Abstract
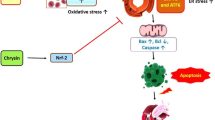
Aging is known as a main risk factor in the development of cardiovascular diseases. Naringin (NRG) is a flavonoid compound derived from citrus fruits. It possesses a wide spectrum of pharmacological properties, including antioxidant anti-inflammatory, and cardioprotective. This investigation aimed to assess the cardioprotective effect of NRG against the ischemia/reperfusion (I/R) injury in aged rats. In this study, D-galactose (D-GAL) at the dose of 150 mg/kg/day for 8 weeks was used to induce aging in rats. Rats were orally gavaged with NRG (40 or 100 mg/kg/day), in co-treatment with D-GAL, for 8 weeks. The Langendorff isolated heart was used to evaluate the effect of NRG on I/R injury in aged rats. NRG treatment diminished myocardial hypertrophy and maximum contracture level in aged animals. During the pre-ischemic phase, reduced heart rate was normalized by NRG. The effects of D-GAL on the left ventricular end diastolic pressure (LVDP), the rate pressure product (RPP), and the minimum and maximum rate of left ventricular pressure (±dp/dt) improved by NRG treatment in the perfusion period. NRG also enhanced post-ischemic recovery of cardiac functional parameters (± dp/dt, and RPP) in isolated hearts. An increase in serum levels of the lactate dehydrogenase (LDH), the creatine kinase-MB (CK-MB), and the tumor necrosis factor-alpha (TNF-α) were reversed by NRG in aged rats. It also normalized the D-GAL-decreased the superoxide dismutase (SOD) activity in the heart tissue. NRG treatment alleviated cardiac injury in aged hearts under conditions of I/R. NRG may improve aging-induced cardiac dysfunction through anti-oxidative and anti-inflammatory mechanisms.










Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- ANOVA:
-
Analysis of variance
- BW:
-
Body weight
- CaCl2:
-
Calcium chloride
- CF:
-
Coronary flow
- CK-MB:
-
Creatine kinase-MB
- CVDs:
-
Cardiovascular diseases
- D-GAL:
-
D-Galactose
- dp/dt Max :
-
Maximum rate of left ventricular pressure
- dp/dt Min :
-
Minimum rate of left ventricular pressure
- ELISA:
-
Enzyme-linked immunosorbent assay
- GCL:
-
Glutamate cysteine ligase
- GPx:
-
Glutathione peroxidase
- HR:
-
Heart rate
- HO-1:
-
Heme oxygenase-1
- HW:
-
Heart weight
- I/R:
-
Ischemia/reperfusion
- KCl:
-
Potassium chloride
- KH2PO4:
-
Potassium dihydrogenephosphate
- LDH:
-
Lactate dehydrogenase
- LVDP:
-
Left ventricular developed pressure
- LVEDP:
-
Left ventricular end diastolic pressure
- LVSP:
-
Left ventricular systolic pressure
- MC:
-
Maximum contracture
- MgSO4:
-
Magnesium sulfate
- MI:
-
Myocardial infarction
- NaCl:
-
Sodium chloride
- NaHCO3:
-
Sodium bicarbonate
- NOAEL:
-
No-observed-adverse-effect level
- Nrf2:
-
Nuclear factor erythroid 2-related factor 2
- NRG:
-
Naringin
- PBS:
-
Phosphate buffer saline
- ROS:
-
Reactive oxygen species
- RPP:
-
Rate pressure product
- SD:
-
Standard deviation
- SEM:
-
Standard error of measurement
- SOD:
-
Superoxide dismutase
- TNF-α:
-
Tumor necrosis factor-alpha
- U/L:
-
Units per liter of sample
- U/mg:
-
Unit per milligram
References
Adebiyi AO, Adebiyi OO, Owira PMO (2016) Naringin mitigates cardiac hypertrophy by reducing oxidative stress and inactivating c-Jun nuclear kinase-1 protein in type I diabetes. J Cardiovasc Pharmacol 67:136–144. https://doi.org/10.1097/FJC.0000000000000325
Aki̇n AT, El Bechir ML, Kaymak E, et al (2022) Anti-inflammatory and anti-apoptotic effects of naringin on bacterial endotoxin-induced small intestine damage in rats. Cukurova Med J 47:1137–1146. https://doi.org/10.17826/cumj.1124641
