Abstract
Rationale
Previous studies demonstrate the neuroprotective effects of progesterone in numerous animal injury models, but a systematic dose–response study in a transient ischemic stroke model is lacking.
Objectives
We investigated the effects of progesterone at different doses on post-stroke brain infarction and functional deficits in middle-aged rats.
Methods
Cerebral ischemia was induced in 13-month-old male Sprague–Dawley rats by right middle cerebral artery occlusion for 2 h followed by reperfusion. Rats received intraperitoneal injections of 8, 16, or 32 mg/kg of progesterone (P8, P16, P32) or vehicle at 2 h post-occlusion followed by subcutaneous injections at 6 h and every 24 h post-injury for 7 days. Functional recovery was evaluated at intervals over 22 days using motor, sensory, and cognitive tests. Infarct size was evaluated at 22 days post-stroke.
Results
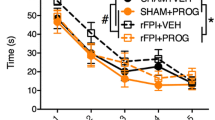
Repeated-measures ANOVA showed significant group effects on grip strength, rotarod, and sensory neglect. All progesterone-treated groups had improved (p < 0.05) spatial memory performance. The P8 and P16 groups showed maximum improvement in long-term memory compared to vehicle. Significant (p < 0.05) gait impairments were observed in the vehicle group compared to shams. Animals receiving the P8 dose showed maximum gait improvement compared to vehicle. Post hoc analysis revealed that the P8 and P16 groups showed significant attenuation in infarct volume compared to vehicle. Animals receiving the P32 dose did not show any effect on infarct volume.
Conclusions
Although all doses were somewhat effective, progesterone given at 8 mg/kg led to the most consistent improvements across a panel of behavioral/functional tests and reduced the severity of ischemic infarct injury.







Similar content being viewed by others
References
Albers GW, Goldstein LB, Hess DC, Wechsler LR, Furie KL, Gorelick PB, Hurn P, Liebeskind DS, Nogueira RG, Saver JL, STAIR VII Consortium (2011) Stroke Treatment Academic Industry Roundtable (STAIR) recommendations for maximizing the use of intravenous thrombolytics and expanding treatment options with intra-arterial and neuroprotective therapies. Stroke 42:2645–2650
Aronowski J, Samways E, Strong R, Rhoades H, Grotta J (1996) An alternative method for the quantitation of neuronal damage after experimental middle cerebral artery occlusion in rats: analysis of behavioral deficit. J Cereb Blood Flow Metab 16:705–713
Atif F, Yousuf S, Sayeed I, Ishrat T, Hua F, Stein DG (2013) Combination treatment with progesterone and vitamin D hormone is more effective than monotherapy in ischemic stroke: the role of BDNF/TrkB/Erk1/2 signaling in neuroprotection. Neuropharmacol 67:78–87
Baulieu EE, Robel P, Schumacher M (2001) Neurosteroids: beginning of the story. Int Rev Neurobiol 46:1–32
Borlongon CV, Hida H, Nishino H (1999) Early assessment of motor dysfunctions aids in successful occlusion of the middle cerebral artery. Neuroreport 9:3615–3621
Cai W, Zhu Y, Furuya K, Li Z, Sokabe M, Chen L (2008) Two different molecular mechanisms underlying progesterone neuroprotection against ischemic brain damage. Neuropharmacol 55:127–138
Cutler SM, Pettus EH, Hoffman SW, Stein DG (2005) Tapered progesterone withdrawal enhances behavioral and molecular recovery after traumatic brain injury. Exp Neurol 195(2):423–429
Cutler SM, VanLandingham JW, Murphy AZ, Stein DG (2006a) Slow-release and injected progesterone treatments enhance acute recovery after traumatic brain injury. Pharmacol Biochem Behav 84(3):420–428
Cutler SM, Vanlandingham JW, Stein DG (2006b) Tapered progesterone withdrawal promotes long-term recovery following brain trauma. Exp Neurol 200(2):378–385
De Nicola AF, Coronel F, Li G, Gargiulo-Monachelli G, McG Deniselle EY, Gonzalez SL, Labombarda F, Meyer M, Guennoun R, Schumacher M (2013) Therapeutic effects of progesterone in animal models of neurological disorders. CNS Neurol Disord Drug Targets. 2013 Sep 4
Deutsch ER, Espinoza TR, Atif F, Woodall E, Kaylor J, Wright DW (2013) Progesterone’s role in neuroprotection, a review of the evidence. Brain Res 1530:82–105
