Abstract
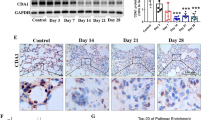
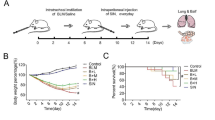
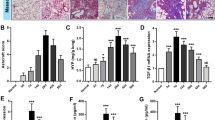
Pulmonary fibrosis (PF) occurs in various end stages of lung disease, and it is characterized by persistent scarring of the lung parenchyma with excessive deposition of extracellular matrix (ECM), leading to degressive quality of life and earlier mortality. FOXO4-D-Retro-Inverso (FOXO4-DRI), a synthesis peptide as a specific FOXO4 blocker, selectively induced dissociation of the FOXO4-p53 complex and nuclear exclusion of p53. Simultaneously, the p53 signaling pathway has been reported to activate in fibroblasts isolated from IPF fibrotic lung tissues and the p53 mutants cooperate with other factors that have the ability to disturb the synthesis of ECM. Yet, whether FOXO4-DRI influences the nuclear exclusion of p53 and then obstructs PF progress is still unknown. In this research, we explored the effect of FOXO4-DRI on bleomycin (BLM)-induced PF mouse model and activated fibroblasts model. The animal group of FOXO4-DRI therapeutic administration shows a milder pathologic change and less collagen deposition compared with the BLM-induced group. We also found the FOXO4-DRI resets the distribution of intranuclear p53 and concurrently decreased the total ECM proteins content. After further validation, FOXO4-DRI may well be a promising therapeutic approach to treating pulmonary fibrosis.






Similar content being viewed by others
Data availability
All the data presented in this study are available on request from the corresponding author.
References
Altomonte J, Cong L, Harbaran S, Richter A, Xu J, Meseck M, Dong HH (2004) Foxo1 mediates insulin action on apoC-III and triglyceride metabolism. J Clin Invest 114:1493–1503
Ashcroft T, Simpson JM, Timbrell V (1988) Simple method of estimating severity of pulmonary fibrosis on a numerical scale. J Clin Pathol 41:467–470
Baar MP, Brandt RMC, Putavet DA, Klein JDD, Derks KWJ, Bourgeois BRM, Stryeck S, Rijksen Y, van Willigenburg H, Feijtel DA, van der Pluijm I, Essers J, van Cappellen WA, van IWF, Houtsmuller AB, Pothof J, de Bruin RWF, Madl T, Hoeijmakers JHJ, Campisi J, de Keizer PLJ (2017) Targeted apoptosis of senescent cells restores tissue homeostasis in response to chemotoxicity and aging. Cell 169: 132-147 e116
Born E, Lipskaia L, Breau M, Houssaini A, Beaulieu DP, Marcos E, Pierre R, Do Cruzeiro M, Lefevre M, Derumeaux G, Bulavin DV, Delcroix M, Quarck R, Reen V, Gil J, Bernard D, Flaman JM, Adnot S, Abid S (2022) Eliminating senescent cells can promote pulmonary hypertension development and progression. Circulation 147(8):650–666
Cabrera S, Maciel M, Herrera I, Nava T, Vergara F, Gaxiola M, Lopez-Otin C, Selman M, Pardo A (2015) Essential role for the ATG4B protease and autophagy in bleomycin-induced pulmonary fibrosis. Autophagy 11:670–684
Chambers RC, Mercer PF (2015) Mechanisms of alveolar epithelial injury, repair, and fibrosis. Ann Am Thorac Soc 12(Suppl 1):S16-20
Chanda D, Otoupalova E, Smith SR, Volckaert T, De Langhe SP, Thannickal VJ (2019) Developmental pathways in the pathogenesis of lung fibrosis. Mol Aspects Med 65:56–69
Ding BS, Cao Z, Lis R, Nolan DJ, Guo P, Simons M, Penfold ME, Shido K, Rabbany SY, Rafii S (2014) Divergent angiocrine signals from vascular niche balance liver regeneration and fibrosis. Nature 505:97–102
Geng Y, Li L, Yan J, Liu K, Yang A, Zhang L, Shen Y, Gao H, Wu X, Noth I, Huang Y, Liu J, Fan X (2022) PEAR1 regulates expansion of activated fibroblasts and deposition of extracellular matrix in pulmonary fibrosis. Nat Commun 13:7114
