Abstract
Rationale
Atomoxetine is a potent and selective norepinephrine transporter (NET) reuptake inhibitor acting as a nonstimulant for the treatment of attention-deficit/hyperactivity disorder (ADHD). Previous positron emission tomography (PET) studies had failed to demonstrate the feasibility of measuring a dose-dependent and saturable NET occupancy in human brain using [11C]MeNER.
Objectives
To determine if atomoxetine occupies NET in a dose-dependent fashion using (S,S)-[18F]FMeNER-D2 in nonhuman primate brain.
Methods
A total of eight PET measurements were performed in two cynomolgus monkeys. Each monkey was examined four times with PET: under baseline conditions and after steady-state infusion with 0.03, 0.06, or 0.12 mg/kg/h of atomoxetine. A prolonged intravenous (i.v.) infusion design was developed rather than an i.v. bolus to better mimic an oral absorption profile and to reach plasma steady state.
Results
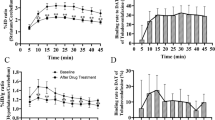
During baseline conditions, (S,S)-[18F]FMeNER-D2 uptake was highest in the locus coeruleus, thalamus, mesencephalon, and the cingulate gyrus, whereas the radioactivity in the caudate was low. Peak equilibrium measurements were achieved using (S,S)-[18F]FMeNER-D2 in contrast to the previously reported data for [11C]MeNER. After administration of atomoxetine, a dose-dependent occupancy from 38 to 82% was observed for various brain regions known to contain high densities of NET.
Conclusions
This is the first in vivo PET study to successfully demonstrate the ability to measure a dose-dependent change in NET occupancy in brain using (S,S)-[18F]FMeNER-D2. Furthermore, an asymptotic relationship between N-desmethylatomoxetine plasma concentration and NET occupancy was established. In total, these data encourage further PET studies using (S,S)-[18F]FMeNER-D2 in humans.





Similar content being viewed by others
References
Andree B, Seneca N, Schou M, Mozley PD, Potter WZ, Farde L, Gulyas B, Halldin C (2004) Regional central norepinephrine transporter occupancy induced by reboxetine determined in man using [11C]MeNER. Neuroimage 22(Suppl 2):30
Aston-Jones G, Shipley MT, Chouvet G, Ennis M, van Bockstaele E, Pieribone V, Shiekhattar R, Akaoka H, Drolet G, Astier B et al (1991) Afferent regulation of locus coeruleus neurons: anatomy, physiology and pharmacology. Prog Brain Res 88:47–75
Bobb AJ, Addington AM, Sidransky E, Gornick MC, Lerch JP, Greenstein DK, Clasen LS, Sharp WS, Inoff-Germain G, Wavrant-De Vrieze F, Arcos-Burgos M, Straub RE, Hardy JA, Castellanos FX, Rapoport JL (2005) Support for association between ADHD and two candidate genes: NET1 and DRD1. Am J Med Genet B Neuropsychiatr Genet 134:67–72
Bymaster FP, Katner JS, Nelson DL, Hemrick-Luecke SK, Threlkeld PG, Heiligenstein JH, Morin SM, Gehlert DR, Perry KW (2002) Atomoxetine increases extracellular levels of norepinephrine and dopamine in prefrontal cortex of rat: a potential mechanism for efficacy in attention deficit/hyperactivity disorder. Neuropsychopharmacology 27:699–711
Carson RE, Wu Y, Lang L, Ma Y, Der MG, Herscovitch P, Eckelman WC (2003) Brain uptake of the acid metabolites of F-18-labeled WAY 100635 analogs. J Cereb Blood Flow Metab 23:249–260
Charnay Y, Leger L, Vallet PG, Hof PR, Jouvet M, Bouras C (1995) [3H]Nisoxetine binding sites in the cat brain: an autoradiographic study. Neuroscience 69:259–270
Ding YS, Lin KS, Logan J, Benveniste H, Carter P (2005) Comparative evaluation of positron emission tomography radiotracers for imaging the norepinephrine transporter: (S,S) and (R,R) enantiomers of reboxetine analogs ([11C]methylreboxetine, 3-Cl-[11C]methylreboxetine and [18F]fluororeboxetine), (R)-[11C]nisoxetine, [11C]oxaprotiline and [11C]lortalamine. J Neurochem 94:337–351
