Abstract
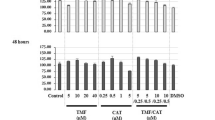
In pulmonary fibrosis, the proliferation of fibroblasts and their differentiation into myofibroblasts is often caused by tissue damage, such as oxidative damage caused by reactive oxygen species, which leads to progressive rupture and thus destruction of the alveolar architecture, resulting in cell proliferation and tissue remodeling. Bezafibrate (BZF) is an important member of the peroxisome proliferator-activated receptor (PPARs) family agonists, used in clinical practice as antihyperlipidemic. However, the antifibrotic effects of BZF are still poorly studied. The objective of this study was to evaluate the effects of BZF on pulmonary oxidative damage in lung fibroblast cells. MRC-5 cells were treated with hydrogen peroxide (H2O2) to induce oxidative stress activation and BZF treatment was administered at the same moment as H2O2 induction. The outcomes evaluated were cell proliferation and cell viability; oxidative stress markers such as reactive oxygen species (ROS), catalase (CAT) levels and thiobarbituric acid reactive substances (TBARS); col-1 and α-SMA mRNA expression and cellular elasticity through Young's modulus analysis evaluated by atomic force microscopy (AFM). The H2O2-induced oxidative damage decreased the cell viability and increased ROS levels and decreased CAT activity in MRC-5 cells. The expression of α-SMA and the cell stiffness increased in response to H2O2 treatment. Treatment with BZF decreased the MRC-5 cell proliferation, ROS levels, reestablished CAT levels, decreased the mRNA expression of type I collagen protein (col-1) and α-smooth muscle actin (α-SMA), and cellular elasticity even with H2O2 induction. Our results suggest that BZF has a potential protective effect on H2O2-induced oxidative stress. These results are based on an in vitro experiment, derived from a fetal lung cell line and may emerge as a possible new therapy for the treatment of pulmonary fibrosis.
Graphical Abstract





Similar content being viewed by others
Data availability
When requested, the authors provided the data and materials used for the development of the manuscript.
Abbreviations
- α-SMA:
-
Alfa Smooth Muscle Actin and Type 1
- AFM:
-
Atomic Force Microscopy
- BZF:
-
Bezafibrate
- CAT:
-
Catalase
- Col-1:
-
Collagen 1
- DCFH-DA:
-
2’,7’-Dichlorodihydrofluorescein Diacetate
- DMEM:
-
Dulbecco´s Modified Eagle´s Medium
- ECM:
-
Extracellular Matrix
- FBS:
-
Fetal Bovine Serum
- H2O2 :
-
Hydrogen Peroxide
- PPARs:
-
Peroxisome Proliferator-Activated Receptor Agonists
- ROS:
-
Reactive Oxygen Species
- TBARS:
-
Thiobarbituric Acid-Reactive Substances
References
Aebi H (1984) Catalase in vitro. In: Methods in enzymology. pp 121–126
Aguilar Diaz De Leon J, Borges CR (2020) Evaluation of oxidative stress in biological samples using the thiobarbituric acid reactive substances assay. J Vis Exp.https://doi.org/10.3791/61122
Avouac J, Konstantinova I, Guignabert C et al (2017) Pan-PPAR agonist IVA337 is effective in experimental lung fibrosis and pulmonary hypertension. Ann Rheum Dis 76:1931–1940. https://doi.org/10.1136/annrheumdis-2016-210821
Awonuga AO, Chatzicharalampous C, Thakur M et al (2022) Genetic and epidemiological similarities, and differences between postoperative intraperitoneal adhesion development and other benign fibro-proliferative disorders. Reprod Sci 29:3055–3077. https://doi.org/10.1007/s43032-021-00726-9
Cameli P, Carleo A, Bergantini L et al (2020) Oxidant/antioxidant disequilibrium in idiopathic pulmonary fibrosis pathogenesis. Inflammation 43:1–7. https://doi.org/10.1007/s10753-019-01059-1
Cannavò L, Perrone S, Viola V et al (2021) Oxidative stress and respiratory diseases in preterm newborns. Int J Mol Sci 22:12504. https://doi.org/10.3390/ijms222212504
Cheresh P, Kim S-J, Tulasiram S, Kamp DW (2013) Oxidative stress and pulmonary fibrosis. Biochim Biophys Acta - Mol Basis Dis 1832:1028–1040. https://doi.org/10.1016/j.bbadis.2012.11.021
Cuenot S, Bouchara J-P (2018) Atomic force microscopy: a promising tool for deciphering the pathogenic mechanisms of fungi in cystic fibrosis. Mycopathologia 183:291–310. https://doi.org/10.1007/s11046-017-0201-1
