Abstract
Rationale
Reexposure to ethanol during acute withdrawal might facilitate the transition to alcoholism by enhancing the rewarding effect of ethanol.
Objective
The conditioned place preference (CPP) procedure was used to test whether ethanol reward is enhanced during acute withdrawal.
Methods
DBA/2J mice were exposed to an unbiased one-compartment CPP procedure. Ethanol (0.75, 1.0, or 1.5 g/kg IP) was paired with a distinctive floor cue (CS+), whereas saline was paired with a different floor cue (CS−). The withdrawal (W) group received CS+ trials during acute withdrawal produced by a large dose of ethanol (4 g/kg) given 8 h before each trial. The no-withdrawal (NW) group did not experience acute withdrawal during conditioning trials but was matched for acute withdrawal experience. Floor preference was tested in the absence of ethanol or acute withdrawal.
Results
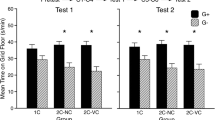
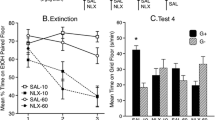
All groups eventually showed a dose-dependent preference for the ethanol-paired cue, but development of CPP was generally more rapid and stable in the W groups than in the NW groups. Acute withdrawal suppressed the normal activating effect of ethanol during CS+ trials, but there were no group differences in test activity.
Conclusions
Acute withdrawal enhanced ethanol’s rewarding effect as indexed by CPP. Since this effect depended on ethanol exposure during acute withdrawal, the enhancement of ethanol reward was likely mediated by the alleviation of acute withdrawal, i.e., negative reinforcement. Enhancement of ethanol reward during acute withdrawal may be a key component in the shift from episodic to chronic ethanol consumption that characterizes alcoholism.



Similar content being viewed by others
References
American Psychiatric Association (2000) Diagnostic and statistical manual of mental disorders, 4th edn. American Psychiatric Association, Washington, Text revision
Becker HC (2008) Alcohol dependence, withdrawal, and relapse. Alcohol Res Health 31:348–361, Journal of the National Institute on Alcohol Abuse and Alcoholism
Becker HC, Lopez MF (2004) Increased ethanol drinking after repeated chronic ethanol exposure and withdrawal experience in C57BL/6 mice. Alcohol Clin Exp Res 28:1829–1838
Boyce-Rustay JM, Cunningham CL (2004) The role of NMDA receptor binding sites in ethanol place conditioning. Behav Neurosci 118:822–834
Brown G, Jackson A, Stephens DN (1998) Effects of repeated withdrawal from chronic ethanol on oral self-administration of ethanol on a progressive ratio schedule. Behav Pharmacol 9:149–161
Buck KJ, Metten P, Belknap JK, Crabbe JC (1997) Quantitative trait loci involved in genetic predisposition to acute ethanol withdrawal in mice. J Neurosci 17:3946–3955
Chester J, Cunningham C (1999) Baclofen alters ethanol-stimulated activity but not conditioned place preference or taste aversion in mice. Pharmacol Biochem Behav 63:325–331
Crabbe JC (1998) Provisional mapping of quantitative trait loci for chronic ethanol withdrawal severity in BXD recombinant inbred mice. J Pharmacol Exp Ther 286:263–271
Crabbe JC, Merrill C, Belknap JK (1991) Acute dependence on depressant drugs is determined by common genes in mice. J Pharmacol Exp Ther 257:663–667
Cunningham CL, Tull LE, Rindal KE, Meyer PJ (2002) Distal and proximal pre-exposure to ethanol in the place conditioning task: tolerance to aversive effect, sensitization to activating effect, but no change in rewarding effect. Psychopharmacol (Berl) 160:414–424
Cunningham CL, Ferree NK, Howard MA (2003) Apparatus bias and place conditioning with ethanol in mice. Psychopharmacol (Berl) 170:409–422
Cunningham CL, Gremel CM, Groblewski PA (2006) Drug-induced conditioned place preference and aversion in mice. Nat Protoc 1:1662–1670
Cunningham CL, Groblewski PA, Voorhees CM (2011) Place conditioning. In: Olmstead MC (ed) Animal Models of Drug Addiction, Neuromethods, vol 53. Humana, Totowa, pp 167–189
Cunningham CL, Fidler TL, Murphy KV, Mulgrew JA, Smitasin PJ (2013) Time-dependent negative reinforcement of ethanol intake by alleviation of acute withdrawal. Biol Psychiatry 73:249–255
Deutsch JA, Cannis JT (1980) Rapid induction of voluntary alcohol choice in rats. Behav Neural Biol 30:292–298
Fidler TL, Clews TW, Cunningham CL (2006) Reestablishing an intragastric ethanol self-infusion model in rats. Alcohol Clin Exp Res 30:414–428
Fidler TL, Oberlin BG, Struthers AM, Cunningham CL (2009) Schedule of passive ethanol exposure affects subsequent intragastric ethanol self-infusion. Alcohol Clin Exp Res 33:1909–1923
Fidler TL, Dion AM, Powers MS, Ramirez JJ, Mulgrew JA, Smitasin PJ, Crane AT, Cunningham CL (2011) Intragastric self-infusion of ethanol in high- and low-drinking mouse genotypes after passive ethanol exposure. Genes Brain Behav 10:264–275
Fidler TL, Powers MS, Ramirez JJ, Crane A, Mulgrew J, Smitasin P, Cunningham CL (2012) Dependence induced increases in intragastric alcohol consumption in mice. Addict Biol 17:13–32
Finn DA, Snelling C, Fretwell AM, Tanchuck MA, Underwood L, Cole M, Crabbe JC, Roberts AJ (2007) Increased drinking during withdrawal from intermittent ethanol exposure is blocked by the CRF receptor antagonist D-Phe-CRF(12–41). Alcohol Clin Exp Res 31:939–949
