Abstract
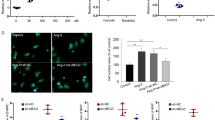
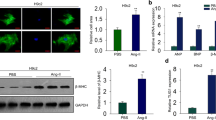
Cardiomyocyte hypertrophy is a fatal factor in heart disease resulting in heart failure and even mortality. Although many studies have been focusing on the pathogenesis of cardiomyocyte hypertrophy, the exact molecular mechanisms are still unexclusive. In this study, we first found that the expression level of lncRNA Tincr was significantly decreased in the myocardial tissues of TAC mouse models of cardiomyocyte hypertrophy, and this result was further confirmed in H9C2 cells, a widely used rat myoblast cell lines. More intriguingly, we demonstrated that the aberration of Tincr is essential to the pathogenesis of cardiomyocyte hypertrophy, indicated by the re-induction of Tincr improving the heart functions of hypertrophic mice. In mechanism, we identified miR-31-5p as a direct target of Tincr using a widely used online bioinformatics tool StarBase, and this result was further experimentally validated using dual-luciferase reporter assay and real-time PCR. Also, we identified PRKCE as a direct target of miR-31-5p, and loss function of miR-31-5p significantly blocks the positive regulatory effect of Tincr on PRKCE expression in H9C2 cells. The knockdown of Tincr resulted in increased cardiomyocyte size, and, however, inhibition of miR-31-5p or overexpression of PRKCE significantly reversed the increased cardiomyocyte size. Taken together, our study showed that a novel Tincr-miR-31-5p axis targeting PRKCE was involved in cardiomyocyte hypertrophy, indicating that it may provide potential therapy in cardiomyocyte hypertrophy.





Similar content being viewed by others
References
Chien KR, Zhu H, Knowlton KU, Miller-Hance W, van-Bilsen M, O'Brien TX, Evans SM (1993) Transcriptional regulation during cardiac growth and development. Annu Rev Physiol 55:77–95
Delpon E, Caballero R, Gomez R, Nunez L, Tamargo J (2005) Angiotensin II, angiotensin II antagonists and spironolactone and their modulation of cardiac repolarization. Trends Pharmacol Sci 26:155–161
Dong H, Hu J, Zou K, Ye M, Chen Y, Wu C, Chen X, Han M (2019) Activation of LncRNA TINCR by H3K27 acetylation promotes Trastuzumab resistance and epithelial-mesenchymal transition by targeting MicroRNA-125b in breast Cancer. Mol Cancer 18:3
Frey N, Olson EN (2003) Cardiac hypertrophy: the good, the bad, and the ugly. Annu Rev Physiol 65:45–79
Frey N, Richardson JA, Olson EN (2000) Calsarcins, a novel family of sarcomeric calcineurin-binding proteins. Proc Natl Acad Sci U S A 97:14632–14637
Han P, Li W, Lin CH, Yang J, Shang C, Nuernberg ST, Jin KK, Xu W, Lin CY, Lin CJ, Xiong Y, Chien H, Zhou B, Ashley E, Bernstein D, Chen PS, Chen HV, Quertermous T, Chang CP (2014) A long noncoding RNA protects the heart from pathological hypertrophy. Nature 514:102–106
Heineke J, Molkentin JD (2006) Regulation of cardiac hypertrophy by intracellular signalling pathways. Nat Rev Mol Cell Biol 7:589–600
Hilfiker-Kleiner D, Shukla P, Klein G, Schaefer A, Stapel B, Hoch M, Muller W, Scherr M, Theilmeier G, Ernst M, Hilfiker A, Drexler H (2010) Continuous glycoprotein-130-mediated signal transducer and activator of transcription-3 activation promotes inflammation, left ventricular rupture, and adverse outcome in subacute myocardial infarction. Circulation 122:145–155
Hunter JJ, Chien KR (1999) Signaling pathways for cardiac hypertrophy and failure. N Engl J Med 341:1276–1283
Jiang F, Zhou X, Huang J (2016) Long non-coding RNA-ROR mediates the reprogramming in cardiac hypertrophy. PLoS One 11:e0152767
Kretz M, Siprashvili Z, Chu C, Webster DE, Zehnder A, Qu K, Lee CS, Flockhart RJ, Groff AF, Chow J, Johnston D, Kim GE, Spitale RC, Flynn RA, Zheng GX, Aiyer S, Raj A, Rinn JL, Chang HY, Khavari PA (2013) Control of somatic tissue differentiation by the long non-coding RNA TINCR. Nature 493:231–235
Kuusisto J, Karja V, Sipola P, Kholova I, Peuhkurinen K, Jaaskelainen P, Naukkarinen A, Yla-Herttuala S, Punnonen K, Laakso M (2012) Low-grade inflammation and the phenotypic expression of myocardial fibrosis in hypertrophic cardiomyopathy. Heart 98:1007–1013
