Abstract
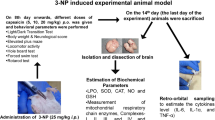
Asperuloside (ASP) and geniposide (GP) are iridoids that have shown various biological properties, such as reduction of inflammation, oxidative stress, and neuroprotection. The aim of this study was to investigate the mechanism of action of ASP and GP through the experimental model of pilocarpine-induced seizures. Mice were treated daily with saline, valproic acid (VPA), GP (5, 25, or 50 mg/kg), or ASP (20 or 40 mg/kg) for 8 days. Pilocarpine (PILO) treatment was administered after the last day of treatment, and the epileptic behavior was recorded for 1 h and analyzed by an adapted scale. Afterward, the hippocampus and blood samples were collected for western blot analyses, ELISA and comet assay, and bone marrow to the micronucleus test. We evaluated the expression of the inflammatory marker cyclooxygenase-2 (COX-2), GluN2B, a subunit of the NMDA receptor, pGluR1, an AMPA receptor, and the enzyme GAD-1 by western blot and the cytokine TNF-α by ELISA. The treatments with GP and ASP were capable to decrease the latency to the first seizure, although they did not change the latency to status epilepticus (SE). ASP demonstrated a genotoxic potential analyzed by comet assay; however, the micronuclei frequency was not increased in the bone marrow. The GP and ASP treatments were capable to reduce COX-2 and GluN2B receptor expression after PILO exposure. This study suggests that GP and ASP have a protective effect on PILO-induced seizures, decreasing GluN2B receptor and COX-2 expression.







Similar content being viewed by others
Data availability
The datasets generated during the current study are available in the supplementary material.
References
Akyuz E, Polat AK, Eroglu E et al (2021) Revisiting the role of neurotransmitters in epilepsy: an updated review. Life Sci 265:1–21. https://doi.org/10.1016/J.LFS.2020.118826
Alachkar A, Azimullah S, Lotfy M et al (2020) Antagonism of histamine H3 receptors alleviates pentylenetetrazole-induced kindling and associated memory deficits by mitigating oxidative stress, central neurotransmitters, and c-Fos protein expression in rats. Molecules 25:1–16. https://doi.org/10.3390/MOLECULES25071575
Asgharzade S, Rabiei Z, Rabiei S et al (2020) Therapeutic effects of oleuropein in improving seizure, oxidative stress and cognitive disorder in pentylenetetrazole kindling model of epilepsy in mice. Iran J Pharm Res 19:98–110. https://doi.org/10.22037/ijpr.2019.14212.12209
Azqueta A, Slyskova J, Langie SAS et al (2014) Comet assay to measure DNA repair: approach and applications. Front Genet 5:1–8. https://doi.org/10.3389/fgene.2014.00288
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254. https://doi.org/10.1016/0003-2697(76)90527-3
Buddhala C, Hsu CC, Wu JY (2009) A novel mechanism for GABA synthesis and packaging into synaptic vesicles. Neurochem Int 55:9–12. https://doi.org/10.1016/J.NEUINT.2009.01.020
Chan Y, Ng SW, Xin Tan JZ et al (2020) Emerging therapeutic potential of the iridoid molecule, asperuloside: a snapshot of its underlying molecular mechanisms. Chem Biol Interact 315:108911. https://doi.org/10.1016/j.cbi.2019.108911
Chen S, Zeng X, Zong W et al (2019) Aucubin alleviates seizures activity in Li-pilocarpine-induced epileptic mice: involvement of inhibition of neuroinflammation and regulation of neurotransmission. Neurochem Res 44:472–484. https://doi.org/10.1007/s11064-018-2700-y
Chen Y er, Xu S jie, Lu Y yu et al (2021) Asperuloside suppressing oxidative stress and inflammation in DSS-induced chronic colitis and RAW 264.7 macrophages via Nrf2/HO-1 and NF-κB pathways. Chem Biol Interact 344:1–13. https://doi.org/10.1016/J.CBI.2021.109512
Collins AR (2004) The comet assay for DNA damage and repair: principles, applications, and limitations. Appl Biochem Biotechnol - Part B Mol Biotechnol 26:249–261. https://doi.org/10.1385/MB:26:3:249