DeVries AC, Nelson RJ, Traystman RJ, Hurn PD (2001) Cognitive and behavioral assessment in experimental stroke research: will it prove useful? Neurosci Biobehav Rev 25:325–342
Djebaili M, Hoffman SW, Stein DG (2004) Allopregnanolone and progesterone decrease cell death and cognitive deficits after a contusion of the rat pre-frontal cortex. Neuroscience 123:349–359
Donnan GA, Fisher M, Macleod M, Davis SM (2008) Stroke. Lancet 371:1612–1623
Encarnacion A, Horie N, Keren-Gill H, Bliss TM, Steinberg GK, Shamloo M (2011) Long-term behavioral assessment of function in an experimental model for ischemic stroke. J Neurosci Methods 196:247–257
Esneault E, Castagne V, Moser P, Bonny C, Bernaudin M (2008) D-JNKi, a peptide inhibitor of c-Jun N-terminal kinase, promotes functional recovery after transient focal cerebral ischemia in rats. Neuroscience 152:308–320
Espinosa-Garcia C, Aguilar-Hernandez A, Cervantes M, Morali G (2014) Effects of progesterone on neurite growth inhibitors in the hippocampus following global cerebral ischemia. Brain Res 1545:23–34
Gibson CL, Murphy SP (2004) Progesterone enhances functional recovery after middle cerebral artery occlusion in male mice. J Cereb Blood Flow Metab 24:805–813
Gibson CL, Constantin D, Prior MJ, Bath PM, Murphy SP (2005) Progesterone suppresses the inflammatory response and nitric oxide synthase-2 expression following cerebral ischemia. Exp Neurol 193:522–530
Gibson CL, Coomber B, Murphy SP (2011) Progesterone is neuroprotective following cerebral ischaemia in reproductively ageing female mice. Brain 134(Pt 7):2125–2133
Goss CW, Hoffman SW, Stein DG (2003) Behavioral effects and anatomic correlates after brain injury: a progesterone dose–response study. Pharmacol Biochem Behav 76:231–242
Hua F, Wang J, Ishrat T, Wei W, Atif F, Sayeed I, Stein D (2011) Genomic profile of Toll-like receptor pathways in traumatically brain-injured mice: effect of exogenous progesterone. J Neuroinflammation 8:42
Ishrat T, Sayeed I, Atif F, Stein DG (2009) Effects of progesterone administration on infarct volume and functional deficits following permanent focal cerebral ischemia in rats. Brain Res 1257:94–101
Ishrat T, Sayeed I, Atif F, Hua F, Stein DG (2010) Progesterone and allopregnanolone attenuate blood–brain barrier dysfunction following permanent focal ischemia by regulating the expression of matrix metalloproteinases. Exp Neurol 226:183–190
Ishrat T, Sayeed I, Atif F, Hua F, Stein DG (2012) Progesterone is neuroprotective against ischemic brain injury through its effects on the phosphoinositide 3-kinase/protein kinase B signaling pathway. Neurosci 210:442–450
Jones TA, Schallert T (1992) Subcortical deterioration after cortical damage: effects of diazepam and relation to recovery of function. Behav Brain Res 51:1–13
Longa EZ, Weinstein PR, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20:84–91
Meffre D, Labombarda F, Delespierre B, Chastre A, De Nicola AF, Stein DG, Schumacher M, Guennoun R (2013) Distribution of membrane progesterone receptor alpha in the male mouse and rat brain and its regulation after traumatic brain injury. Neuroscience 231:111–124
Millard WB (2013) New guidelines on tPA in stroke: putting out fires with gasoline? Ann Emerg Med 62:A13–A18
Muntner P, DeSalvo KB, Wildman RP, Raggi P, He J, Whelton PK (2006) Trends in the prevalence, awareness, treatment, and control of cardiovascular disease risk factors among noninstitutionalized patients with a history of myocardial infarction and stroke. Am J Epidemiol 163:913–920
Peeling J, Corbett D, Del Bigio MR, Hudzik TJ, Campbell TM, Palmer GC (2001) Rat middle cerebral artery occlusion: correlations between histopathology, T2-weighted magnetic resonance imaging, and behavioral indices. J Stroke Cerebrovasc Dis 10:166–177
Petersen SL, Intlekofer KA, Moura-Conlon PJ, Brewer DN, Del Pino SJ, Lopez JA (2013) Non-classical progesterone signalling molecules in the nervous system. J Neuroendocrinol. doi:10.1111/jne.12060
