Abstract
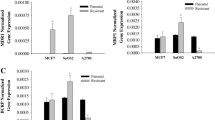
The effects of insulin on the doxorubicin (Dox) sensitivity of breast cancer cell line MCF-7 and its Dox-resistant counterpart MCF-7/Dox were studied and glucose metabolism, content of essential minerals, and the expression of several microRNAs in these cells upon exposure to insulin and Dox were compared. Cell viability colorimetric assay, colorimetric enzymatic technique, flow cytometry, immunocytochemical techniques, inductively-coupled plasma atomic emission spectroscopy, and quantitative polymerase chain reaction were used in the study. We found that insulin in high concentration significantly suppressed Dox toxicity, especially in parental MCF-7 cell line. The increase in proliferative activity triggered by insulin in MCF-7 but not MCF-7/Dox cells occurred in the setting of the increased level of specific binding sites for insulin and increased glucose uptake. Insulin treatment of MCF-7 cells in low and high concentrations resulted in the increase of Mg, Ca, and Zn content while in DOX-resistant cells, only Mg content increased upon exposure to insulin. High concentration of insulin increased the expression of kinase Akt1, P-glycoprotein 1 (P-gp1) and DNA excision repair protein ERCC-1 in MCF-7 cells, while in MCF-7/Dox cells, Akt1 expression decreased, and cytoplasmic expression of P-gp1 increased. In addition, insulin treatment affected expression of miR-122-5p, miR-133a-3p, miR-200b-3p, and miR-320a-3p. The decreased manifestation of biological effects of insulin in Dox-resistant cells could be partly explained by the different patterns of energy metabolism in MCF-7 cells and their Dox-resistant counterpart.






Similar content being viewed by others
Data availability
Data will be made available on reasonable request.
Abbreviations
- BC:
-
Breast cancer
- Dox:
-
Doxorubicin
- ER:
-
Estrogen receptor
- ERCC-1:
-
DNA excision repair protein
- FITC:
-
Fluorescein isothiocyanate
- IGF-1:
-
Insulin-like growth factor 1
- miR:
-
microRNA
- P-gp1:
-
P-glycoprotein 1
- RT-qPCR:
-
Reverse transcription quantitative real-time polymerase chain reaction
References
Agrawal S, Łuc M, Ziółkowski P et al (2017) Insulin-induced enhancement of MCF-7 breast cancer cell response to 5-fluorouracil and cyclophosphamide. Tumour Biol 39(6):1010428317702901. https://doi.org/10.1177/1010428317702901
Aljada A, Saleh AM, Al Suwaidan S (2014) Modulation of insulin/IGFs pathways by sirtuin-7 inhibition in drug-induced chemoresistance. Diagn Pathol 9(1):94. https://doi.org/10.1186/1746-1596-9-94
Bar-On O, Shapira M, Hershko DD (2007) Differential effects of doxorubicin treatment on cell cycle arrest and Skp2 expression in breast cancer cells. Anticancer Drugs 18(10):1113–1121. https://doi.org/10.1097/CAD.0b013e3282ef4571
Berry DA, Cirrincione C, Henderson IC et al (2006) Estrogen-receptor status and outcomes of modern chemotherapy for patients with node-positive breast cancer. JAMA 295(14):1658–1667. https://doi.org/10.1001/jama.295.14.1658
Borst P, Evers R, Kool M et al (2000) A family of drug transporters: the multidrug resistance-associated proteins. J Natl Cancer Inst 92(16):1295–1302. https://doi.org/10.1093/jnci/92.16.1295
Boura-Halfon S, Zick Y (2009) Phosphorylation of IRS proteins, insulin action, and insulin resistance. Am J Physiol Endocrinol Metab 296(4):E581–E591. https://doi.org/10.1152/ajpendo.90437.2008
