Abstract
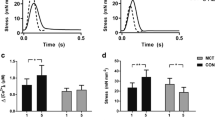
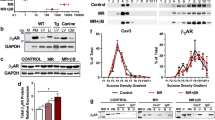
The TASK-1 channel belongs to the two-pore domain potassium channel family. It is expressed in several cells of the heart, including the right atrial (RA) cardiomyocytes and the sinus node, and TASK-1 channel has been implicated in the pathogenesis of atrial arrhythmias (AA). Thus, using the rat model of monocrotaline-induced pulmonary hypertension (MCT-PH), we explored the involvement of TASK-1 in AA. Four-week-old male Wistar rats were injected with 50 mg/kg of MCT to induce MCT-PH and isolated RA function was studied 14 days later. Additionally, isolated RA from six-week-old male Wistar rats were used to explore the ability of ML365, a selective blocker of TASK-1, to modulate RA function. The hearts developed right atrial and ventricular hypertrophy, inflammatory infiltrate and the surface ECG demonstrated increased P wave duration and QT interval, which are markers of MCT-PH. The isolated RA from the MCT animals showed enhanced chronotropism, faster contraction and relaxation kinetics, and a higher sensibility to extracellular acidification. However, the addition of ML365 to extracellular media was not able to restore the phenotype. Using a burst pacing protocol, the RA from MCT animals were more susceptible to develop AA, and simultaneous administration of carbachol and ML365 enhanced AA, suggesting the involvement of TASK-1 in AA induced by MCT. TASK-1 does not play a key role in the chronotropism and inotropism of healthy and diseased RA; however, it may play a role in AA in the MCT-PH model.







Similar content being viewed by others
Data availability
The data presented in this study is available along with the manuscript..
References
Allessie MA, Lammers WJ, Bonke IM, Hollen J (1984) Intra-atrial reentry as a mechanism for atrial flutter induced by acetylcholine and rapid pacing in the dog. Circ 70:123–135. https://doi.org/10.1161/01.cir.70.1.123
Antigny F, Hautefort A, Meloche J, Belacel-Ouari M, Manoury B, Rucker-Martin C, Pechoux C, Potus F, Nadeau V, Tremblay E, Ruffenach G, Bourgeois A, Dorfmuller P, Breuils-Bonnet S, Fadel E, Ranchoux B, Jourdon P, Girerd B, Montani D et al (2016) Potassium channel subfamily K member 3 (KCNK3) contributes to the development of pulmonary arterial hypertension. Circ 133:1371–1385. https://doi.org/10.1161/CIRCULATIONAHA.115.020951
Avula UMR, Abrams J, Katchman A, Zakharov S, Mironov S, Bayne J, Roybal D, Gorti A, Yang L, Iyer V, Waase M, Saluja D, Ciaccio EJ, Garan H, Marks AR, Marx SO, Wan EY (2019) Heterogeneity of the action potential duration is required for sustained atrial fibrillation. JCI insight 5.https://doi.org/10.1172/jci.insight.128765
Bell RM, Mocanu MM, Yellon DM (2011) Retrograde heart perfusion: the Langendorff technique of isolated heart perfusion. J Mol Cell Cardiol 50:940–950. https://doi.org/10.1016/j.yjmcc.2011.02.018
Bohnen MS, Roman-Campos D, Terrenoire C, Jnani J, Sampson KJ, Chung WK, Kass RS (2017) The impact of heterozygous KCNK3 mutations associated with pulmonary arterial hypertension on channel function and pharmacological recovery. J Am Heart Assoc 6.https://doi.org/10.1161/JAHA.117.006465
