Abstract
Rationale
Nicotine administration potentiates conditioned reinforcement in rats, an effect that persists for weeks after chronic exposure. Little is known regarding the nicotinic receptor subtypes that may mediate this effect.
Objective
The purpose of this study was to determine whether β2-subunit-containing nicotinic acetylcholine receptors (β2*nAChRs) are necessary for lasting effects of nicotine on conditioned and primary reinforcement in mice.
Methods
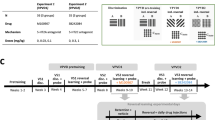
β2 knockout (β2KO) and wild-type (WT) mice received 14 days of nicotine exposure (NIC, 200 μg/ml in 2% saccharin) or saccharin alone (SAC) in their drinking water. Five days later, mice received paired presentations of a conditioned stimulus (CS) with water unconditioned stimulus (US) or explicitly unpaired presentations of the CS and US during Pavlovian discriminative approach training. Training was followed by two conditioned reinforcement tests. Mice were subsequently tested for food-reinforced responding in the absence of explicit cues followed by a progressive ratio test.
Results
During conditioned reinforcement testing, only mice in the paired condition showed increased responding in the CS-reinforced aperture over inactive apertures. WT-NIC mice showed enhanced conditioned reinforcement compared to WT-SAC animals. β2KO-SAC mice showed elevated conditioned reinforcement compared to WT-SAC subjects, but β2KO-NIC and β2KO-SAC mice did not differ in responding with conditioned reinforcement. Prior nicotine exposure did not alter food-reinforced responding but resulted in elevated break points for food in both genotypes.
Conclusion
These data show that nicotine exposure enhances conditioned reinforcement in mice and indicate that β2*nAChRs are necessary for nicotine-dependent enhancement of incentive aspects of motivation but not motivation for primary reinforcement measured by progressive ratio responding.





Similar content being viewed by others
References
Arthur D, Levin ED (2002) Chronic inhibition of alpha4beta2 nicotinic receptors in the ventral hippocampus of rats: impacts on memory and nicotine response. Psychopharmacology (Berl) 160:140–145
Bancroft A, Levin ED (2000) Ventral hippocampal alpha4beta2 nicotinic receptors and chronic nicotine effects on memory. Neuropharmacology 39:2770–2778
Benwell ME, Balfour DJ (1992) The effects of acute and repeated nicotine treatment on nucleus accumbens dopamine and locomotor activity. Br J Pharmacol 105:849–856
Benwell ME, Balfour DJ, Anderson JM (1988) Evidence that tobacco smoking increases the density of (−)-[3H]nicotine binding sites in human brain. J Neurochem 50:1243–1247
Benwell ME, Balfour DJ, Khadra LF (1994) Studies on the influence of nicotine infusions on mesolimbic dopamine and locomotor responses to nicotine. Clin Investig 72:233–239
Brody AL, Mandelkern MA, London ED, Childress AR, Lee GS, Bota RG, Ho ML, Saxena S, Baxter LR Jr, Madsen D, Jarvik ME (2002) Brain metabolic changes during cigarette craving. Arch Gen Psychiatry 59:1162–1172
Brunzell DH, Russell DS, Picciotto MR (2003) In vivo nicotine treatment regulates mesocorticolimbic CREB and ERK signaling in C57Bl/6J mice. J Neurochem 84:1431–1441
Buisson B, Bertrand D (2001) Chronic exposure to nicotine upregulates the human (alpha)4((beta)2 nicotinic acetylcholine receptor function. J Neurosci 21:1819–1829
Burns LH, Robbins TW, Everitt BJ (1993) Differential effects of excitotoxic lesions of the basolateral amygdala, ventral subiculum and medial prefrontal cortex on responding with conditioned reinforcement and locomotor activity potentiated by intra-accumbens infusions of d-amphetamine. Behav Brain Res 55:167–183