Cao M, Isaac R, Yan W et al (2022) Cancer-cell-secreted extracellular vesicles suppress insulin secretion through miR-122 to impair systemic glucose homeostasis and contribute to tumour growth. Nat Cell Biol 24(6):954–967. https://doi.org/10.1038/s41556-022-00919-7
Chakraborty C, Doss CGP, Bandyopadhyay S et al (2014) Influence of miRNA in insulin signaling pathway and insulin resistance: micro-molecules with a major role in type-2 diabetes. Wiley Interdiscip Rev RNA 5(5):697–712. https://doi.org/10.1002/wrna.1240
Chekhun VF, Borikun TV, Lukianova NY (2016) Effect of 5-azacytidine on miRNA expression in human breast cancer cells with different sensitivity to cytostatics. Exp Oncol 38(1):26–30. https://doi.org/10.31768/2312-8852.2016.38
Chen S, Tu Y, Yuan H et al (2022) Regulatory functions of miR-200b-3p in tumor development (Review). Oncol Rep 47(5):96. https://doi.org/10.3892/or.2022.8307
Ciftci K, Su J, Trovitch PB (2003) Growth factors and chemotherapeutic modulation of breast cancer cells. J Pharm Pharmacol 55(8):1135–1141. https://doi.org/10.1211/002235703322277177
Cui W, Zhang S, Shan C et al (2013) MicroRNA-133a regulates the cell cycle and proliferation of breast cancer cells by targeting epidermal growth factor receptor through the EGFR/Akt signaling pathway. FEBS J 280(16):3962–3974. https://doi.org/10.1111/febs.12398
Detre S, Saclani Jotti G, Dowsett M (1995) A “quickscore” method for immunohistochemical semiquantitation: validation for oestrogen receptor in breast carcinomas. J Clin Pathol 48(9):876–878. https://doi.org/10.1136/jcp.48.9.876
Due C, Linnet K, Langeland Johansen N et al (1985) Analysis of insulin receptors on heterogeneous eukaryotic cell populations with fluorochrome-conjugated insulin and fluorescence-activated cell sorter. Advantages and limitations to the 125I-labelled insulin methodology. Diabetologia 28(10):749–755. https://doi.org/10.1007/bf00265023
Eketunde AO (2020) Diabetes as a risk factor for breast cancer. Cureus 12(5):e8010. https://doi.org/10.7759/cureus.8010
Fedorenko ZP, Goulak LO, YeL G et al (2022) Cancer in Ukraine 2020–2021. Incidence, mortality, prevalence and other relevant statistics. Bulletin of the National Cancer Registry of Ukraine 23 Available at: http://www.ncru.inf.ua/publications/BULL_23/index_e.htm
Feoktistova M, Geserick P, Leverkus M (2016) Crystal Violet assay for determining viability of cultured cells. Cold Spring Harb Protoc 2016(4):db.prot087379. https://doi.org/10.1101/pdb.prot087379
Fong MY, Zhou W, Liu L et al (2015) Breast-cancer-secreted miR-122 reprograms glucose metabolism in premetastatic niche to promote metastasis. Nat Cell Biol 17(2):183–194. https://doi.org/10.1038/ncb3094
Furberg AS, Veierød MB, Wilsgaard T et al (2004) Serum high-density lipoprotein cholesterol, metabolic profile, and breast cancer risk. J Natl Cancer Inst 96(15):1152–1160. https://doi.org/10.1093/jnci/djh216
Gallagher EJ, LeRoith D (2013) Epidemiology and molecular mechanisms tying obesity, diabetes, and the metabolic syndrome with cancer. Diabetes Care 36(Suppl 2):S233–S239. https://doi.org/10.2337/dcs13-2001
Geier A, Beery R, Haimsohn M et al (1995) Insulin-like growth factor-1 inhibits cell death induced by anticancer drugs in the MCF-7 cells: involvement of growth factors in drug resistance. Cancer Invest 13(5):480–486. https://doi.org/10.3109/07357909509024911
