Abstract
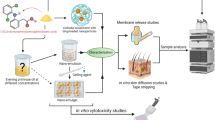
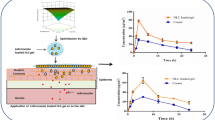
Iloperidone (ILO) is a poorly soluble and bioavailable WHO-approved schizophrenia drug. Microneedles are a revolutionary delivery technology that overcomes many of the issues associated with traditional drug administration. The current research aimed to compare the antipsychotic activity and pharmacokinetics of ILO-loaded dissolving microneedles (DMNs) and transdermal film with a solid microneedle (STF). The DMNs were fabricated using the micromolding process, while the transdermal film was created using the solvent casting approach. Furthermore, an in vivo pharmacokinetic, pharmacodynamic, and skin irritation study was performed on Wistar rats. Studies were compared with transdermal film (TF) on untreated skin as a passive control. STF and DMNs had considerably greater AUC and Cmax (p ≤ 0.001) than transdermal film. In pharmacodynamic tests, STF and DMNs demonstrated significant (p ≤ 0.001) forelimb retraction time (FRT) and hindlimb retraction time (HRT) delay responses as compared to control and TF. In the skin irritation test, no adverse effects such as erythema or edema were observed at the end of the 48 h. Thus, antipsychotic activity (paw test) and pharmacokinetics studies revealed sustained action of DMN and STF. This research revealed that improved efficacy of DMN and STF for antipsychotic drug delivery may be an alternative to the existing dosage form.




Similar content being viewed by others
Data availability
Access to data will be granted from the authors on reasonable request.
All institutional and national guidelines for the care and use of laboratory animals were followed.
References
Accessdata.fda.gov. (2022). Retrieved 23 March 2022, from https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/215413s000lbl.pdf
Bhadale R, Londhe V (2021) A systematic review of carbohydrate-based microneedles: current status and future prospects. J Mater Sci Mater Med 32(8). https://doi.org/10.1007/s10856-021-06559-x
Bhadale R, Londhe V (2022) Inclusion complexed iloperidone loaded dissolving microneedles: characterization, in-vitro study, and dermatopharmacokinetics. J Drug Deliv Sci Technol 68:103063. https://doi.org/10.1016/j.jddst.2021.103063
Bošković M, Grabnar I, Terzič T, Kores Plesničar B, Vovk T (2013) Oxidative stress in schizophrenia patients treated with long-acting haloperidol decanoate. Psychiatry Res 210(3):761–768. https://doi.org/10.1016/j.psychres.2013.08.035
Citrome L, Zeni C, Correll C (2019) Patches. J Clin Psychiatry 80(4). https://doi.org/10.4088/jcp.18nr12554
Dosage & application | SECUADO® (asenapine) Patch. Secuado. (2022). Retrieved 23 April 2022, from https://www.secuado.com/hcp/prescribing-secuado/
Ellenbroek B, Peeters B, Honig W, Cools A (1987) The paw test: a behavioural paradigm for differentiating between classical and atypical neuroleptic drugs. Psychopharmacology 93(3). https://doi.org/10.1007/bf00187254
Ema.europa.eu. (2022). Retrieved 15 January 2022, from https://www.ema.europa.eu/en/documents/assessment-report/fanaptum-epar-refusal-public-assessment-report_en.pdf.HighlightsofPrescribingMedication. www.accessdata.fda.gov/drugsatfda_docs/label/2013/022192s007lbl.pdf
Hoang M, Ita K, Bair D (2015) Solid microneedles for transdermal delivery of amantadine hydrochloride and pramipexole dihydrochloride. Pharmaceutics 7(4):379–396. https://doi.org/10.3390/pharmaceutics7040379
Londhe V, Bhasin B (2019) Transdermal lipid vesicular delivery of iloperidone: formulation, in vitro and in vivo evaluation. Colloids Surf B Biointerfaces 183:110409. https://doi.org/10.1016/j.colsurfb.2019.110409
Londhe V, Shirsat R (2018) Formulation and characterization of fast-dissolving sublingual film of iloperidone using Box-Behnken design for enhancement of oral bioavailability. AAPS PharmSciTech 19(3):1392–1400. https://doi.org/10.1208/s12249-018-0954-y
Londhe V, Deshmane A, Singh S, Kulkarni Y (2018) Lurasidone-β-cyclodextrin complexes: physicochemical characterization and comparison of their antidepressant, antipsychotic activities against that of self microemulsifying formulation. J Mol Struct 1157:395–400. https://doi.org/10.1016/j.molstruc.2017.12.042
McCrudden M, Alkilani A, McCrudden C, McAlister E, McCarthy H, Woolfson A, Donnelly R (2014) Design and physicochemical characterisation of novel dissolving polymeric microneedle arrays for transdermal delivery of high dose, low molecular weight drugs. J Control Release 180:71–80. https://doi.org/10.1016/j.jconrel.2014.02.007
Mental disorders. Who.int. (2022). Retrieved 23 January 2022, from https://www.who.int/news-room/fact-sheets/detail/mental-disorders
Omolu A, Bailly M, Day R (2017) Assessment of solid microneedle rollers to enhance transmembrane delivery of doxycycline and inhibition of MMP activity. Drug Delivery 24(1):942–951. https://doi.org/10.1080/10717544.2017.1337826
Pillai A, Parikh V, Terry A, Mahadik S (2007) Long-term antipsychotic treatments and crossover studies in rats: differential effects of typical and atypical agents on the expression of antioxidant enzymes and membrane lipid peroxidation in rat brain. J Psychiatr Res 41(5):372–386. https://doi.org/10.1016/j.jpsychires.2006.01.011
Schizophrenia facts and statistics. Schizophrenia.com. (2022). Retrieved 23 January 2022, from http://schizophrenia.com/szfacts.htm
Shirzadi A, Ghaemi N (2006) Side effects of atypical antipsychotics: extrapyramidal symptoms and the metabolic syndrome. Harv Rev Psychiatry 14(3):152–164. https://doi.org/10.1080/10673220600748486
Singh O, Chakraborty I, Dasgupta A, Datta S (2008) A comparative study of oxidative stress and interrelationship of important antioxidants in haloperidol and olanzapine treated patients suffering from schizophrenia. Indian J Psychiatry 50(3):171. https://doi.org/10.4103/0019-5545.43627
Test No. 404: acute dermal irritation/corrosion | en | OECD. Oecd.org. (2022). Retrieved 23 May 2022, from https://www.oecd.org/env/test-no-404-acute-dermal-irritation-corrosion-9789264242678-en.htm
Van Os J, Kapur S (2009) Schizophrenia. The Lancet 374(9690):635–645. https://doi.org/10.1016/s0140-6736(09)60995-8
Funding
This work was supported by the Science and Engineering Research Board, File Number: CRG/2018/003176.
Author information
Authors and Affiliations
Contributions
The study conception and design were done by VL. Material preparation, data collection, and analysis were performed by RB. The first draft of the manuscript was written by RB and approved by VL. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All animal samples were obtained and ethical committee approval (Protocol number: CPCSEA/IAEC/P-19/2019). All authors have reviewed and agreed with the content of the manuscript.
Consent for publication
All authors have reviewed, agreed with the content of the manuscript, and consented to its publication.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bhadale, R.S., Londhe, V.Y. A comparison of dissolving microneedles and transdermal film with solid microneedles for iloperidone in vivo: a proof of concept. Naunyn-Schmiedeberg's Arch Pharmacol 396, 239–246 (2023). https://doi.org/10.1007/s00210-022-02309-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-022-02309-0