Abstract
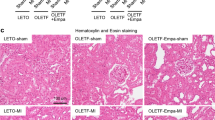
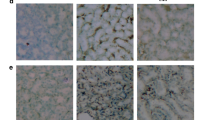
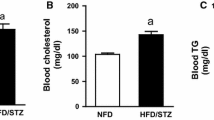
Roflumilast is a potent selective inhibitor of the phosphodiesterase-4 enzyme which greatly manifest an anti-inflammatory activity in chronic obstructive pulmonary patients. Inflammation is a prominent factor in the prevalence of diabetic nephropathy, one of the most prevalent microvascular complications of Diabetes Mellitus. The present study was undertaken to assess the potential role of roflumilast in diabetic nephropathy. The model was developed by feeding a high-fat diet for four weeks and following streptozotocin (30 mg/kg) injection intraperitoneally. The rats with > 13.8 mmol/L blood glucose were treated with roflumilast (0.25, 0.5, 1 mg/kg) and standard metformin (100 mg/kg) orally once a day for eight weeks. Roflumilast (1 mg/kg) remarkably improved renal damage, indicated by an increase in 16% albumin, a decrease in 5% serum creatinine, 12% BUN, 19% HbA1c, and 34% blood glucose. It also significantly improves the oxidative stress levels, which was indicated by a decrease in 18% MDA level and an increase in GSH, SOD, and catalase by 6%, 4%, and 5%, respectively. In addition, Roflumilast (1 mg/kg) decreased the HOMA-IR index by 28% and increased the pancreatic β-cells functioning by 30%. Moreover, significant improvement in histopathological abnormalities were observed in roflumilast treatment groups. Roflumilast treatment was shown to down-regulate the gene expressions of TNF-α (2.1-fold), NF-kB (2.3-fold), MCP-1 (2.5-fold), fibronectin (2.7-fold), collagen IV (2.7-fold), STAT 1(1.06-fold), and STAT 3 (1.20-fold) and upregulated the expression of the Nrf2 (1.43-fold) gene. Roflumilast manifested a potential role in diabetic nephropathy as a renoprotective agent. Roflumilast effectively down-regulates the JAK/STAT pathway and restores renal functions.






Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- AGEs:
-
Advanced glycation End Products
- AMP:
-
Adenosine Monophosphate
- BUN:
-
Blood Urea Nitrogen
- BW:
-
Body Weight
- cAMP:
-
Cyclic Adenosine Monophosphate
- cDNA:
-
Complementary DNA
- CKD:
-
Chronic Kidney Disease
- CMC:
-
Carboxymethyl Cellulose
- COPD:
-
Chronic Obstructive Pulmonary Disease
- CTGF:
-
Connective Tissue Growth Factor
- DM:
-
Diabetes Mellitus
- DN:
-
Diabetic Nephropathy
- ECM:
-
Extracellular Matrix
- FBS:
-
Fasting Blood Sugar
- FINS:
-
Fasting Insulin
- GSH:
-
Reduced Glutathione
- GLUT 2:
-
Glucose transporter 2
- HbA1c:
-
Glycated Hemoglobin
- HFD:
-
High Fat Diet
- IL-6:
-
Interleukin 6
- IR:
-
Insulin Resistance
- JAK/STAT:
-
Janus kinase/signal transducers and activators of transcription
- KWI:
-
Kidney Weight Index
- MCP-1:
-
Monocyte Chemoattractant Protein-1
- MDA:
-
Malondialdehyde
- PDE:
-
Phosphodiesterase
- PKC:
-
Protein Kinase C
- ROS:
-
Reactive Oxygen Species
- SOD:
-
Superoxide Dismutase
- STZ:
-
Streptozotocin
- T2DM:
-
Type II Diabetes Mellitus
- TC:
-
Total Cholesterol
- TGF-β:
-
Transforming Growth Factor β
- TNF-α:
-
Tumor Necrosis Factor α
References
Abdel-Rahman EM, Alhamad T, Reeves WB, Awad AS (2012) Management of diabetic nephropathy in the elderly: Special considerations. J Nephrol Ther 2:1–17. https://doi.org/10.4172/2161-0959.1000124
Aebi H (1984) [13] Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Aghadavoud E, Nasri H, Amiri M (2017) Molecular signaling pathways of diabetic kidney disease; new concepts. J Prev Epidemiol 2:e09–e09. https://doi.org/10.15171/JPE.2017.03
Al-Attar AM, Alsalmi FA (2019) Influence of olive leaves extract on hepatorenal injury in streptozotocin diabetic rats. Saudi J Biol Sci 26:1865–1874. https://doi.org/10.1016/J.SJBS.2017.02.005
Alicic RZ, Rooney MT, Tuttle KR (2017) Diabetic kidney disease: Challenges, progress, and possibilities. Clin J Am Soc Nephrol 12:2032–2045. https://doi.org/10.2215/CJN.11491116
Brown PC (2010) Center for drug evaluation and research application number: 022522Orig1s000 Pharmacology Review(s) 1 Tertiary Pharmacology Review. 1-63. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2011/022522Orig1s000PharmR.pdf
