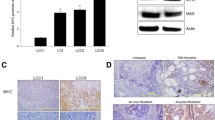
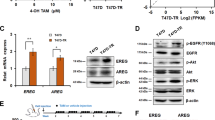
Abstract
Tamoxifen is an estrogen modulator widely used in the treatment of patients with ESR/ER-positive breast cancer; however, resistance limits its clinical application. Autophagy alterations have recently been suggested as a new mechanism for tamoxifen resistance. Glucose transporter 1 (GLUT1) has been reported to be associated with the development and metastasis of breast cancer, but the relationship among GLUT1, autophagy, and endocrine resistance remains unclear. Our present study found that GLUT1 expression and autophagy flux were upregulated in the tamoxifen-resistant breast cancer cell line MCF-7/TAMR-1 and that knockdown of GLUT1 promoted sensitization to tamoxifen. Moreover, knockdown of GLUT1 significantly decreased the enhancement of autophagy flux in tamoxifen-resistant cell lines. Furthermore, inhibiting autophagy in tamoxifen-resistant cells resulted in sensitization to tamoxifen. We conclude that GLUT1 contributes to tamoxifen resistance in breast cancer and that tamoxifen-resistant cells become resensitized to tamoxifen after GLUT1 silencing. These findings suggest GLUT1 as a new factor clinically associated with resistance to tamoxifen.






Similar content being viewed by others
Abbreviations
- Baf-A1:
-
Bafilomycin A 1
- CCK-8:
-
Cell counting Kit-8
- DAB:
-
Diaminobenzidine
- DEGs:
-
Differentially expressed genes
- DFS:
-
Disease-free survival
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- ER/ESR:
-
Estrogen receptor
- FBS:
-
Fetal bovine serum
- FOXO:
-
Forkhead box O
- GAPDH:
-
Glyceraldehyde-3-phosphate dehydrogenase
- GEO:
-
Gene expression omnibus
- GLUT1:
-
Glucose transporter 1
- GO:
-
Gene Ontology
- HRP:
-
Horseradish peroxidase
- IC50:
-
Inhibitory concentration
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- PPI:
-
Protein-protein interaction
- PR:
-
Progesterone receptor
- SEM:
-
Structural equation modeling
- SERMs:
-
Selective ER modulators
- SD:
-
Standard deviation
- siRNA:
-
Small interfering RNA
- siGLUT1:
-
Small interfering GLUT1
- TAM:
-
Tamoxifen
- 4OH-T:
-
4-Hydroxy tamoxifen
References
Kwong D, Davies C, Godwin J et al (2011) Relevance of breast cancer hormone receptors and other factors to the efficacy of adjuvant tamoxifen: patient-level meta-analysis of randomised trials. Lancet 378(9793):771–784
Lee MH, Koh D, Na H, Ka NL, Kim S, Kim HJ, et al. (2017) MTA1 is a novel regulator of autophagy that induces tamoxifen resistance in breast cancer cells. Autophagy
Wood AJJ, Riggs BL, Hartmann LC (2003) Selective estrogen-receptor modulators — mechanisms of action and application to clinical practice. https://doi.org/10.1056/nejmra022219
Viedma-Rodríguezet R et al (2014) Mechanisms associated with resistance to tamoxifen in estrogen receptor-positive breast cancer (review). Oncol Rep 32:3–15. https://doi.org/10.3892/or.2014.3190
Das CK, Mandal M, Kögel D (2018) Pro-survival autophagy and cancer cell resistance to therapy. Cancer Metastasis Rev 37(5):1–18
Samaddar JS, Gaddy VT, Duplantier J, Thandavan SP, Shah M, Smith MJ et al (2009) A role for macroautophagy in protection against 4-hydroxytamoxifen-induced cell death and the development of antiestrogen resistance. Mol Cancer Ther 7(3):2977–2987
Nagelkerke A, Sieuwerts AM, Bussink J, Sweep FCGJ, Look MP, Foekens JA, Martens JWM, Span PN (2014) LAMP3 is involved in tamoxifen resistance in breast cancer cells through the modulation of autophagy. Endocr Relat Cancer 21(1):101–112
Cook KL, Shajahan AN, Wärri A, Jin L, Hilakiviclarke LA, Clarke R (2012) Glucose-regulated protein 78 controls cross-talk between apoptosis and autophagy to determine antiestrogen responsiveness. Autophagy 72(12):3337–3349
