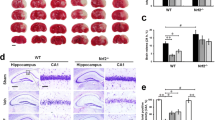
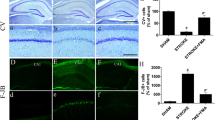
Abstract
With high unmet medical needs, stroke remains an intensely focused research area. Although noscapine is a neuroprotective agent, its mechanism of action in ischemic-reperfusion (I-R) injury is yet to be ascertained. We investigated the effect of noscapine on the molecular mechanisms of cell damage using yeast, and its neuroprotection on cerebral I-R injury in rats. Yeast, both wild-type and Δtrx2 strains, was evaluated for cell growth and viability, and oxidative stress to assess the noscapine effect at 8, 10, 12, and 20 μg/ml concentrations. The neuroprotective activity of noscapine (5 and 10 mg/kg; po for 8 days) was investigated in rats using middle cerebral artery occlusion-induced I-R injury. Infarct volume, neurological deficit, oxidative stress, myeloperoxidase activity, and histological alterations were determined in I-R rats. In vitro yeast assays exhibited significant antioxidant activity and enhanced cell tolerance against oxidative stress after noscapine treatment. Similarly, noscapine pretreatment significantly reduced infarct volume and edema in the brain. The neurological deficit was decreased and oxidative stress biomarkers, superoxide dismutase activity and glutathione levels, were significantly increased while lipid peroxidation showed significant decrease in comparison to vehicle-treated I-R rats. Myeloperoxidase activity, an indicator of inflammation, was also reduced significantly in treated rats; histological changes were attenuated with noscapine. The study demonstrates the protective effect of noscapine in yeast and I-R rats by improving cell viability and attenuating neuronal damage, respectively. This protective activity of noscapine could be attributed to potent free radical scavenging and inhibition of inflammation in cerebral ischemia-reperfusion injury.










Similar content being viewed by others
Data availability
Supplemental original source data presented in form of Excel files, raw images, and pictures.
Abbreviations
- tPA:
-
Tissue plasminogen activator
- FDA:
-
Food and Drug Administration
- MCAO:
-
Middle cerebral artery occlusion
- I-R:
-
Ischemia-reperfusion
- H2O2 :
-
Hydrogen peroxide
- YPD:
-
Yeast extract, peptone, dextrose
- WT:
-
Wild type
- TRX2:
-
Thioredoxin-2
- ROS:
-
Reactive oxygen species
- NOS:
-
Noscapine
- SOD:
-
Superoxide dismutase
- GSH:
-
Glutathione reductase
- LPO:
-
Lipid peroxidation
- MPO:
-
Myeloperoxidaseactivity
- BBB:
-
Blood-brain barrier
- TTC:
-
2,3,5-Triphenyltetrazolium chloride
- DTNB:
-
5,5′-Dithiobis-2-nitrobenzoic acid
- TCA:
-
Trichloroacetic acid
- MDA:
-
Malondialdehyde
- EB:
-
Evans blue
References
Amen T, Kaganovich D (2016) Yeast screening platform identifies FDA-approved drugs that reduce Aβ oligomerization. Microb Cell 3:97–100. https://doi.org/10.15698/mic2016.03.482
Arumugam TV, Selvaraj PK, Woodruff TM, Mattson MP (2008) Targeting ischemic brain injury with intravenous immunoglobulin. Expert Opin Ther Targets 12:19–29. https://doi.org/10.1517/14728222.12.1.19
Bansal S, Sangha KS, Khatri P (2013) Drug treatment of acute ischemic stroke. Am J Cardiovasc Drugs 13:57–69. https://doi.org/10.1007/s40256-013-0007-6
Bond M, Brown R, Rallis C, Bahler J, Mole S (2015) A central role for TOR signalling in a yeast model for juvenile CLN3 disease. Microb Cell 2:466–480. https://doi.org/10.15698/mic2015.12.241
Boussaid AM, Zaouali MA, Hauet T, Ayed KH, Miled AH, Mazgar SG et al (2012) Attenuation of endoplasmic reticulum stresss and mitochondrial injury in kidney with ischemic post conditioning application and trimetazidine treatment. J Biomed Sci 19:17–28. https://doi.org/10.1186/1423-0127-19-71
Bradley PP, Priebat DA, Christensen RD, Rothstein G (1982) Measurement of cutaneous inflammation: estimation of neutrophil content with an enzyme marker. J Invest Dermatol 78:206–209
