Abstract
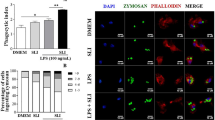
The role of protease-activated receptor (PAR)4 in thrombin-induced platelet aggregation has been studied, and PAR4 blockade is thought to be useful as a new and promising approach in antiplatelet therapy in humans. In recent years, studies have been conducted to clarify the role of PAR4 in the host defense against invading microorganisms and pathogen-induced inflammation; however, to date, the role of PAR4 in mediating the LPS-induced inflammatory repertoire in macrophages remains to be elucidated. Here, we investigated the effects of the synthetic PAR4 agonist peptide (PAR4-AP) AYPGKF-NH2 on the phagocytosis of zymosan-FITC particles; NO, ROS, and iNOS expression; and cytokine production in C57/BL6 macrophages cocultured with PAR4-AP/LPS. The PAR4-AP impaired LPS-induced and basal phagocytosis, which was restored by pharmacological PAR4 blockade. Coincubation with the PAR4-AP/LPS enhanced NO and ROS production and iNOS expression; decreased IL-10, but not TNF-α, in the culture supernatant; and increased translocation of the p65 subunit of the proinflammatory gene transcription factor NF-κ-B. Our results provide evidence for a complex mechanism and new approach by which PAR4 mediates the macrophage response triggered by LPS through counter-regulating the phagocytic activity of macrophages and innate response mechanisms implicated in the killing of invading pathogens.





Similar content being viewed by others
References
Adams MN, Ramachandran R, Yau MK, Suen JY, Fairlie DP, Hollenberg MD, Hooper JD (2011) Structure, function and pathophysiology of protease activated receptors. Pharmacol Ther. 130:248–282. https://doi.org/10.1016/j.pharmthera.2011.01.003
Benoit M, Desnues B, Mege JL (2008) Macrophage polarization in bacterial infections. J Immunol 181:3733–3739. https://doi.org/10.4049/jimmunol.181.6.3733
Bogdan C (2015) Nitric oxide synthase in innate and adaptive immunity: an update. Trends Immunol 36:161–178. https://doi.org/10.1016/j.it.2015.01.003
Braga AD, Miranda JP, Ferreira GM, Bilheiro RP, Duarte ID, Francischi JN, Klein A (2010) Blockade of proteinase-activated receptor-4 inhibits the eosinophil recruitment induced by eotaxin-1 in the pleural cavity of mice. Pharmacology 86:224–230. https://doi.org/10.1159/000319751
Bryniarski K, Maresz K, Szczepanik M, Ptak M, Ptak W (2003) Modulation of macrophage activity by proteolytic enzymes. Differential regulation of IL-6 and reactive oxygen intermediates (ROIs) synthesis as a possible homeostatic mechanism in the control of inflammation. Inflammation 27:333–340. https://doi.org/10.1023/B:IFLA.0000006701.52150.43
Carrim N, Arthur JF, Hamilton JR, Gardiner EE, Andrews RK, Moran N, Berndt MC, Metharom P (2015) Thrombin-induced reactive oxygen species generation in platelets: a novel role for protease-activated receptor 4 and GPIbα. Redox Biol 6:640–647. https://doi.org/10.1016/j.redox.2015.10.009
Cottrell GS, Amadesi S, Grady ES, Bunnett NW (2004) Trypsin IV a novel agonist of protease-activated receptors 2 and 4. J Biol Chem 279:13532–13539. https://doi.org/10.1074/jbc.M312090200
Coughlin SR (2000) Thrombin signalling and protease-activated receptors. Nature 407:258–264. https://doi.org/10.1038/35025229
Estevez B, Du X (2017) New concepts and mechanisms of platelet activation signaling. Physiology 32:162–177. https://doi.org/10.1152/physiol.00020.2016
Fang X, Liao R, Yu Y, Li J, Guo Z, Zhu T (2019) Thrombin induces secretion of multiple cytokines and expression of protease-activated receptors in mouse mast cell line. Mediat Inflamm 2019:1–12. https://doi.org/10.1155/2019/4952131
Fuentes AL, Millis L, Sigoia LB (2011) Laminarin, a soluble beta-glucan, inhibits macrophage phagocytosis of zymosan but has no effect on lipopolysaccharide mediated augmentation of phagocytosis. Int Immunopharmacol 11:1939–1945. https://doi.org/10.1016/j.intimp.2011.08.005
Gomides LF, Duarte ID, Ferreira RG, Perez AC, Francischi JN, Klein A (2012) Proteinase-activated receptor-4 plays a major role in the recruitment of neutrophils induced by trypsin or carrageenan during pleurisy in mice. Pharmacology 89:275–282. https://doi.org/10.1159/000337378
