Abstract
Rationale
Many behavioral effects of delta-9-tetrahydrocannabinol (THC), including its discriminative-stimulus effects, are modulated by endogenous opioid systems.
Objective
To investigate opioid receptor subtypes involved in the discriminative effects of THC.
Methods
Rats trained to discriminate 3 mg/kg i.p. of THC from vehicle using a two-lever operant drug-discrimination procedure, were tested with compounds that bind preferentially or selectively to either mu-, delta- or kappa-opioid receptors.
Results
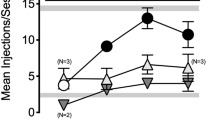
The preferential mu-opioid receptor agonist heroin (0.3–1.0 mg/kg, i.p.), the selective delta-opioid receptor agonist SNC-80 (1–10 mg/kg, i.p.) and the selective kappa-opioid receptor agonist U50488 (1–10 mg/kg, i.p.) did not produce generalization to the discriminative effects of THC when given alone. However, heroin, but not SNC-80 or U50488, significantly shifted the dose–response curve for THC discrimination to the left. Also, the preferential mu-opioid receptor antagonist naltrexone (0.1–1 mg/kg, i.p.), the selective delta-opioid receptor antagonist, naltrindole (1–10 mg/kg, i.p.) and the kappa-opioid receptor antagonist nor-binaltorphimine (n-BNI, 5 mg/kg, s.c.), did not significantly reduce the discriminative effects of the training dose of THC. However, naltrexone, but not naltrindole or n-BNI, significantly shifted the dose–response curve for THC discrimination to the right. Finally, naltrexone, but not naltrindole or n-BNI, blocked the leftward shift in the dose–response curve for THC discrimination produced by heroin.
Conclusions
mu- but not delta- or kappa-opioid receptors are involved in the discriminative effects of THC. Given the role that mu-opioid receptors play in THC’s rewarding effects, the present findings suggest that discriminative-stimulus effects and rewarding effects of THC involve similar neural mechanisms.





Similar content being viewed by others
References
Ameri A. (1999) The effects of cannabinoids on the brain. Prog Neurobiol 58(4):315–348
Barrett RL, Wiley JL, Balster RL, Martin BR (1995) Pharmacological specificity of delta 9-tetrahydrocannabinol discrimination in rats. Psychopharmacology 118:419–424
Bilsky EJ, Calderon SN, Wang T, Bernstein RN, Davis P, Hruby VJ, McNutt RW, Rothman RB, Rice KC, Porreca F (1995) SNC 80, a selective, nonpeptidic and systemically active opioid delta agonist. J Pharmacol Exp Ther 273:359–366
Browne RG, Weissman A (1981) Discriminative stimulus properties of delta 9-tetrahydrocannabinol: mechanistic studies. J Clin Pharmacol 21:227S–234S
Castane A, Robledo P, Matifas A, Kieffer BL, Maldonado R (2003) Cannabinoid withdrawal syndrome is reduced in double mu and delta opioid receptor knockout mice. Eur J Neurosci 17:155–159
Chen JP, Paredes W, Li J, Smith D, Lowinson J, Gardner EL (1990) Delta 9-tetrahydrocannabinol produces naloxone-blockable enhancement of presynaptic basal dopamine efflux in nucleus accumbens of conscious, freely-moving rats as measured by intracerebral microdialysis. Psychopharmacology 102:156–162
Clark JA, Pasternak GW (1988) U50,488: a kappa-selective agent with poor affinity for mu1 opiate binding sites. Neuropharmacology 27:331–332
Colpaert FC (1999) Drug discrimination in neurobiology. Pharmacol Biochem Behav 64:337–345
Colpaert FC (2003) Discovering risperidone: the LSD model of psychopathology. Nat Rev, Drug Discov 2:315–320
Devine DP, Wise RA (1994) Self-administration of morphine, DAMGO, and DPDPE into the ventral tegmental area of rats. J Neurosci 14:1978–1984
De Vries TJ, Shippenberg TS (2002) Neural systems underlying opiate addiction. J Neurosci 22:3321–3325
Gardner EL, Lowinson JH (1991) Marijuana’s interaction with brain reward systems: update 1991. Pharmacol Biochem Behav 40:571–580
Ghozland S, Matthes HW, Simonin F, Filliol D, Kieffer BL, Maldonado R (2002) Motivational effects of cannabinoids are mediated by mu-opioid and kappa-opioid receptors. J Neurosci 22:1146–1154
Goldstein A, Naidu A (1989) Multiple opioid receptors: ligand selectivity profiles and binding site signatures. Mol Pharmacol 36:265–272
Herz A (1997) Endogenous opioid systems and alcohol addiction. Psychopharmacology 129:99–111
Holtzman SG (1985) Drug discrimination studies. Drug Alcohol Depend 14:263–282
Jarbe TU, Henriksson BG (1974) Discriminative response control produced with hashish, tetrahydrocannabinols (delta 8-THC and delta 9-THC), and other drugs. Psychopharmacologia 40:1–16
Jarbe TU, Ohlin GC (1977) Stimulus effects of delta(9)-THC and its interaction with naltrexone and catecholamine blockers in rats. Psychopharmacology 54:193–195
Justinova Z, Tanda G, Munzar P, Goldberg SR (2004) The opioid antagonist naltrexone reduces the reinforcing effects of Delta 9 tetrahydrocannabinol (THC) in squirrel monkeys. Psychopharmacology 173:186–194