Donnan GA, Kaczmarczyk SJ, Paxinos G, Chilco PJ, Kalnins RM, Woodhouse DG, Mendelsohn FA (1991) Distribution of catecholamine uptake sites in human brain as determined by quantitative [3H] mazindol autoradiography. J Comp Neurol 304:419–434
Gehlert DR, Gackenheimer SL, Robertson DW (1993) Localization of rat brain binding sites for [3H]tomoxetine, an enantiomerically pure ligand for norepinephrine reuptake sites. Neurosci Lett 157:203–206
Ghose S, Fujita M, Morrison P, Uhl G, Murphy DL, Mozley PD, Schou M, Halldin C, Innis R (2005) Specific in vitro binding of (S,S)-[3H]MeNER to norepinephrine transporters. Synapse 56:100–104
Gross-Isseroff R, Israeli M, Biegon A (1988) Autoradiographic analysis of [3H]desmethylimipramine binding in the human brain postmortem. Brain Res 456:120–126
Haller J, Bakos N, Rodriguiz RM, Caron MG, Wetsel WC, Liposits Z (2002) Behavioral responses to social stress in noradrenaline transporter knockout mice: effects on social behavior and depression. Brain Res Bull 58:279–284
Haughey HM, Kaiser AL, Johnson TE, Bennett B, Sikela JM, Zahniser NR (2005) Norepinephrine transporter: a candidate gene for initial ethanol sensitivity in inbred long-sleep and short-sleep mice. Alcohol Clin Exp Res 29:1759–1768
Hargreaves R (2002) Imaging substance P receptors (NK1) in the living human brain using positron emission tomography. J Clin Psychiatry 63(Suppl 11):18–24
Ichise M, Liow JS, Lu JQ, Takano A, Model K, Toyama H, Suhara T, Suzuki K, Innis RB, Carson RE (2003) Linearized reference tissue parametric imaging methods: application to [11C]DASB positron emission tomography studies of the serotonin transporter in human brain. J Cereb Blood Flow Metab 23:1096–1112
Iversen L (2006) Neurotransmitter transporters and their impact on the development of psychopharmacology. Br J Pharmacol 147(Suppl 1):S82–S88
Karlsson P, Farde L, Halldin C, Swahn CG, Sedvall G, Foged C, Hansen KT, Skrumsager B (1993) PET examination of [11C]NNC 687 and [11C]NNC 756 as new radioligands for the D1-dopamine receptor. Psychopharmacology (Berl) 113:149–156
Klimek V, Stockmeier C, Overholser J, Meltzer HY, Kalka S, Dilley G, Ordway GA (1997) Reduced levels of norepinephrine transporters in the locus coeruleus in major depression. J Neurosci 17:8451–8458
Lammertsma AA, Hume SP (1996) Simplified reference tissue model for PET receptor studies. Neuroimage 4:153–158
Li W, Knowlton D, Van Winkle DM, Habecker BA (2004) Infarction alters both the distribution and noradrenergic properties of cardiac sympathetic neurons. Am J Physiol Heart Circ Physiol 286:H2229–H2236
Logan J, Fowler JS, Volkow ND, Wang GJ, Ding YS, Alexoff DL (1996) Distribution volume ratios without blood sampling from graphical analysis of PET data. J Cereb Blood Flow Metab 16:834–840
Michelson D, Faries D, Wernicke J, Kelsey D, Kendrick K, Sallee FR, Spencer T (2001) Atomoxetine in the treatment of children and adolescents with attention-deficit/hyperactivity disorder: a randomized, placebo-controlled, dose-response study. Pediatrics 108:E83
Roland PE, Zilles K (1994) Brain atlases-a new research tool. Trends Neurosci 17:458–467
Schou M, Halldin C, Sovago J, Pike VW, Gulyas B, Mozley PD, Johnson DP, Hall H, Innis RB, Farde L (2003) Specific in vivo binding to the norepinephrine transporter demonstrated with the PET radioligand, (S,S)-[11C]MeNER. Nucl Med Biol 30:707–714
Schou M, Halldin C, Sovago J, Pike VW, Hall H, Gulyas B, Mozley PD, Dobson D, Shchukin E, Innis RB, Farde L (2004) PET evaluation of novel radiofluorinated reboxetine analogs as norepinephrine transporter probes in the monkey brain. Synapse 53:57–67
Schou M, Halldin C, Pike VW, Mozley PD, Dobson D, Innis RB, Farde L, Hall H (2005) Post-mortem human brain autoradiography of the norepinephrine transporter using (S,S)-[18F]FMeNER-D2. Eur Neuropsychopharmacol 15:517–520
