Abstract
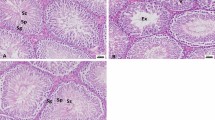
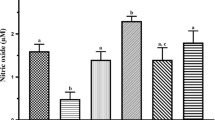
Oxidative stress has been linked with lead toxicity, including lead-induced sexual dysfunction. On the contrary, sodium acetate has been proven to exert antioxidant activity. However, the effect of sodium acetate on lead-induced sexual dysfunction has not been fully explored. This study investigated the effect of sodium acetate on lead-induced sexual dysfunction, exploring the involvement of testosterone, eNOS/NO/cGMP, and Nrf2/HO-1 signaling. Twenty male Wistar rats with similar weights were randomly assigned into four groups (n = 5 rats/group) after two weeks of acclimatization. Animals were vehicle-treated (0.5 ml/day of distilled water, per os), acetate-treated (200 mg/kg/day, per os), lead-treated (20 mg/kg/day, per os), or lead + acetate-treated. The results revealed that sodium acetate treatment attenuated lead-induced rise in penile lead, malondialdehyde and oxidized glutathione concentrations, and acetylcholinesterase activity. In addition, lead exposure prolonged mount, intromission, and ejaculation latency and reduced mount, intromission, and ejaculation frequency, as well as the motivation to mate and penile reflex, which were improved by acetate treatment. More so, acetate treatment ameliorated lead-induced reductions in absolute and relative penile weight, eNOS, NO, cGMP, luteinizing hormone, follicle-stimulating hormone, testosterone, dopamine, Nrf2, HO-1, and reduced glutathione concentrations, as well as glutathione reductase, glutathione peroxidase, glutathione-S-transferase, superoxide dismutase, and catalase activities. In conclusion, this study demonstrates that sodium acetate attenuated lead-induced sexual dysfunction by upregulating testosterone-dependent eNOS/NO/cGMP and Nrf2/HO-1 signaling. Despite the compelling data presented in this study, other possible associated mechanisms in the protective role of acetate should be explored.








Similar content being viewed by others
Data availability
The data used to support the findings of the present study are available from the corresponding author upon request.
References
Adeyemi DH, Obembe OO, Hamed MA, Akhigbe RE (2023) Sodium acetate ameliorates doxorubicin-induced cardiac injury via upregulation of Nrf2/HO-1 signaling and downregulation of NFkB-mediated apoptotic signaling in Wistar rats. Naunyn-Schmiedeberg's Arch Pharmacol: 1–3
Ajayi AF, Akhigbe RE (2020a) Assessment of sexual behaviour and fertility indices in male rabbits. Andrology 8:509–515
Ajayi AF, Akhigbe RE (2020b) Staging of the estrous cycle and induction of estrus in experimental rodents: an update. Fertil Res Pract 6:5
Akhigbe R, Ajayi A (2020) Testicular toxicity following chronic codeine administration is via oxidative DNA damage and up-regulation of NO/TNF-α and caspase 3 activities. PLoS One 15(3):e0224052
Akhigbe R, Ajayi A (2021) The impact of reactive oxygen species in the development of cardiometabolic disorders: a review. Lipids Health Dis 20:23
Akhigbe RE, Dutta S, Sengupta P, Chhikara BS (2021a) Adropin in immune and energy balance: ‘a molecule of interest’ in male reproduction. Chem Biol Lett 8(4):213–223
Akhigbe RE, Ajayi AF, Ram SK (2021b) Oxidative stress and cardiometabolic disorders. BioMed Res Int 2021:9872109. https://doi.org/10.1155/2021/9872109
Akhigbe RE, Hamed MA, Aremu AO (2021c) HAART exacerbates testicular damage and impaired spermatogenesis in anti-Koch-treated rats via dysregulation of lactate transport and glutathione content. Reprod Toxicol 103:96–107
Akhigbe RE, Afolabi OA, Ajayi AF (2022a) L-Arginine reverses maternal and pre-pubertal codeine exposure-induced sexual dysfunction via upregulation of androgen receptor gene and NO/ cGMP signaling. PLoS One 17(9):e0274411
Akhigbe RE, Oluwole DT, Adegoke TE, Hamed MA, Anyogu DC, Ajayi AF (2022b) Suppression of glutathione system and upregulation of caspase 3-dependent apoptosis mediate rohypnol-induced gastric injury. Redox Rep 27(1):111–118