Ding Y, Xu G-K, Wang G-F (2017) On the determination of elastic moduli of cells by AFM based indentation. Sci Rep 7:45575. https://doi.org/10.1038/srep45575
Dong H, Yang J, Wang Y et al (2020) Polysaccharide SAFP from Sarcodon aspratus attenuates oxidative stress-induced cell damage and bleomycin-induced pulmonary fibrosis. Int J Biol Macromol 164:1215–1236. https://doi.org/10.1016/j.ijbiomac.2020.07.120
Draper HH, Hadley M (1990) Malondialdehyde determination as index of lipid Peroxidation. Methods Enzymol 186:421–431. https://doi.org/10.1016/0076-6879(90)86135-I
Duckworth A, Gibbons MA, Allen RJ et al (2021) Telomere length and risk of idiopathic pulmonary fibrosis and chronic obstructive pulmonary disease: a mendelian randomisation study. Lancet Respir Med 9:285–294. https://doi.org/10.1016/S2213-2600(20)30364-7
Dulínska-Molak I, Mao H, Kawazoe N, Chen G (2014) Variation of mechanical property of single-walled carbon nanotubes-treated cells explored by atomic force microscopy. J Biomed Nanotechnol 10:651–659. https://doi.org/10.1166/jbn.2014.1745
Francis LW, Gonzalez D, Ryder T et al (2010) Optimized sample preparation for high-resolution AFM characterization of fixed human cells. J Microsc 240:111–121. https://doi.org/10.1111/j.1365-2818.2010.03392.x
Glass DS, Grossfeld D, Renna HA et al (2022) Idiopathic pulmonary fibrosis: Current and future treatment. Clin Respir J 16:84–96. https://doi.org/10.1111/crj.13466
Glorieux C, Calderon PB (2017) Catalase, a remarkable enzyme: targeting the oldest antioxidant enzyme to find a new cancer treatment approach. Biol Chem 398:1095–1108. https://doi.org/10.1515/hsz-2017-0131
Guz N, Dokukin M, Kalaparthi V, Sokolov I (2014) If cell mechanics can be described by elastic modulus: study of different models and probes used in indentation experiments. Biophys J 107:564–575. https://doi.org/10.1016/j.bpj.2014.06.033
Han X, Wu Y, Yang Q, Cao G (2021) Peroxisome proliferator-activated receptors in the pathogenesis and therapies of liver fibrosis. Pharmacol Ther 222:107791. https://doi.org/10.1016/j.pharmthera.2020.107791
Hu X, Xu Q, Wan H et al (2020) PI3K-Akt-mTOR/PFKFB3 pathway mediated lung fibroblast aerobic glycolysis and collagen synthesis in lipopolysaccharide-induced pulmonary fibrosis. Lab Investig 100:801–811. https://doi.org/10.1038/s41374-020-0404-9
Ibrahim SA, Mohamed MZ, El-Tahawy NF, Abdelrahman AM (2021) Antifibrotic effects of bezafibrate and pioglitazone against thioacetamide-induced liver fibrosis in albino rats. Can J Physiol Pharmacol 99:313–320. https://doi.org/10.1139/cjpp-2020-0159
Illner D, Scherthan H (2013) Ionizing irradiation-induced radical stress stalls live meiotic chromosome movements by altering the actin cytoskeleton. Proc Natl Acad Sci 110:16027–16032. https://doi.org/10.1073/pnas.1306324110
Júnior C, Narciso M, Marhuenda E et al (2021) Baseline stiffness modulates the non-linear response to stretch of the extracellular matrix in pulmonary fibrosis. Int J Mol Sci 22:12928. https://doi.org/10.3390/ijms222312928
Junn E, Lee KN, Ju HR et al (2000) Requirement of hydrogen peroxide generation in TGF-β1 signal transduction in human lung fibroblast cells: involvement of hydrogen peroxide and Ca 2+ in TGF-β1-induced IL-6 expression. J Immunol 165:2190–2197. https://doi.org/10.4049/jimmunol.165.4.2190
Kim HJ, Perlman D, Tomic R (2015) Natural history of idiopathic pulmonary fibrosis. Respir Med 109:661–670. https://doi.org/10.1016/j.rmed.2015.02.002
King TE, Pardo A, Selman M (2011) Idiopathic pulmonary fibrosis. Lancet 378:1949–1961. https://doi.org/10.1016/S0140-6736(11)60052-4
Kishaba T (2019) Acute exacerbation of idiopathic pulmonary fibrosis. Medicina (B Aires) 55:70. https://doi.org/10.3390/medicina55030070
Kuznetsova TG, Starodubtseva MN, Yegorenkov NI et al (2007) Atomic force microscopy probing of cell elasticity. Micron 38:824–833. https://doi.org/10.1016/j.micron.2007.06.011
Li S, Shi J, Tang H (2022) Animal models of drug-induced pulmonary fibrosis: an overview of molecular mechanisms and characteristics. Cell Biol Toxicol 38:699–723. https://doi.org/10.1007/s10565-021-09676-z
Liu X, Liu H, Zhai Y et al (2017) Laminarin protects against hydrogen peroxide-induced oxidative damage in MRC-5 cells possibly via regulating NRF2. PeerJ 5:e3642. https://doi.org/10.7717/peerj.3642