Chang M, Gada KD, Chidipi B, Tsalatsanis A, Gibbons J, Remily-Wood E, Logothetis DE, Oberstaller J, Noujaim SF (2022) I(KACh) is constitutively active via PKC epsilon in aging mediated atrial fibrillation. iSci 25:105442. https://doi.org/10.1016/j.isci.2022.105442
Cirulis MM, Ryan JJ, Archer SL (2019) Pathophysiology, incidence, management, and consequences of cardiac arrhythmia in pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension. Pulm Circ 9:2045894019834890. https://doi.org/10.1177/2045894019834890
da Silva Goncalves Bos D, CEE VDB, Kurakula K, Sun XQ, Casali KR, Casali AG, Rol N, Szulcek R, Dos Remedios C, Guignabert C, Tu L, Dorfmuller P, Humbert M, PJM W, DWD K, van der Velden J, Goumans MJ, Bogaard HJ, Vonk-Noordegraaf A et al (2018) Contribution of impaired parasympathetic activity to right ventricular dysfunction and pulmonary vascular remodeling in pulmonary arterial hypertension. Circ 137:910–924. https://doi.org/10.1161/CIRCULATIONAHA.117.027451
Dobrev D, Graf E, Wettwer E, Himmel HM, Hala O, Doerfel C, Christ T, Schuler S, Ravens U (2001) Molecular basis of downregulation of G-protein-coupled inward rectifying K(+) current (IK,ACh) in chronic human atrial fibrillation: decrease in GIRK4 mRNA correlates with reduced I(K,ACh) and muscarinic receptor-mediated shortening of action potentials. Circ 104:2551–2557. https://doi.org/10.1161/hc4601.099466
Euler DE, Scanlon PJ (1987) Acetylcholine release by a stimulus train lowers atrial fibrillation threshold. Am J Phys 253:H863–H868. https://doi.org/10.1152/ajpheart.1987.253.4.H863
Ferdous Z, Qureshi MA, Jayaprakash P, Parekh K, John A, Oz M, Raza H, Dobrzynski H, Adrian TE, Howarth FC (2016) Different profile of mRNA expression in sinoatrial node from streptozotocin-induced diabetic rat. PLoS One 11:e0153934. https://doi.org/10.1371/journal.pone.0153934
Fulton RM, Hutchinson EC, Jones AM (1952) Ventricular weight in cardiac hypertrophy. Br Heart J 14:413–420. https://doi.org/10.1136/hrt.14.3.413
Gada K, Plant LD (2019) Two-pore domain potassium channels: emerging targets for novel analgesic drugs: IUPHAR Review 26. Br J Pharmacol 176:256–266. https://doi.org/10.1111/bph.14518
Gende OA, Camilion de Hurtado MC, Cingolani EH (1978) Chronotropic response of isolated atria to acid base alterations. Arch Int Physiol Biochim 86:997–1009. https://doi.org/10.3109/13813457809055959
Ghebleh Zadeh N, Vaezi G, Bakhtiarian A, Mousavi Z, Shiravi A, Nikoui V (2019) The potassium channel blocker, dalfampridine diminishes ouabain-induced arrhythmia in isolated rat atria. Arch Physiol Biochem 125:25–29. https://doi.org/10.1080/13813455.2018.1430158
Handoko ML, de Man FS, Happe CM, Schalij I, Musters RJ, Westerhof N, Postmus PE, Paulus WJ, van der Laarse WJ, Vonk-Noordegraaf A (2009) Opposite effects of training in rats with stable and progressive pulmonary hypertension. Circ 120:42–49. https://doi.org/10.1161/CIRCULATIONAHA.108.829713
Henkens IR, Mouchaers KT, Vliegen HW, van der Laarse WJ, Swenne CA, Maan AC, Draisma HH, Schalij I, van der Wall EE, Schalij MJ, Vonk-Noordegraaf A (2007) Early changes in rat hearts with developing pulmonary arterial hypertension can be detected with three-dimensional electrocardiography. Am J Phys Heart Circ Phys 293:H1300–H1307. https://doi.org/10.1152/ajpheart.01359.2006
