Abstract
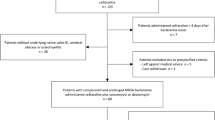
Infections caused by multidrug-resistant (MDR) bacteria are typically associated with high morbidity and mortality, especially in vulnerable individuals such as patients with prolonged hospitalizations, immunocompromised individuals, and the elderly. This study aimed to provide post-marketing surveillance results concerning the prevalence of antibiotic resistance against Gram-negative bacteria through the collaboration of a multidisciplinary team. Patients involved have been treated with new antibacterial drugs, in particular ceftazidime/avibactam (C/A), meropenem/vaborbactam (M/V), cefiderocol, and ceftolozane/tazobactam (C/T). The most resistant bacterial species were Klebsiella spp., Pseudomonas aeruginosa, and Acinetobacter baumannii. Italian Drug Agency (AIFA) monitoring records for inpatients have been collected and analyzed, assessing the characteristics of the patients involved. Adverse drug reactions (ADRs) and drugs involved have been reported using a descriptive analytical approach. All data have been collected retrospectively from patient’s medical records and entered into an electronic case report form (CRF). Among the 104 treated patients, Klebsiella spp. accounted for 50.1% of infections, Pseudomonas aeruginosa for 32.7%, Acinetobacter baumannii for 3%, and other bacterial species for 1.92% configuring polymicrobial infections. Regarding treatment outcomes, healing was achieved in 61 (58.6%) patients, 23 (22.1%) patients died, 8 (7.7%) patients discontinued empirical therapy, and 3 (2.9%) patients were lost to follow-up. Despite the introduction of new antibacterial drugs active against Gram-negative bacteria is improving the clinical scenario, it is crucial that the use of new antibacterial drugs be implemented by appropriate antimicrobial stewardship, surveillance programs, and monitoring efforts to prevent further spread of resistance. This study showed that the new antibiotics have good efficacy against MDR bacteria and cause negligible side effects.
Similar content being viewed by others
Data Availability
No datasets were generated or analysed during the current study.
Code availability
Not applicable.
Abbreviations
- ESBL:
-
Extended spectrum beta-lactamase
- KPC:
-
Klebsiella pneumoniae Carbapenemase
- OXA:
-
Oxacillinases
- MBL:
-
Metallo-beta-lactamase
- MRSA:
-
Methicillin-resistant Staphylococcus aureus
- VIM:
-
Verona integron-encoded
References
Annual Epidemiological Reports (AERs) (2024) https://www.ecdc.europa.eu/en/publications-data/monitoring/all-annual-epidemiological-reports. Accesed January 2024
Catalano A, Iacopetta D, Ceramella J et al (2022) Multidrug resistance (MDR): a widespread phenomenon in pharmacological therapies. Molecules 27(3):616. https://doi.org/10.3390/molecules27030616
Dettori S, Portunato F, Vena A et al (2023) Severe infections caused by difficult-to-treat Gram-negative bacteria. Curr Opin Crit Care 29(5):438–445. https://doi.org/10.1097/MCC.0000000000001074
European Centre for Disease Prevention and Control. ECDC (2019) https://www.ecdc.europa.eu/en/publications-data/surveillance-systems-overview-2019. Accessed April 2024
European Centre for Disease Prevention and Control. ECDC (2023) https://atlas.ecdc.europa.eu/public/index.aspx. Accessed December 2023
Ferrara F, Zovi A, Nava E et al (2023) SARS-CoV-2 caused a surge in antibiotic consumption causing a silent pandemic inside the pandemic. A retrospective analysis of Italian data in the first half of 2022. Ann Pharm Fr 28:S0003-4509(23)00022-6. https://doi.org/10.1016/j.pharma.2023.02.003
Florez-Cuadrado D, Moreno MA, Ugarte-Ruíz M et al (2018) Antimicrobial resistance in the food chain in the European Union. Adv Food Nutr Res 86:115–136
Giddins MJ, Macesic N, Annavajhala MK et al (2018) Successive emergence of ceftazidime-avibactam resistance through distinct genomic adaptations in blaKPC-2-harboring Klebsiella pneumoniae sequence type 307 isolates. Antimicrob Agents Chemother 62. https://doi.org/10.1128/AAC.02101-17
Italian Medicines Agency AIFA (2023). https://www.aifa.gov.it/documents/20142/1787183/AIFA-OPERA_Raccomandazioni_pazienti_ospedalizzati.pdf. Accessed April 2024
Italian Medicines Agency AIFA (2024) https://www.aifa.gov.it/documents/20142/1787183/AIFAOPERA_Raccomandazioni_pazienti_ospedalizzati.pdf. Accessed January 2024