Seneca N, Andree B, Sjoholm N, Schou M, Pauli S, Mozley PD, Stubbs JB, Liow JS, Sovago J, Gulyas B, Innis R, Halldin C (2005) Whole-body biodistribution, radiation dosimetry estimates for the PET norepinephrine transporter probe (S,S)-[18F]FMeNER-D2 in non-human primates. Nucl Med Commun 26:695–700
Spencer T, Biederman J, Wilens T, Prince J, Hatch M, Jones J, Harding M, Faraone SV, Seidman L (1998) Effectiveness and tolerability of tomoxetine in adults with attention deficit hyperactivity disorder. Am J Psychiatry 155:693–695
Stahl SM (2003) Neurotransmission of cognition, part 2. Selective NRIs are smart drugs: exploiting regionally selective actions on both dopamine and norepinephrine to enhance cognition. J Clin Psychiatry 64:110–111
Tejani-Butt SM (1992) [3H]nisoxetine: a radioligand for quantitation of norepinephrine uptake sites by autoradiography or by homogenate binding. J Pharmacol Exp Ther 260:427–436
Tejani-Butt SM, Yang J, Zaffar H (1993) Norepinephrine transporter sites are decreased in the locus coeruleus in Alzheimer’s disease. Brain Res 631:147–150
Tipre D, Zoghbi S, Liow JS, Green M, Seidel J, Ichise M, Innis RB, Pike VW (2006) PET imaging of brain 5-HT1A receptors in rat in vivo with [18F]FCWAY and improvement by successful inhibition of radioligand defluorination with miconazole. J Nucl Med 47:1–9
Xu F, Gainetdinov RR, Wetsel WC, Jones SR, Bohn LM, Miller GW, Wang YM, Caron MG (2000) Mice lacking the norepinephrine transporter are supersensitive to psychostimulants. Nat Neurosci 3:465–471
Wienhard K, Dahlbom M, Eriksson L, Michel C, Bruckbauer T, Pietrzyk U, Heiss WD (1994) The ECAT EXACT HR: performance of a new high resolution positron scanner. J Comput Assist Tomogr 18:110–118
Wilson AA, Johnson DP, Mozley D, Hussey D, Ginovart N, Nobrega J, Garcia A, Meyer J, Houle S (2003) Synthesis and in vivo evaluation of novel radiotracers for the in vivo imaging of the norepinephrine transporter. Nucl Med Biol 30:85–92
Wong DT, Threlkeld PG, Best KL, Bymaster FP (1982) A new inhibitor of norepinephrine uptake devoid of affinity for receptors in rat brain. J Pharmacol Exp Ther 222:61–65
Wong D, Kuwabara H, Mozley PD, Dannals R, Kumar A, Ye W, Brasic J, Alexander M, Mathews W, Holt D, Vandenhende F, Gjedde A (2005) Characterization of dose dependent norepinephrine transporter blockade by atomoxetine in human brain using [11C]MeNER PET. J Cereb Blood Flow Metab 25:S599
Acknowledgements
This study was sponsored by the Eli Lilly and Co., Indianapolis, IN, USA. The authors would like to thank Eli Lilly Research Laboratories for providing the precursor, standard, and atomoxetine. We are appreciative of G. Douglas Ponsler from Eli Lilly Research Laboratories and Heather Coales from SFBC Taylor for their bioanalytical support. We are also grateful to the members of the Karolinska PET psychiatry group for their assistance in the PET experiments. Andrea Varrone was supported by a grant from the “Short-Term Mobility Program” of the National Research Council of Italy. Nicholas Seneca and Magnus Schou are graduate students in the NIH–Karolinska Institutet joint Ph.D. program in neuroscience, and this research was supported in part by the Intramural Research Program of the NIH, National Institute of Mental Health, Bethesda, Maryland, USA.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Seneca, N., Gulyás, B., Varrone, A. et al. Atomoxetine occupies the norepinephrine transporter in a dose-dependent fashion: a PET study in nonhuman primate brain using (S,S)-[18F]FMeNER-D2 . Psychopharmacology 188, 119–127 (2006). https://doi.org/10.1007/s00213-006-0483-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-006-0483-3