Curia G, Longo D, Biagini G et al (2008) The pilocarpine model of temporal lobe epilepsy. J Neurosci Methods 172:143–157
da Costa e Silva LD, Pereira P, Regner GG et al (2018) DNA damage and oxidative stress induced by seizures are decreased by anticonvulsant and neuroprotective effects of lobeline, a candidate to treat alcoholism. Metab Brain Dis 33:53–61. https://doi.org/10.1007/s11011-017-0130-1
Dang YL, Foster E, Lloyd M et al (2021) Adverse events related to antiepileptic drugs. Epilepsy Behav 115:1–10. https://doi.org/10.1016/J.YEBEH.2020.107657
Dinda B, Dinda M, Kulsi G et al (2019) Therapeutic potentials of plant iridoids in Alzheimer’s and Parkinson’s diseases: a review. Eur J Med Chem 169:185–199. https://doi.org/10.1016/j.ejmech.2019.03.009
Dubey V, Dey S, Dixit AB et al (2022) Differential glutamate receptor expression and function in the hippocampus, anterior temporal lobe and neocortex in a pilocarpine model of temporal lobe epilepsy. Exp Neurol 347:113916. https://doi.org/10.1016/j.expneurol.2021.113916
Esclapez M, Houser CR (1993) Loss of glutamate decarboxylase mRNA-containing neurons in the rat dentate gyrus following pilocarpine-induced seizures. J Neurosci 3:4470–4485
Fisher RS, Acevedo C, Arzimanoglou A et al (2014) ILAE official report: a practical clinical definition of epilepsy. Epilepsia 55:475–482. https://doi.org/10.1111/epi.12550
Fleck J, Temp FR, Marafiga JR et al (2016) Montelukast reduces seizures in pentylenetetrazol-kindledmice. Brazilian J Med Biol Res 49:1–7. https://doi.org/10.1590/1414-431X20155031
Freichel C, Potschka H, Ebert U et al (2006) Acute changes in the neuronal expression of GABA and glutamate decarboxylase isoforms in the rat piriform cortex following status epilepticus. Neuroscience 141:2177–2194. https://doi.org/10.1016/J.NEUROSCIENCE.2006.05.040
He D, Xu Y (2022) Toxicological evaluation of Camellia euphlebia leaves aqueous extract using acute and subacute toxicity studies in mice and genotoxicity studies. Evid Based Complement Alternat Med 2022:1–10. https://doi.org/10.1155/2022/7889199
Jiang H, Panda S, Gekara NO (2019) Comet and micronucleus assays for analyzing DNA damage and genome integrity. Methods Enzymol 625:299–307. https://doi.org/10.1016/BS.MIE.2019.05.015
Johnson EL (2019) Seizures and epilepsy. Med Clin North Am 103:309–324. https://doi.org/10.1016/J.MCNA.2018.10.002
Lee B, Dziema H, Lee KH et al (2007) CRE-mediated transcription and COX-2 expression in the pilocarpine model of status epilepticus. Neurobiol Dis 25:80. https://doi.org/10.1016/J.NBD.2006.08.015
Li C, Wang X, Cheng F et al (2019) Geniposide protects against hypoxia/reperfusion-induced blood-brain barrier impairment by increasing tight junction protein expression and decreasing inflammation, oxidative stress, and apoptosis in an in vitro system. Eur J Pharmacol 854:224–231. https://doi.org/10.1016/J.EJPHAR.2019.04.021
Lima IV de A, Bellozi PMQ, Batista EM et al (2020) Cannabidiol anticonvulsant effect is mediated by the PI3Kγ pathway. Neuropharmacol 176:1–12. https://doi.org/10.1016/j.neuropharm.2020.108156
Lotarski S, Hain H, Peterson J et al (2014) Anticonvulsant activity of pregabalin in the maximal electroshock-induced seizure assay in α2δ1 (R217A) and α2δ2 (R279A) mouse mutants. Epilepsy Res 108:833–842. https://doi.org/10.1016/J.EPLEPSYRES.2014.03.002
Manzione MG, Martorell M, Sharopov F et al (2020) Phytochemical and pharmacological properties of asperuloside, a systematic review. Eur J Pharmacol 883:1–9. https://doi.org/10.1016/J.EJPHAR.2020.173344
Mavournin KH, Blakey DH, Cimino MC et al (1990) The in vivo micronucleus assay in mammalian bone marrow and peripheral blood. A report of the U.S. Environmental Protection Agency Gene-Tox Program. Mutat Res Genet Toxicol 239:29–80