Akhigbe RE, Hamed MA, Odetayo AF, Akhigbe TM, Oyedokun PA (2023) Zinc improves sexual and erectile function in HAART-treated rats via the upregulation of erectogenic enzymes and maintenance of redox balance. Aging Male 26:2205517
Anis TH, ElKaraksy A, Mostafa T, Gadalla A, Imam H, Hamdy L, Abu el-Alla O (2007) Chronic lead exposure may be associated with erectile dysfunction. J Sex Med 4(5):1428–36
Besong EE, Akhigbe TM, Ashonibare PJ, Oladipo AA, Obimma JN, Hamed MA, Adeyemi DH, Akhigbe RE (2023a) Zinc improves sexual performance and erectile function by preventing penile oxidative injury and upregulating circulating testosterone in lead-exposed rats. Redox Rep 28(1):2225675
Besong EE, Ashonibare PJ, Obembe OO, Folawiyo MA, Adeyemi DH, Hamed MA, Akhigbe TM, Akhigbe RE (2023b) Zinc protects against lead-induced testicular damage via modulation of steroidogenic and xanthine oxidase/uric acid/caspase 3-mediated apoptotic signaling in male Wistar rats. Aging Male 26(1):2224428
Beutler E, Duron O, Kelly BM (1963) Improved method for the determination of blood glutathione. J Lab Clin Med 61:882–888
Bhardwaj JK, Paliwal A, Saraf P (2021) Effects of heavy metals on reproduction owing to infertility. J Biochem Mol Toxicol 35(8):e22823
Bivalacqua TJ, Usta MF, Champion HC, Kadowitz PJ, Hellstrom WJG (2003) Endothelial dysfunction in erectile dysfunction: role of the endothelium in erectile physiology and disease. J Androl 24:S17–S37
Cheng X, Ku CH, Siow RC (2013) Regulation of the Nrf2 antioxidant pathway by microRNAs: new players in micromanaging redox homeostasis. Free Radical Biol Med 64:4–11
Euler HV, Josephson K (1972) Uberkatalase. I. Eur J Org Chem 452:158–181
Fridovich I, Misra HP (1972) The role of superoxide anion in the autooxidation of epinephrine and a simple assay for superoxide dismutase. J Biol Chem 247:3170–3175
Frost G, Sleeth ML, Sahuri-Arisoylu M, Lizarbe B, Cerdan S, Brody L, Anastasovska J, Ghourab S, Hankir M, Zhang S, Carling D (2014) The short-chain fatty acid acetate reduces appetite via a central homeostatic mechanism. Nat Commun 5(1):3611
Frye C, Bo E, Calamandrei G, Calza L, Dessì-Fulgheri F, Fernández M, Fusani L, Kah O, Kajta M, Le Page Y, Patisaul HB (2012) Endocrine disrupters: a review of some sources, effects, and mechanisms of actions on behaviour and neuroendocrine systems. J Neuroendocrinol 24(1):144–159
Garban H, Vernet D, Freedman A, Rajfer J, Gonzalez-Cadavid NF (1995) Effect of aging on nitric oxide-mediated penile erection in rats. Am Muscle J Urol 153:442A
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione-S-transferases. The first enzymatic step in mercapturic acid formation. J Biol Chem 249(22):7130–9
Horn HD, Burns FH (1965). In: Bergmeyer H-U (ed) Methods of enzymatic analysis. Academic Press, Cambridge, pp 875–879
Hsu PC, Liu MY, Hsu CC, Chen LY, Guo YL (1997) Lead exposure causes generation of reactive oxygen species and functional impairment in rat sperm. Toxicology 122(1–2):133–143
Hu LL, Zhang KQ, Tian T, Zhang H, Fu Q (2018) Probucol improves erectile function via activation of Nrf2 and coordinates the HO-1/DDAH/PPAR-γ/eNOS pathways in streptozotocin-induced diabetic rats. Biochem Biophys Res Commun 507(1–4):9–14
Ige SF, Akhigbe RE (2012) The role of Allium cepa on aluminum-induced reproductive dysfunction in experimental male rat models. J Hum Reprod Sci 5(2):200
Ige SF, Olaleye SB, Akhigbe RE, Akanbi TA, Oyekunle OA, Udoh UA (2012) Testicular toxicity and sperm quality following cadmium exposure in rats: ameliorative potentials of Allium cepa. J Hum Reprod Sci 5(1):37
Kang TC (2020) Nuclear factor-erythroid 2-related factor 2 (Nrf2) and mitochondrial dynamics/mitophagy in neurological diseases. Antioxidants 9(7):617
Kelainy EG, Ibrahim Laila IM, Ibrahim SR (2019) The effect of ferulic acid against lead-induced oxidative stress and DNA damage in kidney and testes of rats. Environ Sci Pollut Res 26:31675–31684
Lugg JA, Schlunt K, Rajfer J, Gonzalez-Cadavid NF (1995) Modulation of nitric oxide synthase activity in the penis by electrical stimulation of the cavernosal nerve. J Urol 153:509A