Lund M, Andersen KG, Heaton R et al (2021) Bezafibrate activation of PPAR drives disturbances in mitochondrial redox bioenergetics and decreases the viability of cells from patients with VLCAD deficiency. Biochim Biophys Acta - Mol Basis Dis 1867:166100. https://doi.org/10.1016/j.bbadis.2021.166100
Lv Q, Wang J, Xu C et al (2020) Pirfenidone alleviates pulmonary fibrosis in vitro and in vivo through regulating Wnt/GSK-3β/β-catenin and TGF-β1/Smad2/3 signaling pathways. Mol Med 26:49. https://doi.org/10.1186/s10020-020-00173-3
Mandras SA, Mehta HS, Vaidya A (2020) Pulmonary hypertension: a brief guide for clinicians. Mayo Clin Proc 95:1978–1988. https://doi.org/10.1016/j.mayocp.2020.04.039
Martinez FJ, Collard HR, Pardo A et al (2017) Idiopathic pulmonary fibrosis. Nat Rev Dis Prim 3:17074. https://doi.org/10.1038/nrdp.2017.74
Piotrowski W, Martusewicz-Boros M, Białas A et al (2022) Guidelines of the polish respiratory society on the diagnosis and treatment of progressive fibrosing interstitial lung diseases other than idiopathic pulmonary fibrosis. Adv Respir Med 90:425–450. https://doi.org/10.3390/arm90050052
Poch D, Mandel J (2021) Pulmonary hypertension. Ann Intern Med 174:ITC49–ITC64. https://doi.org/10.7326/AITC202104200
Que X, Hung M-Y, Yeang C et al (2018) Oxidized phospholipids are proinflammatory and proatherogenic in hypercholesterolaemic mice. Nature 558:301–306. https://doi.org/10.1038/s41586-018-0198-8
Roehlen N, Crouchet E, Baumert TF (2020) Liver fibrosis: mechanistic concepts and therapeutic perspectives. Cells 9:875. https://doi.org/10.3390/cells9040875
Su H, Wan C, Song A, et al (2019a) Oxidative stress and renal fibrosis: mechanisms and therapies. pp 585–604
Su L-J, Zhang J-H, Gomez H et al (2019b) Reactive oxygen species-induced lipid peroxidation in apoptosis, autophagy, and ferroptosis. Oxid Med Cell Longev 2019:1–13. https://doi.org/10.1155/2019/5080843
Sun K-H, Chang Y, Reed NI, Sheppard D (2016) α-Smooth muscle actin is an inconsistent marker of fibroblasts responsible for force-dependent TGFβ activation or collagen production across multiple models of organ fibrosis. Am J Physiol Cell Mol Physiol 310:L824–L836. https://doi.org/10.1152/ajplung.00350.2015
Sunnetcioglu A, Alp HH, Sertogullar ndan B, et al (2016) Evaluation of oxidative damage and antioxidant mechanisms in COPD, Lung Cancer, and Obstructive Sleep Apnea Syndrome. Respir Care 61:205–211. https://doi.org/10.4187/respcare.04209
Tanaka K, Chamoto K, Saeki S et al (2022) Combination bezafibrate and nivolumab treatment of patients with advanced non–small cell lung cancer. Sci Transl Med 14:eabq0021. https://doi.org/10.1126/scitranslmed.abq0021
Xia F, Youcef-Toumi K (2022) Review: advanced atomic force microscopy modes for biomedical research. Biosensors 12:1116. https://doi.org/10.3390/bios12121116
Funding
This study was funded by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES/Brazil; Financial Code 001), through a fellowship for the first author, and by National Council for Scientific and Technological Development (CNPq/Brazil) (Grant#311424/2018–0).
Author information
Authors and Affiliations
Contributions
DASM and JOR conceived and planned this project. CKR performed all experiments. BSB, BPC, GHV and ACS helped in cell viability and oxidative damage experiments. MSB, KGL, HBG, CL and MCRG performed the gene expression experiments. MVFD and KFR helped during the gene expression experiments as well as writing the manuscript. LSM, FMD, BSA and LLX performed the AFM experiments. CKR analyzed the data and wrote the manuscript with review and input from JOR, DASM, and MVFD. All authors approved the final manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethical approval
Not applicable.
Competing of interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Reghelin, C.K., Bastos, M.S., de Souza Basso, B. et al. Bezafibrate reduces the damage, activation and mechanical properties of lung fibroblast cells induced by hydrogen peroxide. Naunyn-Schmiedeberg's Arch Pharmacol 396, 3857–3866 (2023). https://doi.org/10.1007/s00210-023-02595-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02595-2