Broughton BRS, Reutens DC, Sobey CG (2009) Apoptotic mechanisms after cerebral ischemia. Stroke 40:e331–e339
Broussalis E, Trinka E, Killer M, Harrer A, Mccoy M, Kraus J (2012) Current therapies in ischemic stroke. Part B. Future candidates in stroke therapy and experimental studies. Drug DiscovToday 17:671–684. https://doi.org/10.1016/j.drudis.2012.02.011
Carden DL, Granger DN (2000) Pathophysiology of ischemia-reperfusion injury. J Pathol 190:255–266. https://doi.org/10.1002/(SICI)1096-9896(200002)190
Chomova M, Zitnanova I (2016) Look into brain energy crisis and membrane pathophysiology in ischemia and reperfusion. Stress 19:341–348. https://doi.org/10.1080/10253890.2016.1174848
Dani C, Bonatto D, Salvador M, Pereira MD, Henriques JA, Eleutherio E (2008) Antioxidant protection of resveratrol and catechin in Saccharomyces cerevisiae. J Agric Food Chem 56:4268–4272. https://doi.org/10.1021/jf800752s
Ding-Zhou L, Margaill I, Palmier B, Pruneau D, Plotkine M, Marchand-Verrecchia C (2003) LF 16-0687 Ms, a bradykinin B2 receptor antagonist, reduces ischemic brain injury in a murine model of transient focal cerebral ischemia. Br J Pharmacol 139:1539–1547. https://doi.org/10.1038/sj.bjp.0705385
Dirnagl U, Becker K, Meisel A (2009) Preconditioning and tolerance against cerebral ischaemia: from experimental strategies to clinical use. Lancet Neurol 8:398–412. https://doi.org/10.1016/S1474-4422(09)70054-7
Dobrivojević M, Špiranec K, Aleksandra S (2015) Involvement of bradykinin in brain edema development after ischemic stroke. Pflugers Arch 467:201–212. https://doi.org/10.1007/s00424-014-1519-x
Ekshyyan O, Aw TY (2004) Apoptosis in acute and chronic neurological disorders. Front Biosci 9(1567–1576):1567–1576. https://doi.org/10.2741/1357
Elali A, Doeppner TR, Zechariah A, Hermann DM (2011) Increased blood-brain barrier permeability and brain edema after focal cerebral ischemia induced by hyperlipidemia: role of lipid peroxidation and calpain-1/2, matrix metalloproteinase-2/9, and RhoA overactivation. Stroke 42:3238–3244
Foury F (1997) Human genetic diseases: a cross-talk between man and yeast. Gene 195:1–10. https://doi.org/10.1016/s0378-1119(97)00140-6
Goldstein LB (2014) Modern medical management of acute ischemic stroke. Methodist Debakey Cardiovasc J 10:99–104. https://doi.org/10.14797/mdcj-10-2-99
Golla U, Bhimathati SS (2014) Evaluation of antioxidant and DNA damage protection activity of the hydroalcoholic extract of Desmostachya bipinnata L. Stapf. Sci World J 21:5084–5094. https://doi.org/10.1155/2014/215084
Gursoy-ozdemir Y, Bolay H, Saribas O, Dalkara T (2000) Role of endothelial nitric oxide generation and peroxynitrite formation in reperfusion injury after focal cerebral ischemia. Stroke 31:1974–1981. https://doi.org/10.1161/01.str.31.8.1974
Ishrat T, Sayeed I, Atif F, Stein DG (2009) Effects of progesterone administration on infarct volume and functional deficits following permanent focal cerebral ischemia in rats. Brain Res 1257:94–101
Jablonska A, Lukomska B (2011) Stroke induced brain changes: implications for stem cell transplantation. Acta Neurobiol Exp (Wars) 7:74–85
Kalogeris T, Bao Y, Korthuis RJ (2014) Mitochondrial reactive oxygen species: a double edged sword in ischemia/reperfusion vs preconditioning. Redox Biol 2:702–714
Kamoun Y, Mabrouk I, Delahodde A, Boukid F, Yacoubi-Hadj I, Raja A et al (2015) Overexpression of yeast thioredoxin TRX2 reduces p53-mediated cell death in yeast. Appl Microbiol Biotechnol 99:8619–8628
Kawabori M, Yenari MA (2015) Inflammatory responses in brain ischemia. Curr Med Chem 22:1258–1277. https://doi.org/10.2174/0929867322666150209154036
Khanmoradi M, Ali Mard S, Aboutaleb N, Nobakht M, Mahmoudian M (2014) The protective activity of noscapine on renal ischemia-reperfusion injury in male Wistar rat. Iran J Basic Med Sci 17:244–249