Ghosh AK, Bhattacharyya S, Varga J (2004) The tumor suppressor p53 abrogates Smad-dependent collagen gene induction in mesenchymal cells. J Biol Chem 279:47455–47463
Han X, Yuan T, Zhang J, Shi Y, Li D, Dong Y, Fan S (2022) FOXO4 peptide targets myofibroblast ameliorates bleomycin-induced pulmonary fibrosis in mice through ECM-receptor interaction pathway. J Cell Mol Med 26:3269–3280
Hinz B, Lagares D (2020) Evasion of apoptosis by myofibroblasts: a hallmark of fibrotic diseases. Nat Rev Rheumatol 16:11–31
Ho YY, Lagares D, Tager AM, Kapoor M (2014) Fibrosis–a lethal component of systemic sclerosis. Nat Rev Rheumatol 10:390–402
How CK, Chien Y, Yang KY, Shih HC, Juan CC, Yang YP, Chiou GY, Huang PI, Chang YL, Chen LK, Wang CY, Hsu HS, Chiou SH, Lee CH (2013) Induced pluripotent stem cells mediate the release of interferon gamma-induced protein 10 and alleviate bleomycin-induced lung inflammation and fibrosis. Shock 39:261–270
Huang Y, He Y, Makarcyzk MJ, Lin H (2021) Senolytic peptide FOXO4-DRI selectively removes senescent cells from in vitro expanded human chondrocytes. Front Bioeng Biotechnol 9:677576
Kang J, Brajanovski N, Chan KT, Xuan J, Pearson RB, Sanij E (2021) Ribosomal proteins and human diseases: molecular mechanisms and targeted therapy. Signal Transduct Target Ther 6:323
Kato K, Logsdon NJ, Shin YJ, Palumbo S, Knox A, Irish JD, Rounseville SP, Rummel SR, Mohamed M, Ahmad K, Trinh JM, Kurundkar D, Knox KS, Thannickal VJ, Hecker L (2020) Impaired myofibroblast dedifferentiation contributes to nonresolving fibrosis in aging. Am J Respir Cell Mol Biol 62:633–644
Kim DS, Collard HR, King TE Jr (2006) Classification and natural history of the idiopathic interstitial pneumonias. Proc Am Thorac Soc 3:285–292
King TE, Pardo A, Selman M (2011) Idiopathic pulmonary fibrosis. Lancet 378:1949–1961
Kruiswijk F, Labuschagne CF, Vousden KH (2015) p53 in survival, death and metabolic health: a lifeguard with a licence to kill. Nat Rev Mol Cell Biol 16:393–405
Lederer DJ, Martinez FJ (2018) Idiopathic pulmonary fibrosis. N Engl J Med 378:1811–1823
Meng J, Li Y, Wan C, Sun Y, Dai X, Huang J, Hu Y, Gao Y, Wu B, Zhang Z, Jiang K, Xu S, Lovell JF, Hu Y, Wu G, Jin H, Yang K (2021) Targeting senescence-like fibroblasts radiosensitizes non-small cell lung cancer and reduces radiation-induced pulmonary fibrosis. JCI Insight 6(23):e146334
Montero P, Milara J, Roger I, Cortijo J (2021) Role of JAK/STAT in interstitial lung diseases; molecular and cellular mechanisms. Int J Mol Sci 22(12):6211
Nagaraja MR, Tiwari N, Shetty SK, Marudamuthu AS, Fan L, Ostrom RS, Fu J, Gopu V, Radhakrishnan V, Idell S, Shetty S (2018) p53 expression in lung fibroblasts is linked to mitigation of fibrotic lung remodeling. Am J Pathol 188:2207–2222
O’Reilly MA, Marr SH, Yee M, McGrath-Morrow SA, Lawrence BP (2008) Neonatal hyperoxia enhances the inflammatory response in adult mice infected with influenza A virus. Am J Respir Crit Care Med 177:1103–1110
Raghu G, Collard HR, Egan JJ, Martinez FJ, Behr J, Brown KK, Colby TV, Cordier JF, Flaherty KR, Lasky JA, Lynch DA, Ryu JH, Swigris JJ, Wells AU, Ancochea J, Bouros D, Carvalho C, Costabel U, Ebina M, Hansell DM, Johkoh T, Kim DS, King TE Jr, Kondoh Y, Myers J, Muller NL, Nicholson AG, Richeldi L, Selman M, Dudden RF, Griss BS, Protzko SL, Schunemann HJ, Fibrosis AEJACoIP, (2011) An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med 183:788–824
Raghu G, Weycker D, Edelsberg J, Bradford WZ, Oster G (2006) Incidence and prevalence of idiopathic pulmonary fibrosis. Am J Respir Crit Care Med 174:810–816
Rehan M, Kurundkar D, Kurundkar AR, Logsdon NJ, Smith SR, Chanda D, Bernard K, Sanders YY, Deshane JS, Dsouza KG, Rangarajan S, Zmijewski JW, Thannickal VJ (2021) Restoration of SIRT3 gene expression by airway delivery resolves age-associated persistent lung fibrosis in mice. Nat Aging 1:205–217