Alotaibi F (2023) Naringenin alters the pharmacokinetics of ranolazine in part through the inhibition of cytochrome P450 (3A4) and P-glycoprotein. Futur J Pharm Sci 9:23. https://doi.org/10.1186/s43094-023-00477-1
Aman Y, Schmauck-Medina T, Hansen M et al (2021) Autophagy in healthy aging and disease. Nat Aging 1:634–650. https://doi.org/10.1038/s43587-021-00098-4
Azman KF, Zakaria R (2019) d-Galactose-induced accelerated aging model: an overview. Biogerontology 20:763–782. https://doi.org/10.1007/s10522-019-09837-y
Bhia M, Motallebi M, Abadi B et al (2021) Naringenin nano-delivery systems and their therapeutic applications. Pharmaceutics 13:291. https://doi.org/10.3390/pharmaceutics13020291
Bo-Htay C, Palee S, Apaijai N et al (2018) Effects of d-galactose-induced ageing on the heart and its potential interventions. J Cell Mol Med 22:1392–1410. https://doi.org/10.1111/jcmm.13472
Castelli R, Gidaro A, Casu G, et al (2023) Aging of the arterial system. Int J Mol Sci 24:. https://doi.org/10.3390/ijms24086910
Chen RC, Sun GB, Wang J et al (2015) Naringin protects against anoxia/reoxygenation-induced apoptosis in H9c2 cells via the Nrf2 signaling pathway. Food Funct 6:1331–1344. https://doi.org/10.1039/c4fo01164c
Chen X, Yu J, Zheng L et al (2022) Quercetin and lycopene co-administration prevents oxidative damage induced by d-galactose in mice. Food Biosci 50:102042. https://doi.org/10.1016/j.fbio.2022.102042
Cheng J, Ren C, Cheng R et al (2021) Mangiferin ameliorates cardiac fibrosis in D-galactose-induced aging rats by inhibiting TGF-β/p38/MK2 signaling pathway. Korean J Physiol Pharmacol 25:131–137. https://doi.org/10.4196/kjpp.2021.25.2.131
de Almeida AJPO, de Almeida Rezende MS, Dantas SH et al (2020) Unveiling the role of inflammation and oxidative stress on age-related cardiovascular diseases. Oxid Med Cell Longev 2020:1954398. https://doi.org/10.1155/2020/1954398
Dong M, Chen D, Zhu Y et al (2023) Impaired regulation of MMP2/16–MLCK3 by miR-146a-5p increased susceptibility to myocardial ischaemic injury in aging mice. Cardiovasc Res 119:786–801. https://doi.org/10.1093/cvr/cvac104
Dumea E, Lazar M, Barbu EC et al (2022) Pulmonary involvement in SARS-CoV-2 Infection Estimates Myocardial Injury Risk. Medicina (B Aires) 58:1436. https://doi.org/10.3390/medicina58101436
Ferrucci L, Fabbri E (2018) Inflammageing: chronic inflammation in ageing, cardiovascular disease, and frailty. Nat Rev Cardiol 15:505–522. https://doi.org/10.1038/s41569-018-0064-2
Franceschi C, Garagnani P, Morsiani C et al (2018) The continuum of aging and age-related diseases: common mechanisms but different rates. Front Med 5:61. https://doi.org/10.3389/fmed.2018.00061
Fuhr U, Kummert AL (1995) The fate of naringin in humans: a key to grapefruit juice-drug interactions? Clin Pharmacol Ther 58:365–373. https://doi.org/10.1016/0009-9236(95)90048-9
Gelen V, Şengül E (2020) Antioxidant, anti-inflammatory and antiapoptotic effects of Naringin on cardiac damage induced by cisplatin. Indian J Tradit Knowl 19:459–465. https://doi.org/10.56042/ijtk.v19i2.35371
Ghanbari-Movahed M, Jackson G, Farzaei MH, Bishayee A (2021) A systematic review of the preventive and therapeutic effects of naringin against human malignancies. Front Pharmacol 12:639840. https://doi.org/10.3389/fphar.2021.639840
He F, Ru X, Wen T (2020) NRF2, a transcription factor for stress response and beyond. Int J Mol Sci 21:4777. https://doi.org/10.3390/ijms21134777