Górecki I, Rak B (2021) The role of microRNAs in epithelial to mesenchymal transition and cancers; focusing on mir-200 family. Cancer Treat Res Commun 28(100385):100385. https://doi.org/10.1016/j.ctarc.2021.100385
Gupta C, Tikoo K (2013) High glucose and insulin differentially modulates proliferation in MCF-7 and MDA-MB-231 cells. J Mol Endocrinol 51(1):119–129. https://doi.org/10.1530/jme-13-0062
Kciuk M, Gielecińska A, Mujwar S et al (2023) Doxorubicin–an agent with multiple mechanisms of anticancer activity. Cells 12(4):659. https://doi.org/10.3390/cells12040659
Liang Y, Li S, Tang L (2021) MicroRNA 320, an anti-oncogene target miRNA for cancer therapy. Biomedicines 9(6):591. https://doi.org/10.3390/biomedicines9060591
Lipscombe LL, Goodwin PJ, Zinman B et al (2006) Diabetes mellitus and breast cancer: a retrospective population-based cohort study. Breast Cancer Res Treat 98(3):349–356. https://doi.org/10.1007/s10549-006-9172-5
Lozovska YV, Andrusishina IM, Lukianova NY et al (2019) The influence of lactoferrin on elemental homeostasis and activity of metal-containing enzymes in rats with Walker-256 carcinosarcoma. Exp Oncol 41(1):20–25. https://doi.org/10.32471/exp-oncology.2312-8852.vol-41-no-1.12471
Lukyanova NY, Rusetskya NV, Tregubova NA et al (2009) Molecular profile and cell cycle in MCF-7 cells resistant to cisplatin and doxorubicin. Exp Oncol 31(2):87–91
Mirzaei S, Gholami MH, Hashemi F et al (2022) Advances in understanding the role of P-gp in doxorubicin resistance: molecular pathways, therapeutic strategies, and prospects. Drug Discov Today 27(2):436–455. https://doi.org/10.1016/j.drudis.2021.09.020
Morakinyo AO, Samuel TA, Adekunbi DA (2018) Magnesium upregulates insulin receptor and glucose transporter-4 in streptozotocin-nicotinamide-induced type-2 diabetic rats. Endocr Regul 52(1):6–16. https://doi.org/10.2478/enr-2018-0002
Pei ZJ, Zhang ZG, Hu AX et al (2017) miR-122-5p inhibits tumor cell proliferation and induces apoptosis by targeting MYC in gastric cancer cells. Pharmazie 72(6):344–347. https://doi.org/10.1691/ph.2017.6404
Pisano A, Santolla MF, De Francesco EM et al (2017) GPER, IGF-IR, and EGFR transduction signaling are involved in stimulatory effects of zinc in breast cancer cells and cancer-associated fibroblasts. Mol Carcinog 56(2):580–593. https://doi.org/10.1002/mc.22518
Pollak M (2008) Insulin and insulin-like growth factor signalling in neoplasia. Nat Rev Cancer 8(12):915–928. https://doi.org/10.1038/nrc2536
Pritchard JE, Dillon PM, Conaway MR et al (2012) A mechanistic study of the effect of doxorubicin/adriamycin on the estrogen response in a breast cancer model. Oncology 83(6):305–320. https://doi.org/10.1159/000341394
Rencelj A, Gvozdenovic N, Cemazar M (2021) MitomiRs: their roles in mitochondria and importance in cancer cell metabolism. Radiol Oncol 55(4):379–392. https://doi.org/10.2478/raon-2021-0042
Romaniuk А, Lyndin M, Sikora V et al (2017) Heavy metals effect on breast cancer progression. J Occup Med Toxicol 12:32. https://doi.org/10.1186/s12995-017-0178-1
Semple RK (2016) EJE PRIZE 2015: how does insulin resistance arise, and how does it cause disease? Human genetic lessons. Eur J Endocrinol 174(5):R209–R223. https://doi.org/10.1530/eje-15-1131
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71(3):209–249. https://doi.org/10.3322/caac.21660
Trapani V, Luongo F, Arduini D et al (2016) Magnesium modulates doxorubicin activity through drug lysosomal sequestration and trafficking. Chem Res Toxicol 3:317–322. https://doi.org/10.1021/acs.chemrestox.5b00478