Kamendulis LM, Brzezinski MR, Pindel EV, Bosron WF, Dean RA (1996) Metabolism of cocaine and heroin is catalyzed by the same human liver carboxylesterases. J Pharmacol Exp Ther 279:713–717
Kamien JB, Bickel WK, Hughes JR, Higgins ST, Smith BJ (1993) Drug discrimination by humans compared to nonhumans: current status and future directions. Psychopharmacology 111:259–270
Kieffer BL, Gaveriaux-Ruff C (2002) Exploring the opioid system by gene knockout. Prog Neurobiol 66:285–306
Maldonado R (2002) Study of cannabinoid dependence in animals. Pharmacol Ther 95:153–164
Maldonado R, Rodriguez de Fonseca F (2002) Cannabinoid addiction: behavioral models and neural correlates. J Neurosci 22:3326–3331
Maldonado R, Valverde O (2003) Participation of the opioid system in cannabinoid-induced antinociception and emotional-like responses. Eur Neuropsychopharmacol 13:401–410
Mansour A, Fox CA, Akil H, Watson SJ (1995) Opioid-receptor mRNA expression in the rat CNS: anatomical and functional implications. Trends Neurosci 18:22–29
Manzanares J, Corchero J, Romero J, Fernandez-Ruiz JJ, Ramos JA, Fuentes JA (1999) Pharmacological and biochemical interactions between opioids and cannabinoids. Trends Pharmacol Sci 20:287–294
Mello NK, Negus SS (1996) Preclinical evaluation of pharmacotherapies for treatment of cocaine and opioid abuse using drug self-administration procedures. Neuropsychopharmacology 14:375–424
Navarro M, Carrera MR, Fratta W, Valverde O, Cossu G, Fattore L, Chowen JA, Gomez R, del Arco I, Villanua MA, Maldonado R, Koob GF, Rodriguez de Fonseca F (2001) Functional interaction between opioid and cannabinoid receptors in drug self-administration. J Neurosci 21:5344–5350
Portoghese PS, Lipkowski AW, Takemori AE (1987) Binaltorphimine and nor-binaltorphimine, potent and selective kappa-opioid receptor antagonists. Life Sci 40:1287–1292
Prescott WR, Gold LH, Martin BR (1992) Evidence for separate neuronal mechanisms for the discriminative stimulus and catalepsy induced by delta 9-THC in the rat. Psychopharmacology 107:117–124
Schuster CR, Johanson CE (1988) Relationship between the discriminative stimulus properties and subjective effects of drugs. Psychopharmacol Ser 4:161–175
Shippenberg T, Herz A (1987) Motivational properties of opioids. Pol J Pharmacol Pharm 39:577–583
Solinas M, Panlilio LV, Antoniou K, Pappas LA, Goldberg SR (2003) The cannabinoid CB1 antagonist N-piperidinyl-5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methylpyrazole-3-carboxamide (SR-141716A) differentially alters the reinforcing effects of heroin under continuous reinforcement, fixed ratio, and progressive ratio schedules of drug self-administration in rats. J Pharmacol Exp Ther 306:93–102
Solinas M, Zangen A, Thiriet N, SR Goldberg (2004) β-Endorphin elevations in the ventral tegmental area regulate the discriminative effects of delta-9-tetrahydrocannabinol. Eur J Neurosci 19:3183–3192
Spanagel R (1996) The influence of opioid antagonists on the discriminative stimulus effects of ethanol. Pharmacol Biochem Behav 54:645–649
Spanagel R, Almeida OF, Shippenberg TS (1994) Evidence that nor-binaltorphimine can function as an antagonist at multiple opioid receptor subtypes. Eur J Pharmacol 264:157–162
Stolerman I (1992) Drugs of abuse: behavioural principles, methods and terms. Trends Pharmacol Sci 13:170–176
Tanda G, Goldberg SR (2003) Cannabinoids: reward, dependence, and underlying neurochemical mechanisms—a review of recent preclinical data. Psychopharmacology 169:115–134
Tanda G, Pontieri FE, Di Chiara G (1997) Cannabinoid and heroin activation of mesolimbic dopamine transmission by a common mu1 opioid receptor mechanism. Science 276:2048–2050
van Ree JM, Gerrits MA, Vanderschuren LJ (1999) Opioids, reward and addiction: an encounter of biology, psychology, and medicine. Pharmacol Rev 51:341–396
Wiley JL (1999) Cannabis: discrimination of “internal bliss”? Pharmacol Biochem Behav 64:257–260
Wiley JL, Lowe JA, Balster RL, Martin BR (1995) Antagonism of the discriminative stimulus effects of delta 9-tetrahydrocannabinol in rats and rhesus monkeys. J Pharmacol Exp Ther 275:1–6
Yamamura MS, Horvath R, Toth G, Otvos F, Malatynska E, Knapp RJ, Porreca F, Hruby VJ, Yamamura HI (1992) Characterization of [3H]naltrindole binding to delta opioid receptors in rat brain. Life Sci 50:PL119–PL124
Zimmer A, Valjent E, Konig M, Zimmer AM, Robledo P, Hahn H, Valverde O, Maldonado R (2001) Absence of delta-9-tetrahydrocannabinol dysphoric effects in dynorphin-deficient mice. J Neurosci 21:9499–9505
Acknowledgements
The authors want to thank C. Wertheim for her excellent technical assistance and G. Tanda for helpful comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Solinas, M., Goldberg, S.R. Involvement of mu-, delta- and kappa-opioid receptor subtypes in the discriminative-stimulus effects of delta-9-tetrahydrocannabinol (THC) in rats. Psychopharmacology 179, 804–812 (2005). https://doi.org/10.1007/s00213-004-2118-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-004-2118-x