Qadir MA, Kwok B, Dragowska WH, To KH, le D, Bally MB, Gorski SM (2008) Macroautophagy inhibition sensitizes tamoxifen-resistant breast cancer cells and enhances mitochondrial depolarization. Breast Cancer Res Treat 112(3):389–403
None (2016) Correction: hydroxychloroquine inhibits autophagy to potentiate antiestrogen responsiveness in er + breast cancer. Clin Cancer Res 22(11):2825–2825
Deng D, Xu C, Sun P, Wu J, Yan C, Hu M, Yan N et al (2014) Crystal structure of the human glucose transporter glut1. Nature 510(7503):121–125
Haber RS, Rathan A, Weiser KR et al (2015) GLUT1 glucose transporter expression in colorectal carcinoma. Cancer 83(1):34–40
Pelicano H, Martin DS, Xu RH, et al (2006) Glycolysis inhibition for anticancer treatment. 25(34):4633-4646
Wang T, Ning K, Lu TX, Hua D (2016) Elevated expression of trpc5 and glut1 is associated with chemoresistance in colorectal cancer. Oncol Reports
Li P, Yang X, Cheng Y, Zhang X, Yang C, Deng X, Li P, Tao J, Yang H, Wei J, Tang J, Yuan W, Lu Q, Xu X, Gu M (2017) MicroRNA-218 increases the sensitivity of bladder cancer to cisplatin by targeting Glut1. Cell Physiol Biochem 41:921–932. https://doi.org/10.1159/000460505
Zhao F, Ming J, Zhou Y et al (2016) Inhibition of Glut1 by WZB117 sensitizes radioresistant breast cancer cells to irradiation. Cancer Chemother Pharmacol 77(5):963–972
Iitaka M, Katayama S (2000) [Insulin resistance in pituitary, thyroid, and adrenal diseases]. Nihon Rinsho Japanese Journal of. Clin Med 58(2):451
Alò PL, Visca P, Botti C, Galati GM, Sebastiani V, Andreano T, di Tondo U, Pizer ES (2001) Immunohistochemical expression of human erythrocyte glucose transporter and fatty acid synthase in infiltrating breast carcinomas and adjacent typical/atypical hyperplastic or normal breast tissue. Am J Clin Pathol 116(1):129–134
Grover-Mckay M, Walsh SA, Seftor EA et al (1998) Role for glucose transporter 1 protein in human breast cancer. Pathol Oncol Res 4(2):115–120
Younes M, Brown RW, Mody DR, Fernandez L, Laucirica R (2015) Glut1 expression in human breast carcinoma: correlation with known prognostic markers. Anticancer Res 15(6B):2895
Yan SX, Luo XM, Zhou SH, Bao YY, Fan J, Lu ZJ, Liao XB, Huang YP, Wu TT, Wang QY (2013) Effect of antisense oligodeoxynucleotides glucose transporter-1 on enhancement of radiosensitivity of laryngeal carcinoma. Int J Med Sci 10(10):1375–1386
Abouzeid AH, Patel NR, Rachman IM, Senn S, Torchilin VP (2013) Anti-cancer activity of anti-GLUT1 antibody-targeted polymeric micelles co-loaded with curcumin and doxorubicin. J Drug Target 21(10):994–1000
Wang (2013) Inhibition of glucose transporter 1 (glut1) chemosensitized head and neck cancer cells to cisplatin. Technol Cancer Res Treatment
Chan DA, Sutphin PD, Nguyen P et al (2011) Targeting GLUT1 and the Warburg effect in renal cell carcinoma by chemical synthetic lethality. Sci Transl Med 3(94):94–94
Shimanishi M, Ogi K, Sogabe Y, Kaneko T, Dehari H, Miyazaki A, Hiratsuka H (2013) Silencing of GLUT-1 inhibits sensitization of oral cancer cells to cisplatin during hypoxia. J Oral Pathol Med 42(5):382–388
Cook KL, Clarke PAG, Parmar J, Hu R, Schwartz-Roberts JL, Abu-Asab M, Wärri A, Baumann WT, Clarke R (2014) Knockdown of estrogen receptor-? induces autophagy and inhibits antiestrogen-mediated unfolded protein response activation, promoting ROS-induced breast cancer cell death. FASEB J 28(9):3891–3905
Hsieh JY, Kuo WW, Lai YP et al (2015) 17β-Estradiol and/or estrogen receptor ? Attenuate the autophagic and apoptotic effects induced by prolonged hypoxia through HIF-1a-mediated BNIP3 and IGFBP-3 signaling blockage. Cell Physiol Biochem 36(1):274–284