Wani B, Aziz SA, Ganaie MA, Mir MH et al (2017) Metabolic syndrome and breast cancer risk. Indian J Med Paediatr Oncol 38(04):434–439. https://doi.org/10.4103/ijmpo.ijmpo_168_16
Wei ML, Duan P, Wang ZM et al (2017) High glucose and high insulin conditions promote MCF-7 cell proliferation and invasion by upregulating IRS1 and activating the Ras/Raf/ERK pathway. Mol Med Rep 16(5):6690–6696. https://doi.org/10.3892/mmr.2017.7420
Wolf FI, Cittadini ARM, Maier JAM (2009) Magnesium and tumors: ally or foe? Cancer Treat Rev 35(4):378–382. https://doi.org/10.1016/j.ctrv.2009.01.003
Xiao Y, Wang J, Chen Y et al (2014) Up-regulation of miR-200b in biliary atresia patients accelerates proliferation and migration of hepatic stallate cells by activating PI3K/Akt signaling. Cell Signal 26(5):925–932. https://doi.org/10.1016/j.cellsig.2014.01.003
Yang H, Yu J, Wang L et al (2014) miR-320a is an independent prognostic biomarker for invasive breast cancer. Oncol Lett 8(3):1043–1050. https://doi.org/10.3892/ol.2014.2298
Yang L, Li Y, Wang X et al (2017) MicroRNA-320a inhibition decreases insulin-induced KGN cell proliferation and apoptosis by targeting PCGF1. Mol Med Rep 16(4):5706–5712. https://doi.org/10.3892/mmr.2017.7270
Ye J (2021) Mechanism of insulin resistance in obesity: a role of ATP. Front Med 15(3):372–382. https://doi.org/10.1007/s11684-021-0862-5
Ye J, Xu M, Tian X et al (2019) Research advances in the detection of miRNA. J Pharm Anal 9(4):217–226. https://doi.org/10.1016/j.jpha.2019.05.004
Yuan Y, Yao YF, Hu SN et al (2015) MiR-133a is functionally involved in doxorubicin-resistance in breast cancer cells MCF-7 via its regulation of the expression of uncoupling protein 2. PLoS One 10(6):e0129843. https://doi.org/10.1371/journal.pone.0129843
Zhang W, Jiang H, Chen Y et al (2019) Resveratrol chemosensitizes adriamycin-resistant breast cancer cells by modulating miR-122-5p. J Cell Biochem 120(9):16283–16292. https://doi.org/10.1002/jcb.28910
Acknowledgements
The authors would like to thank Dr. N.N. Khranovskaya for assisting in flow cytometry work.
Funding
The work was supported by scientific grant No. 0117U002034 “Molecular and Biological Factors of Malignant Cells Heterogeneity and Variability of Clinical Course of Hormone Dependent Tumors” to Dr. Vasyl Chekhun from the target research program of the National Academy of Sciences of Ukraine “Molecular Genetics and Biochemical Mechanisms of Regulation of Cellular and Systemic Interactions in Physiological and Pathological Conditions.”
Author information
Authors and Affiliations
Contributions
O.L., M.Z., T.K., and N.V.: investigation and validation; T.B., N.L., Y.L., and I.A.: methodology and investigation; A.P.: data curation and visualization; A.P. and M.Z.: writing—original draft; V.C. and A.B.: conceptualization; writing—reviewing and editing; and supervision; All authors have read and approved the article. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding authors
Ethics declarations
Ethical approval
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lykhova, O., Zavelevich, M., Philchenkov, A. et al. Does insulin make breast cancer cells resistant to doxorubicin toxicity?. Naunyn-Schmiedeberg's Arch Pharmacol 396, 3111–3122 (2023). https://doi.org/10.1007/s00210-023-02516-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02516-3