Cadoni C, Di Chiara G (2000) Differential changes in accumbens shell and core dopamine in behavioral sensitization to nicotine. Eur J Pharmacol 387:R23–R25
Cador M, Taylor JR, Robbins TW (1991) Potentiation of the effects of reward-related stimuli by dopaminergic-dependent mechanisms in the nucleus accumbens. Psychopharmacology (Berl) 104:377–385
Caggiula AR, Donny EC, White AR, Chaudhri N, Booth S, Gharib MA, Hoffman A, Perkins KA, Sved AF (2001) Cue dependency of nicotine self-administration and smoking. Pharmacol Biochem Behav 70:515–530
Caggiula AR, Donny EC, Chaudhri N, Perkins KA, Evans-Martin FF, Sved AF (2002a) Importance of nonpharmacological factors in nicotine self-administration. Physiol Behav 77:683–687
Caggiula AR, Donny EC, White AR, Chaudhri N, Booth S, Gharib MA, Hoffman A, Perkins KA, Sved AF (2002b) Environmental stimuli promote the acquisition of nicotine self-administration in rats. Psychopharmacology (Berl) 163:230–237
Chiamulera C (2005) Cue reactivity in nicotine and tobacco dependence: a “multiple-action” model of nicotine as a primary reinforcement and as an enhancer of the effects of smoking-associated stimuli. Brain Res Brain Res Rev 48:74–97
Cohen C, Perrault G, Griebel G, Soubrie P (2005) Nicotine-associated cues maintain nicotine-seeking behavior in rats several weeks after nicotine withdrawal: reversal by the cannabinoid (CB1) receptor antagonist, rimonabant (SR141716). Neuropsychopharmacology 30:145–155
Collins AC, Romm E, Wehner JM (1990) Dissociation of the apparent relationship between nicotine tolerance and up-regulation of nicotinic receptors. Brain Res Bull 25:373–379
Cunningham ST, Kelley AE (1992) Opiate infusion into nucleus accumbens: contrasting effects on motor activity and responding for conditioned reward. Brain Res 588:104–114
Di Ciano P, Everitt BJ (2004) Contribution of the ventral tegmental area to cocaine-seeking maintained by a drug-paired conditioned stimulus in rats. Eur J Neurosci 19:1661–1667
Donny EC, Chaudhri N, Caggiula AR, Evans-Martin FF, Booth S, Gharib MA, Clements LA, Sved AF (2003) Operant responding for a visual reinforcer in rats is enhanced by noncontingent nicotine: implications for nicotine self-administration and reinforcement. Psychopharmacology (Berl) 169:68–76
Due DL, Huettel SA, Hall WG, Rubin DC (2002) Activation in mesolimbic and visuospatial neural circuits elicited by smoking cues: evidence from functional magnetic resonance imaging. Am J Psychiatry 159:954–960
Epping-Jordan MP, Watkins SS, Koob GF, Markou A (1998) Dramatic decreases in brain reward function during nicotine withdrawal. Nature 393:76–79
Epping-Jordan MP, Picciotto MR, Changeux JP, Pich EM (1999) Assessment of nicotinic acetylcholine receptor subunit contributions to nicotine self-administration in mutant mice. Psychopharmacology (Berl) 147:25–26
Fuchs RA, Evans KA, Parker MC, See RE (2004) Differential involvement of the core and shell subregions of the nucleus accumbens in conditioned cue-induced reinstatement of cocaine seeking in rats. Psychopharmacology (Berl) 176:459–465
Grabus SD, Martin BR, Batman AM, Tyndale RF, Sellers E, Damaj MI (2005) Nicotine physical dependence and tolerance in the mouse following chronic oral administration. Psychopharmacology (Berl) 178:183–192
Grady SR, Meinerz NM, Cao J, Reynolds AM, Picciotto MR, Changeux JP, McIntosh JM, Marks MJ, Collins AC (2001) Nicotinic agonists stimulate acetylcholine release from mouse interpeduncular nucleus: a function mediated by a different nAChR than dopamine release from striatum. J Neurochem 76:258–268
Harmer CJ, Phillips GD (1998) Enhanced appetitive conditioning following repeated pretreatment with d-amphetamine. Behav Pharmacol 9:299–308
Hatfield T, Han JS, Conley M, Gallagher M, Holland P (1996) Neurotoxic lesions of basolateral, but not central, amygdala interfere with Pavlovian second-order conditioning and reinforcer devaluation effects. J Neurosci 16:5256–5265