Gomides LF, Lima OCO, Matos NA, Freitas KM, Francischi JN, Tavares JC, Klein A (2014) Blockade of proteinase-activated receptor 4 inhibits neutrophil recruitment in experimental inflammation in mice. Inflamm Res 63:935–941. https://doi.org/10.1007/s00011-014-0767-8
Gordon S (2016) Phagocytosis: an immunobiologic process. Immunity 44:463–475. https://doi.org/10.1016/j.immuni.2016.02.026
Gordon S, Martinez FO (2010) Alternative activation of macrophages: mechanism and functions. Immunity 32:593–604. https://doi.org/10.1016/j.immuni.2010.05.007
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite and [‘5N] nitrate in biological fluids. Anal Biochem 126:131–138. https://doi.org/10.1016/0003-2697(82)90118-X
Hollenberg MD, Compton SJ (2002) International Union of Pharmacology. XXVIII Proteinase-activated receptors. Pharmacol Rev 54:203–217. https://doi.org/10.1124/pr.54.2.203
Hosokawa K, Ohnishi T, Miura N, Sameshima H, Koide T, Tanaka KA, Maruyama I (2014) Antithrombotic effects of PAR1 and PAR4 antagonists evaluated under flow and static conditions. Thromb Res 133:66–72. https://doi.org/10.1016/j.thromres.2013.10.037
Jesmin S, Gando S, Zaedi S, Sakuraya F (2007) Differential expression, time course and distribution of four PARs in rats with endotoxin-induced acute lung injury. Inflammation 30:14–27. https://doi.org/10.1007/s10753-006-9017-8
Kadish J, Choi CC, Czop JK (1986) Phagocytosis of unopsonized zymosan particles by trypsin-sensitive and beta-glucan-inhibitable receptors on bone marrow-derived murine macrophages. Immunol Res 5:129–138. https://doi.org/10.1007/BF02917587
Kahn ML, Zheng YW, Huang W, Bigornia V, Zeng D, Moff S, Farese RV Jr, Tam C, Coughlin SR (1998) A dual thrombin receptor system for platelet activation. Nature 394:690–694. https://doi.org/10.1038/29325
Kahn ML, Nakanishi-Matsui M, Shapiro MJ, Ishihara H, Coughlin SR (1999) Protease-activated receptors 1 and 4 mediate activation of human platelets by thrombin. J Clin Invest 103:879–887. https://doi.org/10.1172/JCI6042
Klein A, Talvani A, Silva PM, Martins MA, Wells TN, Proudfoot A, Luckacs NW, Teixeira MM (2001) Stem cell factor-induced leukotriene B4 production cooperates with eotaxin to mediate the recruitment of eosinophils during allergic pleurisy in mice. J Immunol 167:524–531. https://doi.org/10.4049/jimmunol.167.1.524
Lê VB, Schneider JG, Boergeling Y, Berri F, Ducatez M, Guerin JL, Adrian I, Errazuriz-Cerda E, Frasquilho S, Antunes L, Lina B, Bordet JC, Jandrot-Perrus M, Ludwig S, Riteau B (2015) Platelet activation and aggregation promote lung inflammation and influenza virus pathogenesis. Am J Respir Crit Care Med 191:804–819. https://doi.org/10.1164/rccm.201406-1031OC
Li Q, Verma IM (2002) NF-κB regulation in the immune system. Nat Rev Immunol 2:725–734. https://doi.org/10.1038/nri910
Li S, Tarlac V, Hamiltn JR (2019) Using PAR4 inhibition as an anti-thrombotic approach: why, how, and when? Int J Mol Sci 20:5629. https://doi.org/10.3390/ijms20225629
Lin CC, Shih CH, Yang YL, Bien MY, Lin CH, Yu MC, Sureshbabu M, Chen BC (2011) Thrombin induces inducible nitric oxide synthase expression via the MAPK, MSK1, and NF-κB signaling pathways in alveolar macrophages. Eur J Pharmacol 672:180–187. https://doi.org/10.1016/j.ejphar.2011.10.005
MacMicking J, Xie QW, Nathan C (1997) Nitric oxide and macrophage function. Annu Rev Immunol 15:323–350. https://doi.org/10.1146/annurev.immunol.15.1.323
McDougall JJ, Zhang C, Cellars L, Joubert E, Dixon CM, Vergnolle N (2009) Triggering of proteinase-activated receptor 4 leads to joint pain and inflammation in mice. Arthritis Rheum 60:728–737. https://doi.org/10.1002/art.24300
Mogensen TH, Melchjorsen J, Höllsberg P, Paludan SR (2003) Activation of NF-κB in virus-infected macrophages is dependent on mitochondrial oxidative stress and intracellular calcium: downstream involvement of the kinases TGF-β-activated kinase 1, mitogen-activated kinase/extracellular signal-regulated kinase kinase 1, and IκB kinase. J Immunol 2003 170(12):6224–6233. https://doi.org/10.4049/jimmunol.170.12.6224
Mosman T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytoxicity assays. J Immunol Methods 65:55–63. https://doi.org/10.1016/0022-1759(83)90303-4
Mosser DM, Edwards JP (2008) Exploring the full spectrum of macrophage activation. Nat Rev Immunol. 8(12):958–969. https://doi.org/10.1038/nri2448