Gorin-Meyer RE, Wiren KM, Tanchuck MA, Long SL, Yoneyama N, Finn DA (2007) Sex differences in the effect of finasteride on acute ethanol withdrawal severity in C57BL/6J and DBA/2J mice. Neuroscience 146:1302–1315
Groblewski PA, Bax LS, Cunningham CL (2008) Reference-dose place conditioning with ethanol in mice: empirical and theoretical analysis. Psychopharmacol (Berl) 201:97–106
Heilig M, Egli M, Crabbe JC, Becker HC (2010) Acute withdrawal, protracted abstinence and negative affect in alcoholism: are they linked? Addict Biol 15:169–184
Koob GF (2011) Animal models of drug dependence: motivational perspective. In: Johnson BA (ed) Addiction medicine: science and practice. Springer, New York, pp 333–357
Linakis JG, Cunningham CL (1979) Effects of concentration of ethanol injected intraperitoneally on taste aversion, body temperature, and activity. Psychopharmacology 64:61–65
Lopez MF, Becker HC (2005) Effect of pattern and number of chronic ethanol exposures on subsequent voluntary ethanol intake in C57BL/6J mice. Psychopharmacol (Berl) 181:688–696
McCaul ME, Turkkan JS, Svikis DS, Bigelow GE (1991) Alcohol and secobarbital effects as a function of familial alcoholism: extended intoxication and increased withdrawal effects. Alcohol Clin Exp Res 15:94–101
Metten P, Crabbe JC (1994) Common genetic determinants of severity of acute withdrawal from ethanol, pentobarbital and diazepam in inbred mice. Behav Pharmacol 5:533–547
O’Dell LE, Roberts AJ, Smith RT, Koob GF (2004) Enhanced alcohol self-administration after intermittent versus continuous alcohol vapor exposure. Alcohol Clin Exp Res 28:1676–1682
Rimondini R, Arlinde C, Sommer W, Heilig M (2002) Long-lasting increase in voluntary ethanol consumption and transcriptional regulation in the rat brain after intermittent exposure to alcohol. Faseb J 16:27–35
Ripley TL, Brown G, Dunworth SJ, Stephens DN (2003a) Aversive conditioning following repeated withdrawal from ethanol and epileptic kindling. Eur J Neurosci 17:1664–1670
Ripley TL, O’Shea M, Stephens DN (2003b) Repeated withdrawal from ethanol impairs acquisition but not expression of conditioned fear. Eur J Neurosci 18:441–448
Ripley TL, Borlikova G, Lyons S, Stephens DN (2004) Selective deficits in appetitive conditioning as a consequence of ethanol withdrawal. Eur J Neurosci 19:415–425
Risinger FO, Dickinson SD, Cunningham CL (1992) Haloperidol reduces ethanol-induced motor activity stimulation but not conditioned place preference. Psychopharmacology 107:453–456
Roberts A, Cole M, Koob G (1996) Intra-amygdala muscimol decreases operant ethanol self-administration in dependent rats. Alcohol Clin Exp Res 20:1289–1298
Roberts AJ, Heyser CJ, Cole M, Griffin P, Koob GF (2000) Excessive ethanol drinking following a history of dependence: animal model of allostasis. Neuropsychopharmacology 22:581–594
Schulteis G, Hyytia P, Heinrichs SC, Koob GF (1996) Effects of chronic ethanol exposure on oral self-administration of ethanol or saccharin by Wistar rats. Alcohol Clin Exp Res 20:164–171
Shimamoto A, Liu JY, Fujimiya T (2010) SIAM-like phenomenon caused by low doses of alcohol. Alcohol Clin Exp Res 34(Suppl 1):S2–S6
Thurman RG, Paschal D, Abu-Murad C, Pekkanen L, Bradford BU, Bullock K, Glassman E (1982) Swift increase in alcohol metabolism (SIAM) in the mouse: comparison of the effect of short-term ethanol treatment on ethanol elimination in four inbred strains. J Pharmacol Exp Ther 223:45–49
Ting-A-Kee R, Dockstader C, Heinmiller A, Grieder T, van der Kooy D (2009) GABA(A) receptors mediate the opposing roles of dopamine and the tegmental pedunculopontine nucleus in the motivational effects of ethanol. Eur J Neurosci 29:1235–1244
Ting-A-Kee R, Vargas-Perez H, Bufalino MR, Bahi A, Dreyer JL, Tyndale RF, van der Kooy D (2012) Infusion of brain-derived neurotrophic factor into the ventral tegmental area switches the substrates mediating ethanol motivation. Eur J Neurosci 37:996–1003
Windle M, Zucker RA (2010) Reducing underage and young adult drinking: how to address critical drinking problems during this developmental period. Alcohol Res Health 33:29–44
Acknowledgments
Research reported in this paper was supported by the National Institute on Alcohol Abuse and Alcoholism of the National Institutes of Health under award numbers R01AA007702, R01AA020866, and T32AA007468. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Thanks are extended to Nancy Bormann for her preliminary work on this topic and her help in developing an earlier version of the experimental design and procedure used here. We also thank Melanie Pina for her comments on an earlier draft of the paper. Experiments reported here comply with the current laws of the USA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dreumont, S.E., Cunningham, C.L. Effects of acute withdrawal on ethanol-induced conditioned place preference in DBA/2J mice. Psychopharmacology 231, 777–785 (2014). https://doi.org/10.1007/s00213-013-3291-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-013-3291-6