Roger VL, Go AS, Lloyd-Jones DM, Benjamin EJ, Berry JD, Borden WB et al (2012) Heart disease and stroke statistics—2012 update: a report from the American Heart Association. Circulation 125:e2–e220
Savva GM, Stephan BC (2010) Epidemiological studies of the effect of stroke on incident dementia: a systematic review. Stroke 41:e41–e46
Scalha TB, Miyasaki E, Lima NM, Borges G (2011) Correlations between motor and sensory functions in upper limb chronic hemiparetics after stroke. Arq Neuropsiquiatr 69(4):624–629
Schumacher M, Robel P, Baulieu EE (1996) Development and regeneration of the nervous system: a role for neurosteroids. Dev Neurosci 18(1–2):6–21
Stein DG (2013) A clinical/translational perspective: can a developmental hormone play a role in the treatment of traumatic brain injury? Horm Behav 63:291–300
Strbian D, Sairanen T, Meretoja A, Pitkäniemi J, Putaala J, Salonen O, Helsinki Stroke Thrombolysis Registry Group et al (2011) Patient outcomes from symptomatic intracerebral hemorrhage after stroke thrombolysis. Neurology 77:341–348
Stroke Therapy Academic Industry Roundtable (1999) Recommendations for standards regarding preclinical neuroprotective and restorative drug development. Stroke 30:2752–2578
The NINDS t-PA Stroke Study Group (1997) Intracerebral hemorrhage after intravenous t-PA therapy for ischemic stroke. Stroke 28:2109–2118
Turner RC, Lucke-Wold B, Lucke-Wold N, Elliott AS, Logsdon AF, Rosen CL, Huber JD (2013) Neuroprotection for ischemic stroke: moving past shortcomings and identifying promising directions. Int J Mol Sci 14(1):1890–1917. doi:10.3390/ijms14011890
Wahl F, Allix M, Plotkin M, Boulu RG (1992) Neurological and behavioural outcomes of focal cerebral ischemia in rats. Stroke 23:267–272
Wallace TL, Gudelsky GA, Vorhees CV (1999) Methamphetamine-induced neurotoxicity alters locomoter activity, stereotypic behavior, and stimulated dopamine release in the rat. J Neurosci 19:9141–9148
Wang Y, Bontempi B, Hong SM, Mehta K, Weinstein PR, Abrams GM, Liu J (2008) A comprehensive analysis of gait impairment after experimental stroke and the therapeutic effect of environmental enrichment in rats. J Cereb Blood Flow Metab 28:1936–1950
Ward NS, Cohen LG (2004) Mechanisms underlying recovery of motor function after stroke. Arch Neurol 61:1844–1848
Wong R, Renton C, Gibson CL, Murphy SJ, Kendall DA, Bath PM (2013) Progesterone treatment for experimental stroke: an individual animal meta-analysis. J Cereb Blood Flow Metab 33(9):1362–1372
Wright DW, Kellermann AL, Hertzberg VS, Clark PL, Frankel M, Goldstein FC et al (2007) ProTECT: a randomized clinical trial of progesterone for acute traumatic brain injury. Ann Emerg Med 49(4):391–402
Xiao G, Wei J, Yan W, Wang W, Lu Z (2008) Improved outcomes from the administration of progesterone for patients with acute severe traumatic brain injury: a randomized controlled trial. Crit Care 12(2):R61
Yousuf S, Atif F, Sayeed I, Wang J, Stein DG (2013) Post-stroke infections exacerbate ischemic brain injury in middle-aged rats: immunomodulation and neuroprotection by progesterone. Neuroscience 239:92–102
Acknowledgements
This research was supported by a NIH award U01 NS062676, BHR Pharma, and Allen and Company. The authors would like to thank Leslie McCann for her invaluable editorial assistance. SY is thankful to Jun Wang for his assistance in brain histology.
Conflict of interest
DGS is entitled to royalties from products of BHR Pharma LLC (BHR) related to the use of PROG in TBI and stroke, and may receive research funding from BHR, which is developing products related to this research. In addition, he serves as a consultant to BHR and receives compensation for these services. The terms of this arrangement have been reviewed and approved by Emory University in accordance with its conflict of interest policies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yousuf, S., Atif, F., Sayeed, I. et al. Progesterone in transient ischemic stroke: a dose–response study. Psychopharmacology 231, 3313–3323 (2014). https://doi.org/10.1007/s00213-014-3556-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-014-3556-8