Richeldi L, Collard HR, Jones MG (2017) Idiopathic pulmonary fibrosis. Lancet 389:1941–1952
Shetty SK, Tiwari N, Marudamuthu AS, Puthusseri B, Bhandary YP, Fu J, Levin J, Idell S, Shetty S (2017) p53 and miR-34a feedback promotes lung epithelial injury and pulmonary fibrosis. Am J Pathol 187:1016–1034
Thannickal VJ, Toews GB, White ES, Lynch JP 3rd, Martinez FJ (2004) Mechanisms of pulmonary fibrosis. Annu Rev Med 55:395–417
Thoreen CC, Sabatini DM (2005) AMPK and p53 help cells through lean times. Cell Metab 1:287–288
Wollin L, Wex E, Pautsch A, Schnapp G, Hostettler KE, Stowasser S, Kolb M (2015) Mode of action of nintedanib in the treatment of idiopathic pulmonary fibrosis. Eur Respir J 45:1434–1445
Wolters PJ, Collard HR, Jones KD (2014) Pathogenesis of idiopathic pulmonary fibrosis. Annu Rev Pathol 9:157–179
Yang L, Zhou F, Zheng D, Wang D, Li X, Zhao C, Huang X (2021) FGF/FGFR signaling: from lung development to respiratory diseases. Cytokine Growth Factor Rev 62:94–104
Yee M, Buczynski BW, Lawrence BP, O’Reilly MA (2013) Neonatal hyperoxia increases sensitivity of adult mice to bleomycin-induced lung fibrosis. Am J Respir Cell Mol Biol 48:258–266
Yee M, Vitiello PF, Roper JM, Staversky RJ, Wright TW, McGrath-Morrow SA, Maniscalco WM, Finkelstein JN, O’Reilly MA (2006) Type II epithelial cells are critical target for hyperoxia-mediated impairment of postnatal lung development. Am J Physiol Lung Cell Mol Physiol 291:L1101-1111
Zhang C, Xie Y, Chen H, Lv L, Yao J, Zhang M, Xia K, Feng X, Li Y, Liang X, Sun X, Deng C, Liu G (2020) FOXO4-DRI alleviates age-related testosterone secretion insufficiency by targeting senescent Leydig cells in aged mice. Aging (Albany NY) 12:1272–1284
Zhou W, Cao Q, Peng Y, Zhang QJ, Castrillon DH, DePinho RA, Liu ZP (2009) FoxO4 inhibits NF-kappaB and protects mice against colonic injury and inflammation. Gastroenterology 137:1403–1414
Funding
This work was supported by funds from the National Natural Science Foundation of China (Grant No. 81870057 and 82070062), the Natural Science Foundation of Hubei Province (Grant No. 2020CFA018), the Translational Medicine and Interdisciplinary Research Joint Found of Zhongnan Hospital of Wuhan University (Grant. No. ZNJC202218), and the Climbing Project for Medical Talent of Zhongnan Hospital, Wuhan University (Grant No. PDJH202205).
Author information
Authors and Affiliations
Contributions
YiL conceived and designed research, performed experiments, analyzed data, and drafted the manuscript. QH performed experiments, analyzed data, and drafted the manuscript. RW performed animal experiments. YuL conceived and designed research and edited and revised the manuscript. ZC conceived and designed research, analyzed data, drafted the manuscript, and edited and revised the manuscript. YiL, QH, RW, YuL, and ZC approved the final version of the manuscript. The authors declare that all data were generated in-house and that no paper mill was used.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics approval
The animal study was reviewed and approved by the Animal Care and Use Committee of Zhongnan Hospital at Wuhan University (Approval NO. ZN2021250).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Liu, Y., Hou, Q., Wang, R. et al. FOXO4-D-Retro-Inverso targets extracellular matrix production in fibroblasts and ameliorates bleomycin-induced pulmonary fibrosis in mice. Naunyn-Schmiedeberg's Arch Pharmacol 396, 2393–2403 (2023). https://doi.org/10.1007/s00210-023-02452-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-023-02452-2