Hiram R, Naud P, Xiong F, Al-U'datt D, Algalarrondo V, Sirois MG, Tanguay JF, Tardif JC, Nattel S (2019) Right atrial mechanisms of atrial fibrillation in a rat model of right heart disease. J Am Coll Cardiol 74:1332–1347. https://doi.org/10.1016/j.jacc.2019.06.066
Jones DK, Peters CH, Tolhurst SA, Claydon TW, Ruben PC (2011) Extracellular proton modulation of the cardiac voltage-gated sodium channel, Nav1.5. Biophys J 101:2147–2156. https://doi.org/10.1016/j.bpj.2011.08.056
Jones SA, Morton MJ, Hunter M, Boyett MR (2002) Expression of TASK-1, a pH-sensitive twin-pore domain K(+) channel, in rat myocytes. Am J Physiol Heart Circ Physiol 283:H181–H185. https://doi.org/10.1152/ajpheart.00963.2001
Kawade A, Yamamura A, Fujiwara M, Kobayashi S, Mori S, Horii C, Hiraku A, Suzumura S, Tsukamoto K, Ohara N, Kondo R, Suzuki Y, Yamamura H (2021) Comparative analysis of age in monocrotaline-induced pulmonary hypertensive rats. J Pharmacol Sci 147:81–85. https://doi.org/10.1016/j.jphs.2021.05.012
Kennedy RH, Akera T, Brody TM (1987) The effect of stimulation frequency and calcium concentration on maintenance of developed tension in isolated heart muscle preparations. J Pharm Methods 17:95–110. https://doi.org/10.1016/0160-5402(87)90021-0
Kmecova J, Klimas J (2010) Heart rate correction of the QT duration in rats. Eur J Pharmacol 641:187–192. https://doi.org/10.1016/j.ejphar.2010.05.038
Kornej J, Börschel CS, Benjamin EJ, Schnabel RB (2020) Epidemiology of atrial fibrillation in the 21st century: novel methods and new insights. Circ Res 127:4–20. https://doi.org/10.1161/circresaha.120.316340
Kraft M, Buscher A, Wiedmann F, L'Hoste Y, Haefeli WE, Frey N, Katus HA, Schmidt C (2021) Current drug treatment strategies for atrial fibrillation and TASK-1 inhibition as an emerging novel therapy option. Front Pharmacol 12:638445. https://doi.org/10.3389/fphar.2021.638445
Lalich JJ, Merkow L (1961) Pulmonary arteritis produced in rat by feeding Crotalaria spectabilis. Lab Inv J Tech Meth Pathol 10: 744-750 PubMed
Lambert M, Boet A, Rucker-Martin C, Mendes-Ferreira P, Capuano V, Hatem S, Adao R, Bras-Silva C, Hautefort A, Michel JB, Dorfmuller P, Fadel E, Kotsimbos T, Price L, Jourdon P, Montani D, Humbert M, Perros F, Antigny F (2018) Loss of KCNK3 is a hallmark of RV hypertrophy/dysfunction associated with pulmonary hypertension. Cardiovasc Res 114:880–893. https://doi.org/10.1093/cvr/cvy016
Lambert M, Capuano V, Boet A, Tesson L, Bertero T, Nakhleh MK, Remy S, Anegon I, Pechoux C, Hautefort A, Rucker-Martin C, Manoury B, Domergue V, Mercier O, Girerd B, Montani D, Perros F, Humbert M, Antigny F (2019) Characterization of Kcnk3-mutated rat, a novel model of pulmonary hypertension. Circ Res 125:678–695. https://doi.org/10.1161/CIRCRESAHA.119.314793
Leist M, Rinne S, Datunashvili M, Aissaoui A, Pape HC, Decher N, Meuth SG, Budde T (2017) Acetylcholine-dependent upregulation of TASK-1 channels in thalamic interneurons by a smooth muscle-like signalling pathway. J Physiol 595:5875–5893. https://doi.org/10.1113/JP274527
Liang F, Fan P, Jia J, Yang S, Jiang Z, Karpinski S, Kornyeyev D, Pagratis N, Belardinelli L, Yao L (2016) Inhibitions of late INa and CaMKII act synergistically to prevent ATX-II-induced atrial fibrillation in isolated rat right atria. J Mol Cell Cardiol 94:122–130. https://doi.org/10.1016/j.yjmcc.2016.04.001
