:427–436
Wise RA (2004) Dopamine, learning and motivation. Nat Rev Neurosci 5:483–494
Wyvell CL, Berridge KC (2000) Intra-accumbens amphetamine increases the conditioned incentive salience of sucrose reward: enhancement of reward “wanting” without enhanced “liking” or response reinforcement. J Neurosci 20:8122–8130
Young SN (2013) Acute tryptophan depletion in humans: a review of theoretical, practical and ethical aspects. J Psychiatry Neurosci JPN 38:294–305
Young SN, Smith SE, Pihl RO, Ervin FR (1985) Tryptophan depletion causes a rapid lowering of mood in normal males. Psychopharmacology (Berlin) 87:173–177
Zuckerman M (1996) Item revisions in the Sensation Seeking Scale form V (SSS-V). Personal Individ Differ 20:515
Acknowledgments
We thank Arnina Frank and Friederike Irmen for their help in data collection, Timo Krämer for taking blood samples, Ulrike Schwarze for introducing us to the depletion protocol, and Dirk Geurts for useful discussions. This work was supported by the “Bernstein Award for Computational Neuroscience” by the German Ministry of Education and Research (BMBF) awarded to JG (Grant No. 01GQ1006).
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 279 kb)
Rights and permissions
About this article
Cite this article
Hebart, M.N., Gläscher, J. Serotonin and dopamine differentially affect appetitive and aversive general Pavlovian-to-instrumental transfer. Psychopharmacology 232, 437–451 (2015). https://doi.org/10.1007/s00213-014-3682-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-014-3682-3