Abstract.
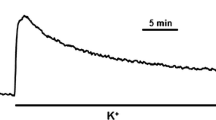
The sarcoplasmic reticulum Ca-ATPase inhibitor thapsigargin (Tg; 0.4–100 nM) produced concentration-related, strong and sustained contractions of the mouse-isolated anococcygeus muscle; these contractions were dependent on extracellular calcium but were only partially reduced (by about 50%) in the presence of verapamil (10 and 100 µM). The verapamil-resistant component of the Tg-induced contraction was relaxed by the general calcium entry blockers SKF96365 (0.4–40 µM) and cadmium (50–300 µM), and by the tyrosine kinase inhibitor genistein (10–180 µM).
In single smooth muscle cells loaded with Fura-2, addition of Tg (100 nM) to calcium-free medium produced a small, transient increase in fluorescence; subsequent addition of calcium (2.5 mM) produced a larger and sustained increase which was abolished on return to calcium-free conditions, but was only partially reduced by verapamil (10 µM; by about 30%). Manganese quenching of Fura-2 was enhanced in cells treated with Tg. The verapamil-resistant calcium influx was reduced by SKF96365 (20 µM) and to a lesser extent by genistein (40 µM); cadmium (200 µM) produced an initial decrease in fluorescence followed by a marked increase.
These results demonstrate that, in the mouse anococcygeus, Tg can cause sustained contractions and elevations of calcium influx in the presence of verapamil; the time-course, calcium dependence and, although to a lesser extent, pharmacology of these effects generally support the proposal that excitation-contraction coupling in this tonic smooth muscle involves sustained capacitative calcium influx.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Wallace, P., Ayman, S., McFadzean, I. et al. Thapsigargin-induced tone and capacitative calcium influx in mouse anococcygeus smooth muscle cells. Naunyn-Schmiedeberg's Arch Pharmacol 360, 368–375 (1999). https://doi.org/10.1007/s002109900100
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s002109900100