Lookin O, Mukhlynina E, Protsenko Y (2022) Contractile behavior of right atrial myocardium of healthy rats and rats with the experimental model of pulmonary hypertension. Int J Mol Sci 23. https://doi.org/10.3390/ijms23084186
Lu YY, Lin FJ, Chen YC, Kao YH, Higa S, Chen SA, Chen YJ (2022) Role of endothelin-1 in right atrial arrhythmogenesis in rabbits with monocrotaline-induced pulmonary arterial hypertension. Int J Mol Sci 23.https://doi.org/10.3390/ijms231910993
Ma L, Roman-Campos D, Austin ED, Eyries M, Sampson KS, Soubrier F, Germain M, Tregouet DA, Borczuk A, Rosenzweig EB, Girerd B, Montani D, Humbert M, Loyd JE, Kass RS, Chung WK (2013) A novel channelopathy in pulmonary arterial hypertension. N Engl J Med 369:351–361. https://doi.org/10.1056/NEJMoa1211097
Mercurio V, Peloquin G, Bourji KI, Diab N, Sato T, Enobun B, Housten-Harris T, Damico R, Kolb TM, Mathai SC, Tedford RJ, Tocchetti CG, Hassoun PM (2018) Pulmonary arterial hypertension and atrial arrhythmias: incidence, risk factors, and clinical impact. Pulm Circ 8:2045894018769874. https://doi.org/10.1177/2045894018769874
Nattel S, Burstein B, Dobrev D (2008) Atrial remodeling and atrial fibrillation: mechanisms and implications. Circ Arrhythm Electrophysiol 1:62–73. https://doi.org/10.1161/circep.107.754564
Orchard CH, Kentish JC (1990) Effects of changes of pH on the contractile function of cardiac muscle. Am J Phys 258:C967–C981. https://doi.org/10.1152/ajpcell.1990.258.6.C967
Putzke C, Wemhoner K, Sachse FB, Rinne S, Schlichthorl G, Li XT, Jae L, Eckhardt I, Wischmeyer E, Wulf H, Preisig-Muller R, Daut J, Decher N (2007) The acid-sensitive potassium channel TASK-1 in rat cardiac muscle. Cardiovasc Res 75:59–68. https://doi.org/10.1016/j.cardiores.2007.02.025
Rodrigues JQD, Camara H, Jurkiewicz A, Godinho RO (2018) Increased Gi protein signaling potentiates the negative chronotropic effect of adenosine in the SHR right atrium. Naunyn Schmiedeberg's Arch Pharmacol 391:513–522. https://doi.org/10.1007/s00210-018-1482-8
Rottlaender D, Motloch LJ, Schmidt D, Reda S, Larbig R, Wolny M, Dumitrescu D, Rosenkranz S, Erdmann E, Hoppe UC (2012) Clinical impact of atrial fibrillation in patients with pulmonary hypertension. PLoS One 7:e33902. https://doi.org/10.1371/journal.pone.0033902
Santos-Miranda A, Joviano-Santos JV, Ribeiro GA, Botelho AFM, Rocha P, Vieira LQ, Cruz JS, Roman-Campos D (2020) Reactive oxygen species and nitric oxide imbalances lead to in vivo and in vitro arrhythmogenic phenotype in acute phase of experimental Chagas disease. PLoS Pathog 16:e1008379. https://doi.org/10.1371/journal.ppat.1008379
Schmidt C, Wiedmann F, Beyersdorf C, Zhao Z, El-Battrawy I, Lan H, Szabo G, Li X, Lang S, Korkmaz-Icoz S, Rapti K, Jungmann A, Ratte A, Muller OJ, Karck M, Seemann G, Akin I, Borggrefe M, Zhou XB et al (2019) Genetic Ablation of TASK-1 (Tandem of P Domains in a Weak Inward Rectifying K(+) Channel-Related Acid-Sensitive K(+) Channel-1) (K2P3.1) K(+) Channels suppresses atrial fibrillation and prevents electrical remodeling. Circ Arrhythmia Electrophys 12:e007465. https://doi.org/10.1161/CIRCEP.119.007465
Schmidt C, Wiedmann F, Voigt N, Zhou XB, Heijman J, Lang S, Albert V, Kallenberger S, Ruhparwar A, Szabo G, Kallenbach K, Karck M, Borggrefe M, Biliczki P, Ehrlich JR, Baczko I, Lugenbiel P, Schweizer PA, Donner BC et al (2015) Upregulation of K(2P)3.1 K+ current causes action potential shortening in patients with chronic atrial fibrillation. Circ 132:82–92. https://doi.org/10.1161/CIRCULATIONAHA.114.012657