OECD (2016) Test No. 474: mammalian erythrocyte micronucleus test, OECD guidelines for the testing of chemicals, Section 4. Paris: OECD. https://doi.org/10.1787/9789264264762-en
Oliveira AA, Nogueira CRA, Nascimento VS et al (2005) Evaluation of levetiracetam effects on pilocarpine-induced seizures: cholinergic muscarinic system involvement. Neurosci Lett 385:184–188. https://doi.org/10.1016/J.NEULET.2005.05.048
Qiu J, Chi G, Wu Q et al (2016) Pretreatment with the compound asperuloside decreases acute lung injury via inhibiting MAPK and NF-κB signaling in a murine model. Int Immunopharmacol 31:109–115. https://doi.org/10.1016/j.intimp.2015.12.013
Racine RJ (1972) Modification of seizure activity by electrical stimulation: II Motor Seizure. Electroencephalogr Clin Neurophysiol 32:281–294. https://doi.org/10.1016/0013-4694(72)90177-0
Salazar P, Tapia R (2015) Epilepsy and hippocampal neurodegeneration induced by glutamate decarboxylase inhibitors in awake rats. Epilepsy Res 116:27–33. https://doi.org/10.1016/J.EPLEPSYRES.2015.06.014
Santulli L, Coppola A, Balestrini S, Striano S (2016) The challenges of treating epilepsy with 25 antiepileptic drugs. Pharmacol Res 107:211–219. https://doi.org/10.1016/J.PHRS.2016.03.016
Shen B, Feng H, Cheng J et al (2020) Geniposide alleviates non-alcohol fatty liver disease via regulating Nrf2/AMPK/mTOR signalling pathways. J Cell Mol Med 24:5097–5108. https://doi.org/10.1111/JCMM.15139
Shishmanova-Doseva M, Peychev L, Yoanidu L et al (2021) Anticonvulsant effects of topiramate and lacosamide on pilocarpine-induced status epilepticus in rats: a role of reactive oxygen species and inflammation. Int J Mol Sci 22:1–13. https://doi.org/10.3390/IJMS22052264
Singh NP, McCoy MT, Tice RR, Schneider EL (1988) A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res 175:184–191. https://doi.org/10.1016/0014-4827(88)90265-0
Stachowicz K (2021) Deciphering the mechanisms of regulation of an excitatory synapse via cyclooxygenase-2. A Review Biochem Pharmacol 192:114729. https://doi.org/10.1016/J.BCP.2021.114729
Stark DT, Bazan NG (2011) Synaptic and extrasynaptic NMDA receptors differentially modulate neuronal cyclooxygenase-2 function, lipid peroxidation, and neuroprotection. J Neurosci 31:13710–13721. https://doi.org/10.1523/JNEUROSCI.3544-11.2011
Su C, Yang X, Lou J (2016) Geniposide reduces α-synuclein by blocking microRNA-21/lysosome-associated membrane protein 2A interaction in Parkinson disease models. Brain Res 1644:98–106. https://doi.org/10.1016/j.brainres.2016.05.011
Takemiya T, Maehara M, Matsumura K et al (2006) Prostaglandin E2 produced by late induced COX-2 stimulates hippocampal neuron loss after seizure in the CA3 region. Neurosci Res 56:103–110. https://doi.org/10.1016/J.NEURES.2006.06.003
Trinka E, Cock H, Hesdorffer D et al (2015) A definition and classification of status epilepticus - report of the ILAE task force on classification of status epilepticus. Epilepsia 56:1515–1523. https://doi.org/10.1111/epi.13121
Turski L, Cavalheiro EA, Sieklucka-Dziuba M et al (1986) Seizures produced by pilocarpine: neuropathological sequelae and activity of glutamate decarboxylase in the rat forebrain. Brain Res 398:37–48. https://doi.org/10.1016/0006-8993(86)91247-3
Vendruscolo MH, das Neves GM, Kagami LP et al (2018) In vitro and in silico activity of iridoids against Leishmania amazonensis. Curr Drug Discov Technol 16:173–183. https://doi.org/10.2174/1570163814666171002102058
Wang J, Li Y, Huang WH et al (2017) The protective effect of aucubin from Eucommia ulmoides against status epilepticus by inducing autophagy and inhibiting necroptosis. Am J Chin Med 45:557–573. https://doi.org/10.1142/S0192415X17500331