HeidaryMoghaddam R, Samimi Z, Moradi SZ et al (2020) Naringenin and naringin in cardiovascular disease prevention: a preclinical review. Eur J Pharmacol 887:173535. https://doi.org/10.1016/j.ejphar.2020.173535
Hosseini L, Vafaee MS, Badalzadeh R (2020) Melatonin and nicotinamide mononucleotide attenuate myocardial ischemia/reperfusion injury via modulation of mitochondrial function and hemodynamic parameters in aged rats. J Cardiovasc Pharmacol Ther 25:240–250. https://doi.org/10.1177/1074248419882002
Kleinbongard P, Bøtker HE, Ovize M et al (2020) Co-morbidities and co-medications as confounders of cardioprotection-does it matter in the clinical setting? Br J Pharmacol 177:5252–5269. https://doi.org/10.1111/bph.14839
Korzick DH, Lancaster TS (2013) Age-related differences in cardiac ischemia–reperfusion injury: effects of estrogen deficiency. Pflügers Arch J Physiol 465:669–685. https://doi.org/10.1007/s00424-013-1255-7
Lee C-H, Shin H-W, Shin D-G (2020) Impact of oxidative stress on long-term heart rate variability: linear versus non-linear heart rate dynamics. Hear Lung Circ 29:1164–1173. https://doi.org/10.1016/j.hlc.2019.06.726
Li F, Zhan Z, Qian J et al (2021) Naringin attenuates rat myocardial ischemia/reperfusion injury via PI3K/Akt pathway-mediated inhibition of apoptosis, oxidative stress and autophagy. Exp Ther Med 22:1–8. https://doi.org/10.3892/etm.2021.10243
Li P, Wang S, Guan X et al (2013) Acute and 13weeks subchronic toxicological evaluation of naringin in Sprague-Dawley rats. Food Chem Toxicol 60:1–9. https://doi.org/10.1016/j.fct.2013.07.019
Li P, Wu H, Wang Y et al (2020) Toxicological evaluation of naringin: acute, subchronic, and chronic toxicity in Beagle dogs. Regul Toxicol Pharmacol 111:104580. https://doi.org/10.1016/j.yrtph.2020.104580
Liberale L, Montecucco F, Tardif J-C et al (2020) Inflamm-ageing: the role of inflammation in age-dependent cardiovascular disease. Eur Heart J 41:2974–2982. https://doi.org/10.1093/eurheartj/ehz961
Maharajan N, Cho G-W (2021) Camphorquinone promotes the antisenescence effect via activating AMPK/SIRT1 in stem cells and D-galactose-induced aging mice. Antioxidants 10:1916. https://doi.org/10.3390/antiox10121916
Martín-Fernández B, Gredilla R (2016) Mitochondria and oxidative stress in heart aging. Age (Omaha) 38:225–238. https://doi.org/10.1007/s11357-016-9933-y
de Mikael RL, de Paiva AMG, Gomes MM et al (2017) Vascular aging and arterial stiffness. Arq Bras Cardiol 109:253–258. https://doi.org/10.5935/abc.20170091
Moghaddam RH, Samimi Z, Moradi SZ et al (2020) Naringenin and naringin in cardiovascular disease prevention: a preclinical review. Eur J Pharmacol 887:173535. https://doi.org/10.1016/j.ejphar.2020.173535
Montllor-Albalate C, Kim H, Thompson AE et al (2022) Sod1 integrates oxygen availability to redox regulate NADPH production and the thiol redoxome. Proc Natl Acad Sci 119:e2023328119. https://doi.org/10.1073/pnas.2023328119
Papaconstantinou J (2019) The role of signaling pathways of inflammation and oxidative stress in development of senescence and aging phenotypes in cardiovascular disease. Cells 8:1383. https://doi.org/10.3390/cells8111383
Park JH, Ku HJ, Kim JK et al (2018) Amelioration of high fructose-induced cardiac hypertrophy by naringin. Sci Rep 8:9464. https://doi.org/10.1038/s41598-018-27788-1
Randhawa PK, Bali A, Virdi JK, Jaggi AS (2018) Conditioning-induced cardioprotection: Aging as a confounding factor. Korean J Physiol Pharmacol Off J Korean Physiol Soc Korean Soc Pharmacol 22:467–479. https://doi.org/10.4196/kjpp.2018.22.5.467