Chaudhary S, Semwal A, Kumar H et al (2016) In-vivo study for anti-hyperglycemic potential of aqueous extract of Basil seeds (Ocimum basilicum Linn) and its influence on biochemical parameters, serum electrolytes and haematological indices. Biomed Pharmacother 84:2008–2013. https://doi.org/10.1016/J.BIOPHA.2016.11.020
Chen D, Liu Y, Chen J et al (2021) JAK/STAT pathway promotes the progression of diabetic kidney disease via autophagy in podocytes. Eur J Pharmacol 902:174121. https://doi.org/10.1016/J.EJPHAR.2021.174121
Crawford K (2017) Review of 2017 Diabetes Standards of Care. Nurs Clin North Am 52:621–663. https://doi.org/10.1016/j.cnur.2017.07.010
Donate-Correa J, Luis-Rodríguez D, Martín-Núñez E et al (2020) Inflammatory targets in diabetic nephropathy. J Clin Med 9:458. https://doi.org/10.3390/JCM9020458
Donate-Correa J, Martín-Núñez E, Muros-De-Fuentes M et al (2015) Inflammatory cytokines in diabetic nephropathy. J Diabetes Res 2015. https://doi.org/10.1155/2015/948417
Duran-Salgado MB, Rubio-Guerra AF (2014) Diabetic nephropathy and inflammation. World J Diabetes 5:393. https://doi.org/10.4239/WJD.V5.I3.393
Forbes JM, Thallas V, Thomas MC et al (2003) The breakdown of pre-existing advanced glycation end products is associated with reduced renal fibrosis in experimental diabetes. FASEB J 17:1762–1764. https://doi.org/10.1096/FJ.02-1102FJE
Gheibi S, Kashfi K, Ghasemi A (2017) A practical guide for induction of type-2 diabetes in rat: Incorporating a high-fat diet and streptozotocin. Biomed Pharmacother 95:605–613. https://doi.org/10.1016/J.BIOPHA.2017.08.098
Goboza M, Aboua YG, Chegou N, Oguntibeju OO (2019) Vindoline effectively ameliorated diabetes-induced hepatotoxicity by docking oxidative stress, inflammation and hypertriglyceridemia in type 2 diabetes-induced male Wistar rats. Biomed Pharmacother 112:108638. https://doi.org/10.1016/J.BIOPHA.2019.108638
Hills CE, Squires PE (2011) The role of TGF-β and epithelial-to mesenchymal transition in diabetic nephropathy. Cytokine Growth Factor Rev 22:131–139. https://doi.org/10.1016/J.CYTOGFR.2011.06.002
Kandhare AD, Mukherjee A, Bodhankar SL (2017) Antioxidant for treatment of diabetic nephropathy: A systematic review and meta-analysis. Chem Biol Interact 278:212–221. https://doi.org/10.1016/J.CBI.2017.10.031
Khanra R, Bhattacharjee N, Dua TK et al (2017) Taraxerol, a pentacyclic triterpenoid, from Abroma augusta leaf attenuates diabetic nephropathy in type 2 diabetic rats. Biomed Pharmacother 94:726–741. https://doi.org/10.1016/J.BIOPHA.2017.07.112
Kotha P, Marella S, Allagadda R et al (2019) Evaluation of biochemical mechanisms of anti-diabetic functions of Anisomeles malabarica. Biomed Pharmacother 112:108598. https://doi.org/10.1016/J.BIOPHA.2019.01.059
Lenzen S (2008) The mechanisms of alloxan- and streptozotocin-induced diabetes. Diabetologia 51:216–226. https://doi.org/10.1007/S00125-007-0886-7
Libetta C, Sepe V, Esposito P et al (2011) Oxidative stress and inflammation: Implications in uremia and hemodialysis. Clin Biochem 44:1189–1198. https://doi.org/10.1016/J.CLINBIOCHEM.2011.06.988
Lim AK, Lim A (2014) Diabetic nephropathy – Complications and treatment. Int J Nephrol Renovasc Dis 7:361–381. https://doi.org/10.2147/IJNRD.S40172
Livak KJ, Schmittgen TD (2001) Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 25:402–408. https://doi.org/10.1006/METH.2001.1262
Lu Q, Wang W, Zhang M et al (2019) ROS induces epithelial-mesenchymal transition via the TGF-β1/PI3K/Akt/mTOR pathway in diabetic nephropathy. Exp Ther Med 17:835–846. https://doi.org/10.3892/ETM.2018.7014
Makinde EA, Radenahmad N, Adekoya AE, Olatunji OJ (2020) Tiliacora triandra extract possesses antidiabetic effects in high fat diet/streptozotocin-induced diabetes in rats. J Food Biochem 44:e13239. https://doi.org/10.1111/JFBC.13239
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47:469–474. https://doi.org/10.1111/J.1432-1033.1974.TB03714.X
Matera MG, Rogliani P, Calzetta L, Cazzola M (2014) Phosphodiesterase inhibitors for chronic obstructive pulmonary disease: What does the future hold? Drugs 74:1983–1992. https://doi.org/10.1007/S40265-014-0303-8/METRICS