Murray PJ, Wynn TA (2011) Protective and pathogenic functions of macrophage subsets. Nat Rev Immunol. 11:723–737. https://doi.org/10.1038/nri3073
Oikonomopoulou HKK, Saifeddine M, Tea I, Blaber M, Blaber SI, Scarisbrick I, Andrade-Gordon P, Cottrell GS, Bunnett NW, Diamandis EP, Hollenberg MD (2006) Proteinase-activated receptors, targets for kallikrein signaling. J Biol Chem 281:32095–32112. https://doi.org/10.1074/jbc.M513138200
Ramachandran, Hollenberg MD (2008) Proteinases and signalling: pathophysiological and therapeutic implications via PARs and more. Br J Pharmacol 153:5263–5282. https://doi.org/10.1038/sj.bjp.0707507
Rosales C, Uribe-Querol E (2017) Phagocytosis: a fundamental process in immunity. Biomed Res Int 2017:1–18. https://doi.org/10.1155/2017/9042851
Rudinga GR, Khan JG, Kong Y (2018) Protease-activated receptor 4 (PAR4): a promising target for antiplatelet therapy. Int J Mol Sci 19:573. https://doi.org/10.3390/ijms19020573
Rullier A, Senant N, Kisiel W, Bioulac-Sage P, Balabaud C, Le Bail B, Rosenbaum J (2006) Expression of protease-activated receptors and tissue factor in human liver. Virchows Arch 448:46–51. https://doi.org/10.1007/s00428-005-0078-0
Sambrano GR, Huang W, Faruqui T, Mahrus S, Carij C, Coughlin SR (2000) Cathepsin G activates protease-activated receptor-4 in human platelets. J Biol Chem 275:6819–6823. https://doi.org/10.1074/jbc.275.10.6819
Shpacovitch V, Feld M, Hollenberg MD, Luger TA, Steinhoff M (2008) Role of protease-activated receptors in inflammatory responses, innate and adaptive immunity. J Leukoc Biol 83:1309–1322. https://doi.org/10.1189/jlb.0108001
Siqueira IR, Cimarosti H, Fochesatto C, Salbego C, Netto CA (2004) Age-related susceptibility to oxygen and glucose deprivation damage in rat hippocampal slices. Brain Res 1025:226–230. https://doi.org/10.1016/j.brainres.2004.08.005
Slofstra SH, Bijlsma MF, Groot AP, Reitsma PH, Lindhout T, Hugo ten Cate HT, Spek CA (2007) Protease-activated receptor-4 inhibition protects from multiorgan failure in a murine model of systemic inflammation. Blood. 110:3176–3182. https://doi.org/10.1182/blood-2007-02-075440
Tatsumi K, Schmedes CM, Houston ER, Butler E, Mackman N, Antoniak S (2019) Protease-activated receptor 4 protects mice from Coxsackievirus B3 and H1N1 influenza A virus infection. Cell Immunol 344:103949. https://doi.org/10.1016/j.cellimm.2019.103949
Vergnolle N, Derian CK, D'Andrea MR, Steinhoff M, Andrade-Gordon P (2002) Characterization of thrombin-induced leukocyte rolling and adherence: a potential proinflammatory role for proteinase-activated receptor-4. J Immunol 169:1467–1473. https://doi.org/10.4049/jimmunol.169.3.1467
Vu TK, Hung DT, Wheaton VI, Coughlin SR (1991) Molecular cloning of a functional thrombin receptor reveals a novel proteolytic mechanism of receptor activation. Cell. 64:1057–1068. https://doi.org/10.1016/0092-8674(91)90261-V
Xu WF, Andersen H, Whitmore TE, Presnell SR, Yee DP, Ching A, Gilbert T, Davie EW, Foster DC (1998) Cloning and characterization of human protease-activated receptor 4. Proc Natl Acad Sci 95:6642–6646. https://doi.org/10.1073/pnas.95.12.6642
Acknowledgments
This work was supported by Fundacao de Amparo a Pesquisa de Minas Gerais (FAPEMIG/Brazil, grant number PPM-00593-16). AB was a graduate student fellow from CNPq/Brazil, and MTPL is a senior research fellow from CNPq/Brazil.
Author information
Authors and Affiliations
Contributions
AB conceived and conducted experiments, analyzed the data, and wrote the manuscript. KMF and DGM conducted experiments and analyzed the data. PFP and MTPL analyzed the data and contributed new reagents and analytical tools. AK supervised, conceived the experiments, designed research, analyzed the data, and wrote the manuscript. All authors read and approved the manuscript and declare that all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Barra, A., Freitas, K.M., Marconato, D.G. et al. Protease-activated receptor 4 plays a role in lipopolysaccharide-induced inflammatory mechanisms in murine macrophages. Naunyn-Schmiedeberg's Arch Pharmacol 394, 853–862 (2021). https://doi.org/10.1007/s00210-020-02014-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-020-02014-w