Sanchez-Morate E, Gimeno-Mallench L, Stromsnes K et al (2020) Relationship between diet, microbiota, and healthy aging. Biomedicines 8:287. https://doi.org/10.3390/biomedicines8080287
Shackebaei D, Hesari M, Ramezani-Aliakbari S et al (2022) Gallic acid protects against isoproterenol-induced cardiotoxicity in rats. Hum Exp Toxicol 41:09603271211064532. https://doi.org/10.1177/09603271211064532
Vatner SF, Zhang J, Vyzas C et al (2021) Vascular stiffness in aging and disease. Front Physiol 12:762437. https://doi.org/10.3389/fphys.2021.762437
Viswanatha GL, Shylaja H, Keni R et al (2022) A systematic review and meta-analysis on the cardio-protective activity of naringin based on pre-clinical evidences. Phyther Res 36:1064–1092. https://doi.org/10.1002/ptr.7368
Wu NN, Zhang Y, Ren J (2019) Mitophagy, mitochondrial dynamics, and homeostasis in cardiovascular aging. Oxid Med Cell Longev 2019:9825061. https://doi.org/10.1155/2019/9825061
Xianchu L, Lan PZ, Qiufang L et al (2016) Naringin protects against lipopolysaccharide-induced cardiac injury in mice. Environ Toxicol Pharmacol 48:1–6. https://doi.org/10.1016/j.etap.2016.09.005
Yarmohammadi F, Barangi S, Aghaee-Bakhtiari SH et al (2023) Melatonin ameliorates arsenic-induced cardiotoxicity through the regulation of the Sirt1/Nrf2 pathway in rats. BioFactors. https://doi.org/10.1002/biof.1934
Yucel C, Demir S, Demir M et al (2015) Left ventricular hypertrophy and arterial stiffness in essential hypertension. Bratisl Lek Listy 116:714–718. https://doi.org/10.4149/bll_2015_140
Zarei M, Sarihi A, Zamani A, et al (2023) Mitochondrial biogenesis and apoptosis as underlying mechanisms involved in the cardioprotective effects of Gallic acid against D-galactose-induced aging. Mol Biol Rep 1–10. https://doi.org/10.1007/s11033-023-08670-4
Zhong W, Rao Z, Rao J et al (2020) Aging aggravated liver ischemia and reperfusion injury by promoting STING-mediated NLRP3 activation in macrophages. Aging Cell 19:e13186. https://doi.org/10.1111/acel.13186
Acknowledgements
The authors are grateful to the Kermanshah University of Medical Sciences, Health Technology Institute, Medical Biology Research Center, Kermanshah, Iran, for financial support.
Funding
This work was supported by Kermanshah University of Medical Sciences, Kermanshah, Iran (IR.KUMS.REC.1397.902).
Author information
Authors and Affiliations
Contributions
D. Sh. contributed to data collection and interpretation, and wrote the manuscript, M. H. performed the experiments, and contributed reagents, S. R-A. Contributed reagents, materials, and analysis tools or data, M. P. conceived and designed the experiments, F. Y. contributed to data collection and analysis, and wrote the manuscript, F. R-A. Designed the study, contributed to data collection and interpretation, and wrote the manuscript. All authors read and approved the final manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethical approval
This study was approved by the Animal Ethics Committee of Kermanshah University of Medical sciences (Ethics Committee Permission No. IR.KUMS.REC.1397.902).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shackebaei, D., Hesari, M., Ramezani-Aliakbari, S. et al. Cardioprotective effect of naringin against the ischemia/reperfusion injury of aged rats. Naunyn-Schmiedeberg's Arch Pharmacol 397, 1209–1218 (2024). https://doi.org/10.1007/s00210-023-02692-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02692-2