Landen JW, Hau V, Wang M, Davis T, Ciliax B, Wainer BH et al (2004) Noscapine crosses the blood-brain barrier and inhibits glioblastoma growth. Clin Cancer Res 10:5187–5201. https://doi.org/10.1158/1078-0432.CCR-04-0360
Longa EZ, Weinstein PR, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniotomy in rats. Stroke 20:84–91. https://doi.org/10.1161/01.str.20.1.84
Lowery OH, Rosenbrough NJ, Farr AI, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Madeo F, Fröhlich E, Ligr M, Grey M, Sigrist SJ, Wolf DH et al (1999) Oxygen stress: a regulator of apoptosis in yeast. J Cell Biol 145:757–767. https://doi.org/10.1083/jcb.145.4.757
Madeo F, Engelhardt S, Herke E, Lehmann N, Maldener C, Proksch A et al (2002) Apoptosis in yeast: a new model system with applications in cell biology and medicine. Curr Genet 41(4):208–216. https://doi.org/10.1007/s00294-002-0310-2
Mahmoudian M, Siadatpour Z, Ziai SA, Mehrpour M, Benaissa F, Nobakht M (2003) Reduction of the prenatal hypoxic-ischemic brain edema with noscapine. Acta Physiol Hung 90:313–318. https://doi.org/10.1556/aphysiol.90.2003.4.4
Mahmoudian M, Rezvani M, Rohani M, Benaissa F, Jalili M, Ghourchian S (2015) A novel effect of noscapine on patients with massive ischemic stroke: a pseudo-randomized clinical trial. Iran J Neurol 14:12–16
Margaritescu O, Mogoanta L, Pirici D, Cernea D, Margaritescu C (2009) Histopathological changes in acute ischemic stroke. Romanian J Morphol Embryol 50:327–339
Marklund S, Marklund G (1974) Involvement of the superoxide anion radical in the autooxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur J Biochem 47:469–474. https://doi.org/10.1111/j.1432-1033.1974.tb03714.x
Ming J, Patrick J, Doherty O, Harvey R, Lyons V, Rogers PJ (2011) An antioxidant screening assay based on oxidant-induced growth arrest in Saccharomyces cerevisiae. FEMS Yeast Res 11:379–387. https://doi.org/10.1111/j.1567-1364.2011.00726.x
Moncada S, Higgs EA (2006) The discovery of nitric oxide and its role in vascular biology. Br J Pharmacol 147:S193–S201. https://doi.org/10.1038/sj.bjp.0706458
Moron MS, Depierre JW, Mannerwik B (1979) Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver. Biochim Biophys Acta 582:67–78. https://doi.org/10.1016/0304-4165(79)90289-7
Moskowitz MA, Lo EH, Iadecola C (2010) The science of stroke: mechanisms in search of treatments. Neuron 67:181–198. https://doi.org/10.1016/j.neuron.2010.07.002
Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M et al (2016) Heart disease and stroke statistics-2016 update a report from the American Heart Association. Circulation 133:e348–e360. https://doi.org/10.1161/CIR.0000000000000350
Nour M, Scalzo F, Liebeskind DS (2013) Ischemia-reperfusion injury in stroke. Interv Neurol 1:185–199. https://doi.org/10.1159/000353125
Ocón E, Garrido, Chris M (2002) Role of thioredoxins in the response of Saccharomyces cerevisiae to oxidative stress induced by hydroperoxides. Mol Microbiol 43:993–1003. https://doi.org/10.1046/j.1365-2958.2002.02795.x
Panicker JN, Thomas M, Pavithran K, Nair D, Sarma PS (2003) Morbidity predictors in ischemic stroke. Neurol India 51:49–51
Rida PC, LiVecche D, Ogden A, Zhou J, Aneja R (2015) The noscapine chronicle: a pharmaco-historic biography of the opiate alkaloid family and its clinical applications. Med Res Rev 35:1072–1096. https://doi.org/10.1002/med.21357
Rogers DC, Campbell CA, Jennifer L, Stretton L, Macky KB (1997) Correlation between motor impairment and infarct volume after permanent and transient middle artery occlusion in rat. Stroke 28:2060–2065. https://doi.org/10.1161/01.str.28.10.2060
Rosenberg GA, Yang Y (2007) Vasogenic edema due to tight junction disruption by matrix metalloproteinases in cerebral ischemia. Neurosurg Focus 22:E4