Hitchcott PK, Harmer CJ, Phillips GD (1997) Enhanced acquisition of discriminative approach following intra-amygdala d-amphetamine. Psychopharmacology (Berl) 132:237–246
Ito R, Robbins TW, Everitt BJ (2004) Differential control over cocaine-seeking behavior by nucleus accumbens core and shell. Nat Neurosci 7:389–397
Jentsch JD, Taylor JR (1999) Impulsivity resulting from frontostriatal dysfunction in drug abuse: implications for the control of behavior by reward-related stimuli. Psychopharmacology (Berl) 146:373–390
King SL, Marks MJ, Grady SR, Caldarone BJ, Koren AO, Mukhin AG, Collins AC, Picciotto MR (2003) Conditional expression in corticothalamic efferents reveals a developmental role for nicotinic acetylcholine receptors in modulation of passive avoidance behavior. J Neurosci 23:3837–3843
King SL, Caldarone BJ, Picciotto MR (2004) Beta2-subunit-containing nicotinic acetylcholine receptors are critical for dopamine-dependent locomotor activation following repeated nicotine administration. Neuropharmacology 47(Suppl 1):132–139
Lesage MG, Burroughs D, Dufek M, Keyler DE, Pentel PR (2004) Reinstatement of nicotine self-administration in rats by presentation of nicotine-paired stimuli, but not nicotine priming. Pharmacol Biochem Behav 79:507–513
Lindgren JL, Gallagher M, Holland PC (2003) Lesions of basolateral amygdala impair extinction of CS motivational value, but not of explicit conditioned responses, in Pavlovian appetitive second-order conditioning. Eur J Neurosci 17:160–166
Mackintosh N (1974) The psychology of animal learning. Academic, New York
Marubio LM, Gardier AM, Durier S, David D, Klink R, Arroyo-Jimenez MM, McIntosh JM, Rossi F, Champtiaux N, Zoli M, Changeux JP (2003) Effects of nicotine in the dopaminergic system of mice lacking the alpha4 subunit of neuronal nicotinic acetylcholine receptors. Eur J Neurosci 17:1329–1337
Niaura R, Abrams D, Demuth B, Pinto R, Monti P (1989) Responses to smoking-related stimuli and early relapse to smoking. Addict Behav 14:419–428
Nooney JM, Peters JA, Lambert JJ (1992) A patch clamp study of the nicotinic acetylcholine receptor of bovine adrenomedullary chromaffin cells in culture. J Physiol 455:503–527
Olausson P, Jentsch JD, Taylor JR (2003) Repeated nicotine exposure enhances reward-related learning in the rat. Neuropsychopharmacology 28:1264–1271
Olausson P, Jentsch JD, Taylor JR (2004a) Nicotine enhances responding with conditioned reinforcement. Psychopharmacology (Berl) 171:173–178
Olausson P, Jentsch JD, Taylor JR (2004b) Repeated nicotine exposure enhances responding with conditioned reinforcement. Psychopharmacology (Berl) 173:98–104
Parkinson JA, Olmstead MC, Burns LH, Robbins TW, Everitt BJ (1999) Dissociation in effects of lesions of the nucleus accumbens core and shell on appetitive pavlovian approach behavior and the potentiation of conditioned reinforcement and locomotor activity by d-amphetamine. J Neurosci 19:2401–2411
Perkins KA, Gerlach D, Vender J, Grobe J, Meeker J, Hutchison S (2001) Sex differences in the subjective and reinforcing effects of visual and olfactory cigarette smoke stimuli. Nicotine Tob Res 3:141–150
Picciotto MR, Zoli M, Lena C, Bessis A, Lallemand Y, Le Novere N, Vincent P, Pich EM, Brulet P, Changeux JP (1995) Abnormal avoidance learning in mice lacking functional high-affinity nicotine receptor in the brain. Nature 374:65–67
Picciotto MR, Zoli M, Rimondini R, Lena C, Marubio LM, Pich EM, Fuxe K, Changeux JP (1998) Acetylcholine receptors containing the beta2 subunit are involved in the reinforcing properties of nicotine. Nature 391:173–177