Massányi P, Massányi M, Madeddu R, Stawarz R, Lukáč N (2020) Effects of cadmium, lead, and mercury on the structure and function of reproductive organs. Toxics 8(4):94
Moriyama M, Kurebayashi R, Kawabe K, Takano K, Nakamura Y (2016) Acetate attenuates lipopolysaccharide-induced nitric oxide production through an anti-oxidative mechanism in cultured primary rat astrocytes. Neurochem Res 41:3138–3146
Oyeleye SI, Adefegha SA, Dada FA et al (2019) Effect of p-coumaric acid on the erectogenic enzyme activities and non-protein thiol level in the penile tissue of normal and doxorubicin-induced oxidative stress male rat. Andrologia 51(6):e13281
Papanikolaou NC, Hatzidaki EG, Belivanis S, Tzanakakis GN, Tsatsakis AM (2005) Lead toxicity update. A brief review. Med Sci Monit 11(10):RA329
Pickard RS, Powell PH, Zar MA (1995) Nitric oxide and cyclic GMP formation following relaxant nerve stimulation in isolated human corpus cavernosum. Br J Urol 75:516–522
Pourbagher-Shahri AM, Farkhondeh T, Talebi M, Kopustinskiene DM, Samarghandian S, Bernatoniene J (2021) An overview of NO signaling pathways in aging. Molecules 26(15):4533
Rajfer J, Aronson WJ, Bush PA, Dorey FJ, Ignarro LJ (1992) Nitric oxide as a mediator of relaxation of the corpus cavernosum on response to NO plays a major role in the physiological regulation of nonadrenergic, noncholinergic neurotransmission. N Engl J Med 326:90–94
Rotruck JT, Pope AL, Ganther HE, Swanson AB, Hafeman DG, Hoekstra WG (1973) Selenium: biochemical role as a component of glutathione peroxidase. Science 179(4073):588–590
Swaney WT, Dubose BN, Curley JP, Champagne FA (2012) Sexual experience affects reproductive behaviour and preoptic androgen receptors in male mice. Horm Behav 61:472–478
Udefa AL, Amama EA, Archibong EA, Nwangwa JN, Adama S, Inyang VU, Inyaka GU, Aju GJ, Okpa S, Inah IO (2020) Antioxidant, anti-inflammatory and anti-apoptotic effects of hydro-ethanolic extract of Cyperus esculentus L.(tigernut) on lead acetate-induced testicular dysfunction in Wistar rats. Biomed Pharmacother 129:110491
Vukelić D, Djordjevic AB, Anđelković M, Repić A, Baralić K, Ćurčić M, Đukić-Ćosić D, Boričić N, Antonijević B, Bulat Z (2023) Derivation of benchmark doses for male reproductive toxicity in a subacute low-level Pb exposure model in rats. Toxicol Lett 375:69–76
Wu T, Li J, Li Y, Song H (2017) Antioxidant and hepatoprotective effect of swertiamarin on carbon tetrachloride-induced hepatotoxicity via the Nrf2/HO-1 pathway. Cell Physiol Biochem 41(6):2242–2254
Zhang Z, Yu J, Xie J et al (2021) Improvement roles of zinc supplementation in low dose lead-induced testicular damage and glycolytic inhibition in mice. Toxicology 462:152933
Zheng Y, Zhang Q, Jing L, Fei Y, Zhao H (2023) The effects of chronic lead exposure on testicular development of Japanese quail (Coturnix japonica): histopathological damages, oxidative stress, steroidogenesis disturbance, and hypothalamus-pituitary-testis axis disruption. Biol Trace Elem Res 201(7):3446–3460
Funding
This study was self-funded.
Author information
Authors and Affiliations
Contributions
Conceptualization and design: BEE and ARE. Data analysis: APJ, ATM, and ARE. Funding acquisition: BEE, APJ, ATM, OJN, and ARE. Investigation: BEE, APJ, ATM, OJN, and ARE. Methodology: BEE, APJ, ATM, OJN, and ARE. Project administration: BEE, APJ, ATM, OJN, and ARE. Supervision: BEE, APJ, ATM, OJN, and ARE. Validation: BEE, APJ, ATM, OJN, and ARE. Writing—original draft: APJ and ARE. Writing—review and editing and final approval: BEE, APJ, ATM, OJN, and ARE. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethical approval
The research was approved by the institution’s Ethics Review Committee (OGH/2022/517).
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Besong, E.E., Ashonibare, P.J., Akhigbe, T.M. et al. Sodium acetate abates lead-induced sexual dysfunction by upregulating testosterone-dependent eNOS/NO/cGMP signaling and activating Nrf2/HO-1 in male Wistar rat. Naunyn-Schmiedeberg's Arch Pharmacol 397, 1233–1243 (2024). https://doi.org/10.1007/s00210-023-02696-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02696-y