Wang Z, Zhou L, An D et al (2019) TRPV4-induced inflammatory response is involved in neuronal death in pilocarpine model of temporal lobe epilepsy in mice. Cell Death Dis 10:1–10. https://doi.org/10.1038/s41419-019-1612-3
Wei H, Duan G, He J et al (2018) Geniposide attenuates epilepsy symptoms in a mouse model through the PI3K/Akt/GSK-3β signaling pathway. Exp Ther Med 15:1136–1142. https://doi.org/10.3892/etm.2017.5512
Wu Q, Gai S, Zhang H (2022) Asperulosidic acid, a bioactive iridoid, alleviates placental oxidative stress and inflammatory responses in gestational diabetes mellitus by suppressing NF-κB and MAPK signaling pathways. Pharmacology 107:1–9. https://doi.org/10.1159/000521080
Yang N, Zhang Y, Wang JT et al (2020) Effects of dexamethasone on remodeling of the hippocampal synaptic filamentous actin cytoskeleton in a model of pilocarpine-induced status epilepticus. Int J Med Sci 17:1683–1691. https://doi.org/10.7150/ijms.44927
Yao H, Liu W et al (2021) Geniposide attenuates postischemic long-term potentiation via GluN2A. Pak J Pharm Sc 34:909–914. https://doi.org/10.36721/PJPS.2021.34.3.REG.909-914.1
Youssef FS, Menze ET, Ashour ML (2020) A potent lignan from prunes alleviates inflammation and oxidative stress in lithium/pilocarpine-induced epileptic seizures in rats. Antioxidants 9:1–15. https://doi.org/10.3390/ANTIOX9070575
Zhang W, Zhang F, Hu Q et al (2021) The emerging possibility of the use of geniposide in the treatment of cerebral diseases: a review. Chin Med 16:1–16. https://doi.org/10.1186/S13020-021-00486-3
Zhang Y, Yin F, Liu J et al (2015) Geniposide attenuates insulin-deficiency-induced acceleration of β-amyloidosis in an APP/PS1 transgenic model of Alzheimer’s disease. Neurochem Int 89:7–16. https://doi.org/10.1016/J.NEUINT.2015.04.002
Zhao Y, Li H, Fang F et al (2018) Geniposide improves repeated restraint stress-induced depression-like behavior in mice by ameliorating neuronal apoptosis via regulating GLP-1R/AKT signaling pathway. Neurosci Lett 676:19–26. https://doi.org/10.1016/j.neulet.2018.04.010
Zhou R, Lu G, Yan Z et al (2019a) Bioaccumulation and biochemical effects of ethylhexyl methoxy cinnamate and its main transformation products in zebrafish. Aquat Toxicol 214:1–9. https://doi.org/10.1016/j.aquatox.2019.105241
Zhou YX, Zhang RQ, Rahman K et al (2019b) Diverse pharmacological activities and potential medicinal benefits of geniposide. Evidence-based Complement Altern Med 2019:1–15. https://doi.org/10.1155/2019
Acknowledgements
This work was supported by the Higher Education Personnel Improvement Coordination (CAPES) and the Federal University of Rio Grande do Sul (UFRGS).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. Mariana Uczay and Pricila Pflüger carried out the assays in the study and participated in the drafting of the manuscript. Gilsane von Poser and Maria Helena Vendruscolo extracted the compounds and participated in the drafting of the manuscript. Iraci Torres provided reagents and participated in the drafting of the manuscript. Helouise Richardt Medeiros helped with the measurement of TNF-α and João Denis Medeiros de Oliveira the comet assay. Jaqueline Nascimento Picada and Patrícia Pereira conceived, designed, supervised the study, and drafted the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All experimental procedures were carried out in accordance with the Guidelines of the Brazilian Council of Animal Experimentation—CONCEA, with the approval of the Committee on the Ethical Use of Animals of UFRGS (authorization number: 37769).
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Uczay, M., Pflüger, P., Picada, J.N. et al. Geniposide and asperuloside alter the COX-2 and GluN2B receptor expression after pilocarpine-induced seizures in mice. Naunyn-Schmiedeberg's Arch Pharmacol 396, 951–962 (2023). https://doi.org/10.1007/s00210-022-02367-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-022-02367-4