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95:351–358. https://doi.org/10.1016/0003-2697(79)90738-3
Rosario RF, Prabhakar S (2006) Lipids and diabetic nephropathy. Curr Diab Rep 6:455–462. https://doi.org/10.1007/S11892-006-0079-7/METRICS
Satirapoj B, Dispan R, Radinahamed P, Kitiyakara C (2018) Urinary epidermal growth factor, monocyte chemoattractant protein-1 or their ratio as predictors for rapid loss of renal function in type 2 diabetic patients with diabetic kidney disease. BMC Nephrol 19:1–10. https://doi.org/10.1186/S12882-018-1043-X/FIGURES/4
Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25:192–205. https://doi.org/10.1016/0003-2697(68)90092-4
Sherwani SI, Khan HA, Ekhzaimy A et al (2016) Significance of HbA1c Test in Diagnosis and Prognosis of Diabetic Patients. Biomark Insights 11:95–104. https://doi.org/10.4137/BMI.S38440
Tikoo K, Lodea S, Karpe PA, Kumar S (2014) Calorie restriction mimicking effects of roflumilast prevents diabetic nephropathy. Biochem Biophys Res Commun 450:1581–1586. https://doi.org/10.1016/J.BBRC.2014.07.039
Umanath K, Lewis JB (2018) Update on Diabetic Nephropathy: Core Curriculum 2018. Am J Kidney Dis 71:884–895. https://doi.org/10.1053/J.AJKD.2017.10.026
Vollert S, Kaessner N, Heuser A et al (2012) The glucose-lowering effects of the PDE4 inhibitors roflumilast and roflumilast-N-oxide in db/db mice. Diabetologia 55:2779–2788. https://doi.org/10.1007/S00125-012-2632-Z/FIGURES/7
Wedzicha JA, Calverley PM, Rabe KF (2016) Roflumilast: A review of its use in the treatment of COPD. Int J COPD 11:81–90. https://doi.org/10.2147/COPD.S89849
Wouters EFM, Bredenbröker D, Teichmann P et al (2012) Effect of the Phosphodiesterase 4 Inhibitor Roflumilast on Glucose Metabolism in Patients with Treatment-Naive, Newly Diagnosed Type 2 Diabetes Mellitus. J Clin Endocrinol Metab 97:E1720–E1725. https://doi.org/10.1210/JC.2011-2886
Zhang S, Xu H, Yu X et al (2017) Metformin ameliorates diabetic nephropathy in a rat model of low-dose streptozotocin-induced diabetes. Exp Ther Med 14:383–390. https://doi.org/10.3892/ETM.2017.4475
Zhong Y, Wu Y, Liu R et al (2012) Roflumilast enhances the renal protective effects of retinoids in an HIV-1 transgenic mouse model of rapidly progressive renal failure. Kidney Int 81:856–864. https://doi.org/10.1038/KI.2011.467
Acknowledgements
The authors are thankful to Ramanbhai Patel College of Pharmacy, CHARUSAT, for providing the necessary infrastructure to carry out the research. We would also like to thank the knowledge consortium of Gujarat (KCG), India, for providing scholarship and contingency.
Funding
The authors are thankful to Ramanbhai Patel College of Pharmacy, CHARUSAT, for providing the necessary infrastructure to carry out the research. We would also like to thank the knowledge consortium of Gujarat (KCG), India, for providing scholarship and contingency. We are thankful to Ms. Pooja Gori for providing access to GraphPad Prism software.
Author information
Authors and Affiliations
Contributions
Sandip Patel, Manan Raval, and Priyal Patel conceptualized and designed the study. Material preparation, data collection, and analysis were performed by Priyal Patel and Shailesh Soni. Sandip Patel, Manan Raval, and Piyush Chudasama validated the data. The first draft of the manuscript was written by Priyal Patel, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Ethics approval
The animal study was approved by the Institutional Animal Ethics Committee at Ramanbhai Patel College of Pharmacy, CHARUSAT. (Registration number: 940/PO/Re/S/06/CPCSEA, Protocol Number: RPCP/IAEC/2021–2022/R01).
Consent to participate
Not Applicable.
Consent to publish
Not Applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Patel, P., Patel, S., Chudasama, P. et al. Roflumilast ameliorates diabetic nephropathy in rats through down-regulation of JAK/STAT signaling pathway. Naunyn-Schmiedeberg's Arch Pharmacol 396, 3285–3297 (2023). https://doi.org/10.1007/s00210-023-02535-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02535-0