Roth JM (2011) Recombinant tissue plasminogen activator for the treatment of acute ischemic stroke. Proc (BaylUniv Med Cent) 24:257–259. https://doi.org/10.1080/08998280.2011.11928729
Schwab M, Bauer R, Zwiener U (1997) The distribution of normal brain water content in Wistar rats and its increase due to ischemia. Brain Res 749:82–87. https://doi.org/10.1016/s0006-8993(96)01165-1
Sharma A, Sharma RR (2017) Ischemic cerebro-vascular strokes. Current research aspects on neuroprotection. Austin J Cerebrovasc Dis & Stroke 4:1072
Shrestha A, Megeney L (2015) Yeast proteinopathy models: a robust tool for deciphering the basis of neurodegeneration. Microb Cell 2:458–465. https://doi.org/10.15698/mic2015.12.243
Slater TF, Sawyer BC (1971) The stimulatory effects of carbon tetrachloride and other halogeno alkanes or peroxidative reactions in rat liver fractions in vitro. Biochemistry 123:805–814. https://doi.org/10.1042/bj1230805
Sommer CJ (2017) Ischemic stroke: experimental models and reality. Acta Neuropathol 133:245–261. https://doi.org/10.1007/s00401-017-1667-0
Stekovic S, Ruckenstuhl C, Royer P, Winker-Hermaden C, Carmona-Gutierrez D, Frohlich KU (2017) The neuroprotective steroid progesterone promotes mitochondrial uncoupling, reduces cytosolic calcium and augments stress resistance in yeast cells. Microb Cell 4:191–199. https://doi.org/10.15698/mic2017.06.577
Stella C, Burgos I, Chapela S, Gamondi O (2011) Ischemia - reperfusion: a look from yeast mitochondria. Curr Med Chem 18:3476–3484. https://doi.org/10.2174/092986711796642553
Thiyagarajan M, Sharma SS (2004) Neuroprotective effect of curcumin in middle cerebral artery occlusion induced focal cerebral ischemia in rats. Life Sci 74:968–985. https://doi.org/10.1016/j.lfs.2003.06.042
Vahabzadeh G, Rahbar-Roshandel N, Ebrahimi SA, Mahmoudian M (2015) Neuroprotective effect of noscapine on cerebral oxygen-glucose deprivation injury. Pharmacol Rep 67:281–288. https://doi.org/10.1016/j.pharep.2014.10.011
Veltkamp R, Gill D (2016) Clinical trials of immunomodulation in ischemic stroke. Neurotherapeutics 13:791–800. https://doi.org/10.1007/s13311-016-0458-y
Ying W, Xiong ZG (2010) Oxidative stress and NAD+ in ischemic brain injury: current advances and future perspectives. Curr Med Chem 17:2152–2158. https://doi.org/10.2174/092986710791299911
Young AR, Ali C, Duretete A, Vivien D (2007) Neuroprotection and stroke: time for a compromise. J Neurochem 103:1302–1309. https://doi.org/10.1111/j.1471-4159.2007.04866.x
Acknowledgments
I would like to thank all members of the Chaperone and Stress Biology Lab, Department of Biological Sciences, Institute of Science Education and Research (IISER), Bhopal, MP, India, for the research facilities and their support.
Author information
Authors and Affiliations
Contributions
MK performed the experiments and data analysis and wrote the manuscript. VD, CS, and ASM helped with the data analysis and contributed to the correction of the manuscript. VS and CT contributed to the correction of the manuscript. VD and CS designed and supervised the entire project. All authors read and approved the manuscript and all data were generated in-house and that no paper mill was used.
Corresponding author
Ethics declarations
All animal procedures were approved by the Institutional Animal Ethics Committee of the VNS Institute of Pharmacy, Bhopal (Madhya Pradesh, India) (Protocol No. PH/IAEC/2K14/007).
Conflict of interest
The authors declare that they have no conflicts of interest.
Code availability
Not applicable
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kawadkar, M., Mandloi, A.S., Saxena, V. et al. Noscapine alleviates cerebral damage in ischemia-reperfusion injury in rats. Naunyn-Schmiedeberg's Arch Pharmacol 394, 669–683 (2021). https://doi.org/10.1007/s00210-020-02005-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-020-02005-x