Haber RS, Rathan A, Weiser KR, Pritsker A, Itzkowitz SH, Bodian C, Slater G, Weiss A, Burstein DE (1998) GLUT1 glucose transporter expression in colorectal carcinoma : a marker for poor prognosis. Cancer 83(1):34–40
Kawamura T, Kusakabe T, Sugino T et al (2011) Expression of glucose transporter-1 in human gastric carcinoma : association with tumor aggressiveness, metastasis, and patient survival. Cancer 92(3):634–641
Younes M, Brown RW, Stephenson M, Gondo M, Cagle PT (1997) Overexpression of glut1 and glut3 in stage I nonsmall cell lung carcinoma is associated with poor survival. Cancer 80(6):1046–1051
Medina RA, Meneses AM, Vera JC, Guzman C, Nualart F, Rodriguez F, de los Angeles Garcia M, Kato S, Espinoza N, Monso C, Carvajal A, Pinto M, Owen GI (2004) Differential regulation of glucose transporter expression by estrogen and progesterone in Ishikawa endometrial cancer cells. J Endocrinol 182(3):467–478
Das CK, Parekh A, Parida PK, Bhutia SK, Mandal M (2019) Lactate dehydrogenase A regulates autophagy and tamoxifen resistance in breast cancer. Biochim Biophys Acta, Mol Cell Res 1866:1004–1018. https://doi.org/10.1016/j.bbamcr
Osborne CK, Neven P, Dirix LY, Mackey JR, Robert J, Underhill C, Schiff R, Gutierrez C, Migliaccio I, Anagnostou VK, Rimm DL, Magill P, Sellers M (2011) Gefitinib or placebo in combination with tamoxifen in patients with hormone receptor-positive metastatic breast cancer: a randomized phase II study. Clin Cancer Res 17(5):1147–1159
Munster PN, Thurn KT, Thomas S et al (2011) A phase II study of the histone deacetylase inhibitor vorinostat combined with tamoxifen for the treatment of patients with hormone therapy-resistant breast cancer. Breast Dis Year Book Quarterly 104(12):1828–1835
Gammoh N, Lam D, Puente C, Ganley I, Marks PA, Jiang X (2012) Role of autophagy in histone deacetylase inhibitor-induced apoptotic and nonapoptotic cell death[J]. Proc Natl Acad Sci 109(17):6561–6565
Dragowska WH, Weppler SA, Wang JC, Wong LY, Kapanen AI, Rawji JS, Warburton C, Qadir MA, Donohue E, Roberge M, Gorski SM, Gelmon KA, Bally MB (2013) Induction of autophagy is an early response to gefitinib and a potential therapeutic target in breast cancer. PLoS ONE 8:e76503. https://doi.org/10.1371/journal.pone.0076503
Acknowledgments
The authors would like to thank the Oncobiology Key Lab of the Heilongjiang Province Common Institution of Higher Learning.
Funding
The study was supported by the grants from the National Natural Science Foundation of China (81730074).
Author information
Authors and Affiliations
Contributions
Conception and design: QZ, MS. Development of methodology: MX, SZ, and YD. Acquisition of data: MS, SZ, YW, and YM. Analysis and interpretation of data: MS, SZ, and YM. Writing, review, and/or revision of the manuscript: MS, QZ, and YD. Administrative, technical, or material support: QZ, MS, and SZ. Study supervision: HJ. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
All procedures performed involving human participants in our study were in accordance with the ethical standards of the Institutional and/or research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethics statement
All experiments were performed in the Oncobiology Key Lab of the Heilongjiang Province Common Institution of Higher Learning and were not outsourced to companies. No papermill was used.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PPTX 817 kb)
Rights and permissions
About this article
Cite this article
Sun, M., Zhao, S., Duan, Y. et al. GLUT1 participates in tamoxifen resistance in breast cancer cells through autophagy regulation. Naunyn-Schmiedeberg's Arch Pharmacol 394, 205–216 (2021). https://doi.org/10.1007/s00210-020-01893-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-020-01893-3