Shiina Y, Funabashi N, Lee K, Daimon M, Sekine T, Kawakubo M, Takahashi M, Yajima R, Tanabe N, Kuriyama T, Komuro I (2009) Right atrium contractility and right ventricular diastolic function assessed by pulsed tissue Doppler imaging can predict brain natriuretic peptide in adults with acquired pulmonary hypertension. Int J Cardiol 135:53–59. https://doi.org/10.1016/j.ijcard.2008.03.090
Skarsfeldt MA, Jepps TA, Bomholtz SH, Abildgaard L, Sorensen US, Gregers E, Svendsen JH, Diness JG, Grunnet M, Schmitt N, Olesen SP, Bentzen BH (2016) pH-dependent inhibition of K(2)P3.1 prolongs atrial refractoriness in whole hearts. Pflugers Arch 468:643–654. https://doi.org/10.1007/s00424-015-1779-0
Stemmer P, Akera T (1986) Concealed positive force-frequency relationships in rat and mouse cardiac muscle revealed by ryanodine. Am J Phys 251:H1106–H1110. https://doi.org/10.1152/ajpheart.1986.251.6.H1106
Sutherland FJ, Hearse DJ (2000) The isolated blood and perfusion fluid perfused heart. Pharmacol Res 41:613–627. https://doi.org/10.1006/phrs.1999.0653
Tanaka Y, Takase B, Yao T, Ishihara M (2013) Right ventricular electrical remodeling and arrhythmogenic substrate in rat pulmonary hypertension. Am J Respir Cell Mol Biol 49:426–436. https://doi.org/10.1165/rcmb.2012-0089OC
Teixeira-Fonseca JL, Santos-Miranda A, da Silva JB, Marques LP, Joviano-Santos JV, Nunes PIC, Roman-Campos D, Gondim ANS (2021) Eugenol interacts with cardiac sodium channel and reduces heart excitability and arrhythmias. Life Sci 282:119761. https://doi.org/10.1016/j.lfs.2021.119761
Verheule S, Schotten U (2021) Electrophysiological consequences of cardiac fibrosis. Cells 10. https://doi.org/10.3390/cells10113220.
Wiedmann F, Beyersdorf C, Zhou XB, Kraft M, Foerster KI, El-Battrawy I, Lang S, Borggrefe M, Haefeli WE, Frey N, Schmidt C (2020) The experimental TASK-1 potassium channel inhibitor A293 can be employed for rhythm control of persistent atrial fibrillation in a translational large animal model. Front Physiol 11:629421. https://doi.org/10.3389/fphys.2020.629421
Wiedmann F, Beyersdorf C, Zhou XB, Kraft M, Paasche A, Javorszky N, Rinne S, Sutanto H, Buscher A, Foerster KI, Blank A, El-Battrawy I, Li X, Lang S, Tochtermann U, Kremer J, Arif R, Karck M, Decher N et al (2022a) Treatment of atrial fibrillation with doxapram: TASK-1 potassium channel inhibition as a novel pharmacological strategy. Cardiovasc Res 118:1728–1741. https://doi.org/10.1093/cvr/cvab177
Wiedmann F, Kraft M, Kallenberger S, Buscher A, Paasche A, Blochberger PL, Seeger T, Javorszky N, Warnecke G, Arif R, Kremer J, Karck M, Frey N, Schmidt C (2022b) MicroRNAs regulate TASK-1 and are linked to myocardial dilatation in atrial fibrillation. J Am Heart Assoc 11:e023472. https://doi.org/10.1161/JAHA.121.023472
Wilson DW, Segall HJ, Pan LC, Lame MW, Estep JE, Morin D (1992) Mechanisms and pathology of monocrotaline pulmonary toxicity. Crit Rev Toxicol 22:307–325. https://doi.org/10.3109/10408449209146311
Yamada Y, Okumura K, Hashimoto H, Ito T, Satake T (1991) Altered myocardial acetylcholine and norepinephrine concentrations in right ventricular hypertrophy and failure. Heart Vessel 6:150–157 PubMed