Pratt WE, Kelley AE (2004) Nucleus accumbens acetylcholine regulates appetitive learning and motivation for food via activation of muscarinic receptors. Behav Neurosci 118:730–739
Rice ME, Cragg SJ (2004) Nicotine amplifies reward-related dopamine signals in striatum. Nat Neurosci 7:583–584
Robbins TW, Everitt BJ (2002) Limbic-striatal memory systems and drug addiction. Neurobiol Learn Mem 78:625–636
Robbins TW, Watson BA, Gaskin M, Ennis C (1983) Contrasting interactions of pipradrol, d-amphetamine, cocaine, cocaine analogues, apomorphine and other drugs with conditioned reinforcement. Psychopharmacology (Berl) 80:113–119
Robinson TE, Berridge KC (2003) Addiction. Annu Rev Psychol 54:25–53
Rose JE, Tashkin DP, Ertle A, Zinser MC, Lafer R (1985) Sensory blockade of smoking satisfaction. Pharmacol Biochem Behav 23:289–293
Schultz W (2002) Getting formal with dopamine and reward. Neuron 36:241–263
Sparks JA, Pauly JR (1999) Effects of continuous oral nicotine administration on brain nicotinic receptors and responsiveness to nicotine in C57Bl/6 mice. Psychopharmacology (Berl) 141:145–153
Stein EA, Pankiewicz J, Harsch HH, Cho JK, Fuller SA, Hoffmann RG, Hawkins M, Rao SM, Bandettini PA, Bloom AS (1998) Nicotine-induced limbic cortical activation in the human brain: a functional MRI study. Am J Psychiatry 155:1009–1015
Tapper AR, McKinney SL, Nashmi R, Schwarz J, Deshpande P, Labarca C, Whiteaker P, Marks MJ, Collins AC, Lester HA (2004) Nicotine activation of alpha4* receptors: sufficient for reward, tolerance, and sensitization. Science 306:1029–1032
Taylor JR, Horger BA (1999) Enhanced responding for conditioned reward produced by intra-accumbens amphetamine is potentiated after cocaine sensitization. Psychopharmacology (Berl) 142:31–40
Taylor JR, Jentsch JD (2001) Repeated intermittent administration of psychomotor stimulant drugs alters the acquisition of Pavlovian approach behavior in rats: differential effects of cocaine, d-amphetamine and 3,4-methylenedioxymethamphetamine (“Ecstasy”). Biol Psychiatry 50:137–143
Taylor JR, Robbins TW (1984) Enhanced behavioural control by conditioned reinforcers following microinjections of d-amphetamine into the nucleus accumbens. Psychopharmacology (Berl) 84:405–412
Tiffany ST, Carter BL (1998) Is craving the source of compulsive drug use? J Psychopharmacol 12:23–30
Waters AJ, Shiffman S, Bradley BP, Mogg K (2003) Attentional shifts to smoking cues in smokers. Addiction 98:1409–1417
Waters AJ, Shiffman S, Sayette MA, Paty JA, Gwaltney CJ, Balabanis MH (2004) Cue-provoked craving and nicotine replacement therapy in smoking cessation. J Consult Clin Psychol 72:1136–1143
Wustenberg DG, Grunewald B (2004) Pharmacology of the neuronal nicotinic acetylcholine receptor of cultured Kenyon cells of the honeybee, Apis mellifera. J Comp Physiol A Neuroethol Sens Neural Behav Physiol 190:807–821
Yin X, Cui W, Hu G, Wang H (2004) Desensitization of alpha7 nicotinic receptors potentiated the inhibitory effect on M-current induced by stimulation of muscarinic receptors in rat superior cervical ganglion neurons. J Neural Transm (in press). DOI 10.1007/s0070200402606
Acknowledgements
This work was supported by grants DA00436, DA14241, and AA15632 from the National Institutes of Health. We wish to thank Chris Kochevar and Elin Lof for experimental assistance and Natalie Tronson for help in modifying the behavioral software.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brunzell, D.H., Chang, J.R., Schneider, B. et al. β2-Subunit-containing nicotinic acetylcholine receptors are involved in nicotine-induced increases in conditioned reinforcement but not progressive ratio responding for food in C57BL/6 mice. Psychopharmacology 184, 328–338 (2006). https://doi.org/10.1007/s00213-005-0099-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-005-0099-z