Abstract
Rationale
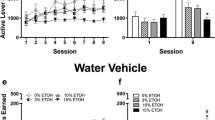
Operant self-administration (SA) is an important model of motivation to consume ethanol (EtOH), but low rates of voluntary consumption in rats are thought to necessitate water deprivation and saccharin/sucrose fading for acquisition of responding.
Objectives
Here, we sought to devise an effective model of SA that does not use water deprivation or saccharin/sucrose fading.
Methods
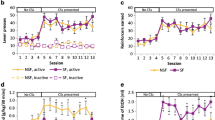
First, we tested if Wistar rats would acquire and maintain SA behavior of 20 % EtOH under two conditions, water deprivation (WD) and non-water deprivation (NWD). Second, we tested the efficacy of our SA procedure by confirming a prior study which found that the NK1 antagonist L822429 specifically blocked stress-induced reinstatement of EtOH seeking but not SA. Finally, we assessed the effect of naltrexone, an FDA-approved medication for alcohol dependence that has been shown to suppress EtOH SA in rodents.
Results
Lever presses (LPs) and rewards were consistent with previous reports that utilized WD and saccharin/sucrose fading. Similar to previous findings, we found that L822429 blocked stress-induced reinstatement but not baseline SA of 20 % EtOH. Moreover, naltrexone dose-dependently decreased alcohol intake and motivation to consume alcohol for rats that are self-administering 20 % EtOH.
Conclusions
Our findings provide a method for voluntary oral EtOH SA in rats that is convenient for experimenters and eliminates the potential confound of sweeteners in EtOH-operant SA studies. Unlike models that use intermittent access to 20 % EtOH, this method does not induce escalation, and based on pharmacological experiments, it appears to be driven by the positive reinforcing effects of EtOH.



Similar content being viewed by others
References
Augier E, Vouillac C, Ahmed SH (2012) Diazepam promotes choice of abstinence in cocaine self-administering rats. Addict Biol 17:378–391
Avena NM (2010) The study of food addiction using animal models of binge eating. Appetite 55:734–737
Avena NM, Rada P, Hoebel BG (2008) Evidence for sugar addiction: behavior effects of excessive intermittent sugar intake. Neurosci Behav Rev 32:20–39
Cantin L, Lenoir M, Augier E, Vanhille N, Dubreucq S, Serre F, Vouillac C, Ahmed SH (2010) Cocaine is low on the value ladder of rats: possible evidence for resilience to addiction. PLoS One 5(7):e11592
Carcinella L, Yowell QV, Ron D (2011) Regulation of operant oral ethanol self-administration: a dose–response curve study in rats. Alcohol Clin Exp Res 35:116–125
Czachowski CL, Delory MJ (2009) Acamprosate and naltrexone treatment effects on ethanol and sucrose seeking and intake in ethanol-dependent and nondependent rats. Psychopharmacology 204:335–348
De Bruin NM, McCreary AC, van Loevezijn A, de Vries TJ, Venhorst J, van Drimmelen M, Kruse CG (2013) A novel highly selective 5-HT6 receptor antagonist attenuates ethanol and nicotine seeking but does not affect inhibitory response control in Wistar rats. Behav Brain Res 236(1):157–165
Gonzales RA, Weiss F (1998) Suppression of ethanol-reinforced behavior by naltrexone is associated with attenuation of the ethanol-induced increase in dialysate dopamine levels in the nucleus accumbens. The Journal of neuroscience: the official journal of the Society for Neuroscience 18:10663–10671
Koob G, Weiss F (1990) Pharmacology of drug self-administration. Alcohol 7(3):193–197
Lenoir M, Serre F, Cantin L, Ahmed SH (2007) Intense sweetness surpasses cocaine reward. PLoS One 2:e698
Morgan D, Sizemore GM (2011) Animal models of addiction: fat and sugar. Curr Pharm Des 17:1168–1172
Radke AK, Holtz NA, Gewirtz JC, Carroll ME (2013) Reduced emotional signs of opiate withdrawal in rats selectively bred for low (LoS) versus high (HiS) saccharin intake. Psychopharmacology 227:116–126
Richardson NR, Roberts DC (1996) Progressive ratio schedules in drug self-administration studies in rats: a method to evaluate reinforcing efficacy. J Neurosci Methods 66:1–11
Samson HH (1986) Initiation of EtOH reinforcement using a sucrose-substitution procedure in food- and water-sated rats. Alcohol Clin Exp Res 10:436–442
Samson HH, Pfeffer AO, Tolliver GA (1988) Oral EtOH self-administration in rats: models of EtOH-seeking behavior. ACER 12:591–598
Schank JR, Pickens CL, Rowe KE, Cheng K, Thorsell A, Rice KC, Shaham Y, Heilig M (2011) Stress-induced reinstatement of alcohol-seeking in rats is selectively suppressed by the neurokinin 1 (NK1) antagonist L822429. Psychopharmacology 218(1):111–119
Schank JR, Tapocik JD, Barbier E, Damadzic R, Eskay RL, Sun H, Rowe KE, King CE, Yao M, Flanigan ME, Solomon MG, Karlsson C, Cheng K, Rice KC, Heilig M (2013) Tacr1 gene variation and neurokinin 1 receptor expression is associated with antagonist efficacy in genetically selected alcohol-preferring rats. Biol Psychiatry 73:774–781
Schulteis G, Hyytia P, Heinrichs SC, Koob GF (1996) Effects of chronic ethanol exposure on oral selfadministration of ethanol or saccharin by Wistar rats. Alcohol Clin Exp Res 20:164–171
Simms JA, Bito-Onon J, Chatterjee S, Bartlett S (2010) Long-evans rats acquire operant self-administration of 20 % ethanol without sucrose fading. Neuropsychopharmacology 35:1453–1463
Spangler R, Wittkowski KM, Goddard NL, Avena NM, Hoebel BG, Leibowitz SF (2004) Opiate-like effects of sugar on gene expression in reward areas of the rat brain. Brain Res Mol Brain Res 124:134–142
Stromberg MF, Volpicelli JR, O'Brien CP (1998) Effects of naltrexone administered repeatedly across 30 or 60 days on ethanol consumption using a limited access procedure in the rat. Alcohol Clin Exp Res 22:2186–2191
Weiss F, Koob GF (1991) The neuropharmacology of ethanol self-administration. In: Meyer RF, Koob GF, Lewis MJ, Paul S (eds) Neuropharmacology of Ethanol. Birkhauser; Boston, pp 125–162
Weiss F, Mitchiner M, Bloom FE, Koob GF (1990) Free-choice responding for ethanol versus water in alcohol preferring (P) and unselected Wistar rats is differentially modified by naloxone, bromocriptine, and methysergide. Psychopharmacology 101(2):178–186
Weiss F, Lorang MT, Bloom FE, Koob GF (1993) Oral alcohol self-administration stimulates dopamine release in the rat nucleus accumbens: genetic and motivational determinants. J Pharmacol Exp Ther 267(1):250–258
Acknowledgments
This work was supported by the Intramural Research Programs of the National Institute on Drug Abuse and the National Institute on Alcohol Abuse and Alcoholism.
Author information
Authors and Affiliations
Corresponding author
Additional information
E. Augier and M. Flanigan equally contributed to this study.
Rights and permissions
About this article
Cite this article
Augier, E., Flanigan, M., Dulman, R.S. et al. Wistar rats acquire and maintain self-administration of 20 % ethanol without water deprivation, saccharin/sucrose fading, or extended access training. Psychopharmacology 231, 4561–4568 (2014). https://doi.org/10.1007/s00213-014-3605-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-014-3605-3