Italian Ministry of Health (2022a) https://www.salute.gov.it/portale/antibioticoresistenza/glossarioAntibioticoResistenza.jsp?lingua=italiano&area=antibiotico-resistenza&menu=glossario&tab=1. Accessed January 2024
Italian Ministry of Health (2022b) Piano Nazionale di Contrasto all’Antibiotico-Resistenza (PNCAR) 2022–2025. https://www.salute.gov.it/imgs/C_17_pubblicazioni_3294_allegato.pdf. Accessed December 2023
Italian Superior Institute of Health (2019) https://www.iss.it/one-health. Accessed December 2023
Lepape A, Jean A, De Waele J et al (2020) European intensive care physicians’ experience of infections due to antibiotic-resistant bacteria. Antimicrob Resist Infect Control 9(1):1
Li D, Huang X, Rao H et al (2023) Klebsiella pneumoniae bacteremia mortality: a systematic review and meta-analysis. Front Cell Infect Microbiol 20(13):1157010. https://doi.org/10.3389/fcimb.2023.1157010
Littmann J, Viens AM (2015) The ethical significance of antimicrobial resistance. Public Health Ethics 8(3):209–224
Liu P, Li X, Luo M et al (2018) Risk factors for carbapenem-resistant Klebsiella pneumoniae infection: a meta-analysis. Microb Drug Resist 24:190–198
McEwen SA, Collignon PJ (2018) Antimicrobial resistance: a one health perspective. Microbiol Spectr 6(2). https://doi.org/10.1128/microbiolspec.ARBA-0009-2017
Paul M, Carrara E, Retamar P et al (2022) European Society of Clinical Microbiology and Infectious Diseases (ESCMID) guidelines for the treatment of infections caused by multidrug-resistant Gram-negative bacilli (endorsed by European Society of Intensive Care Medicine). Clin Microbiol Infect 28:521–547
Pérez-Galera S, Bravo-Ferrer JM, Paniagua M et al (2023) Risk factors for infections caused by carbapenem-resistant Enterobacterales: an international matched case-control-control study (EURECA). E Clin Med 57:101871
Quotidiano Sanità (2024) Antibiotico resistenza. Via libera dell’Aifa alla nuova terapia per contrastarla e razionalizzare l’uso di carbapenemici. https://www.quotidianosanita.it/scienza-e-farmaci/articolo.php?articolo_id=44968. Accesed January 2024
Solomkin J, Hershberger E, Miller B et al (2015) Ceftolozane/tazobactam plus metronidazole for complicated intra-abdominal infections in an era of multidrug resistance: results from a randomized, double-blind, phase 3 trial (ASPECT-cIAI). Clin Infect Dis 60(10):1462–71. https://doi.org/10.1093/cid/civ097
WHO (2015) Global action plan on antimicrobial resistance. https://www.who.int/publications/i/item/9789241509763. Accessed Jan 2024.
Zeng M, Xia J, Zong Z et al (2023) Society of Bacterial Infection and Resistance of Chinese Medical Association; Expert Committee on Clinical Use of Antimicrobial Agents and Evaluation of Antimicrobial Resistance of the National Health Commission; Infectious Diseases Society of Chinese Medical Education Association. Guidelines for the diagnosis, treatment, prevention and control of infections caused by carbapenem-resistant gram-negative bacilli. J Microbiol Immunol Infect 56(4):653–671. https://doi.org/10.1016/j.jmii.2023.01.017
Acknowledgements
We would like to thank the IRCCS San Gerardo dei Tintori Foundation in Monza for the concession and willingness to use the data within the framework of Dr. Anna Chiara Aloisi's specialisation thesis.
Author information
Authors and Affiliations
Contributions
FF: conceptualization, writing—original draft, methodology, supervision, validation. RP: conceptualization, writing—original draft. ACA: writing—original draft. GP: writing—review and editing, supervision. GB: writing—review and editing, validation. MC: writing—review and editing. AZ: conceptualization, writing—original draft, validation. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
The authors consent to the publication of the manuscript.
Competing interests
The authors declare no competing interests.
Disclosure
The authors declare that the opinions expressed are of a personal nature and do not in any way commit the responsibility of the administrations to which they belong.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ferrara, F., Pasquinucci, R., Aloisi, A.C. et al. The use of new antibacterial drugs against infections caused by multidrug-resistant Gram-negative bacteria: an Italian real-world evidence study in a Lombardy hospital. Naunyn-Schmiedeberg's Arch Pharmacol (2024). https://doi.org/10.1007/s00210-024-03117-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00210-024-03117-4