Yang CF, Chen MYC, Chen TI, Cheng CF (2014) Dose-dependent effects of isoflurane on cardiovascular function in rats. Tzu Chi Med J 26(3):119–122. https://doi.org/10.1016/j.tcmj.2014.07.005
Zafalon N Jr, Bassani JW, Bassani RA (2004) Cholinergic-adrenergic antagonism in the induction of tachyarrhythmia by electrical stimulation in isolated rat atria. J Mol Cell Cardiol 37:127–135. https://doi.org/10.1016/j.yjmcc.2004.04.020
Zhang WH, Qiu MH, Wang XJ, Sun K, Zheng Y, Jing ZC (2014) Up-regulation of hexokinase1 in the right ventricle of monocrotaline induced pulmonary hypertension. Respir Res 15:119. https://doi.org/10.1186/s12931-014-0119-9
Zopf DA, das Neves LA, Nikula KJ, Huang J, Senese PB, Gralinski MR (2011) C-122, a novel antagonist of serotonin receptor 5-HT2B, prevents monocrotaline-induced pulmonary arterial hypertension in rats. Eur J Pharmacol 670:195–203. https://doi.org/10.1016/j.ejphar.2011.08.015
Zou B, Flaherty DP, Simpson DS, Maki BE, Miller MR, Shi J, Wu M, McManus OB, Golden JE, Aube J, Li M (2010) ML365: development of bis-amides as selective inhibitors of the KCNK3/TASK1 two pore potassium channel. Probe Reports from the NIH Molecular Libraries Program, Bethesda (MD)
Funding
This work was supported by the São Paulo Research Foundation (FAPESP) (through grants # 2019/21304-4, 2021/05584-7 and 2020/14635-1 to DRC, grant #2019/18918-0 to DSS, and grant #2021/15122-0 to LPM) and the National Council for Scientific and Technological Development [CNPq] # 304257/2020-6.
Author information
Authors and Affiliations
Contributions
Jorge Lucas Teixeira-Fonseca and Danilo Roman-Campos contributed to the study conception and design. Material preparation, data collection, and analysis were performed by Jorge Lucas Teixeira-Fonseca, Julliane V. Joviano-Santos, Samuel Santos Bezerra, Michael Ramon de Lima Conceição, Polyana Leal-Silva, Leisiane Pereira Marques, and Diego Santos Souza. The first draft of the manuscript was written by Jorge Lucas Teixeira-Fonseca and Danilo Roman-Campos and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethics statement
This study was performed in line with the principles of the Declaration of Helsinki. The ARRIVE guidelines were strictly followed during all procedures to ensure that animals used in research are given the most suitable possible care and that all procedures are conducted humanely and ethically.
Ethics approval
All animal handling procedures were approved by the Ethics Committee for Animal Use (CEUA) of the Federal University of São Paulo (number 9073161118) and the animals were obtained from the Center for the Development of Experimental Models for Biology and Medicine (CEDEME) from Federal University of São Paulo.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
ESM 1
(DOCX 0.97 mb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Teixeira-Fonseca, J.L., Joviano-Santos, J.V., Beserra, S.S. et al. Exploring the involvement of TASK-1 in the control of isolated rat right atrium function from healthy animals and an experimental model of monocrotaline-induced pulmonary hypertension. Naunyn-Schmiedeberg's Arch Pharmacol 396, 3775–3788 (2023). https://doi.org/10.1007/s00210-023-02569-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02569-4