Li JH, Liu S, Zhou H, Qu LH, Yang JH (2014) starBase v2.0: decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA interaction networks from large-scale CLIP-Seq data. Nucleic Acids Res 42:D92–D97
Liu L, An X, Li Z, Song Y, Li L, Zuo S, Liu N, Yang G, Wang H, Cheng X, Zhang Y, Yang X, Wang J (2016) The H19 long noncoding RNA is a novel negative regulator of cardiomyocyte hypertrophy. Cardiovasc Res 111:56–65
Liu Y, Wang Y, He X, Zhang S, Wang K, Wu H, Chen L (2018) LncRNA TINCR/miR-31-5p/C/EBP-alpha feedback loop modulates the adipogenic differentiation process in human adipose tissue-derived mesenchymal stem cells. Stem Cell Res 32:35–42
McMullen JR, Ooi JYY (2017) The interplay of protein coding and non-coding RNAs (circRNAs, lncRNAs) during cardiac differentiation. EBioMedicine 25:9–10
McMurray JJ, Pfeffer MA (2005) Heart failure. Lancet (London, England) 365:1877–1889
Mehra MR, Uber PA, Francis GS (2003) Heart failure therapy at a crossroad: are there limits to the neurohormonal model? J Am Coll Cardiol 41:1606–1610
Militello G, Weirick T, John D, Doring C, Dimmeler S, Uchida S (2017) Screening and validation of lncRNAs and circRNAs as miRNA sponges. Brief Bioinform 18:780–788
Molkentin JD (2004) Calcineurin-NFAT signaling regulates the cardiac hypertrophic response in coordination with the MAPKs. Cardiovasc Res 63:467–475
Molkentin JD (2013) Parsing good versus bad signaling pathways in the heart: role of calcineurin-nuclear factor of activated T-cells. Circ Res 113:16–19
Molkentin JD, Lu JR, Antos CL, Markham B, Richardson J, Robbins J, Grant SR, Olson EN (1998) A calcineurin-dependent transcriptional pathway for cardiac hypertrophy. Cell 93:215–228
Ooi JYY, Bernardo BC, Singla S, Patterson NL, Lin RCY, McMullen JR (2017) Identification of miR-34 regulatory networks in settings of disease and antimiR-therapy: implications for treating cardiac pathology and other diseases. RNA Biol 14:500–513
Ren J, Liu W, Li GC, Jin M, You ZX, Liu HG, Hu Y (2018) Atorvastatin attenuates myocardial hypertrophy induced by chronic intermittent hypoxia in vitro partly through miR-31/PKCepsilon pathway. Current medical science 38:405–412
Savarese G, Lund LH (2017) Global public health burden of heart failure. Cardiac failure review 3:7–11
Shao M, Chen G, Lv F, Liu Y, Tian H, Tao R, Jiang R, Zhang W, Zhuo C (2017) LncRNA TINCR attenuates cardiac hypertrophy by epigenetically silencing CaMKII. Oncotarget 8:47565–47573
Shubeita HE, McDonough PM, Harris AN, Knowlton KU, Glembotski CC, Brown JH, Chien KR (1990) Endothelin induction of inositol phospholipid hydrolysis, sarcomere assembly, and cardiac gene expression in ventricular myocytes. A paracrine mechanism for myocardial cell hypertrophy. J biol chem 265:20555–20562
Sussman MA, Lim HW, Gude N, Taigen T, Olson EN, Robbins J, Colbert MC, Gualberto A, Wieczorek DF, Molkentin JD (1998) Prevention of cardiac hypertrophy in mice by calcineurin inhibition. Science (New York, N.Y.) 281:1690–1693
Thomson DW, Dinger ME (2016) Endogenous microRNA sponges: evidence and controversy. Nat Rev Genet 17:272–283
Viereck J, Kumarswamy R, Foinquinos A, Xiao K, Avramopoulos P, Kunz M, Dittrich M, Maetzig T, Zimmer K, Remke J, Just A, Fendrich J, Scherf K, Bolesani E, Schambach A, Weidemann F, Zweigerdt R, de Windt LJ, Engelhardt S, Dandekar T, Batkai S, Thum T (2016) Long noncoding RNA Chast promotes cardiac remodeling. Sci Transl Med 8:326ra322
Wang K, Liu F, Zhou LY, Long B, Yuan SM, Wang Y, Liu CY, Sun T, Zhang XJ, Li PF (2014) The long noncoding RNA CHRF regulates cardiac hypertrophy by targeting miR-489. Circ Res 114:1377–1388
Wang Y, Men M, Yang W, Zheng H, Xue S (2015) MiR-31 downregulation protects against cardiac ischemia/reperfusion injury by targeting protein kinase C epsilon (PKCepsilon) directly. Cell Physiol Biochem 36:179–190
Wang Z, Zhang XJ, Ji YX, Zhang P, Deng KQ, Gong J, Ren S, Wang X, Chen I, Wang H, Gao C, Yokota T, Ang YS, Li S, Cass A, Vondriska TM, Li G, Deb A, Srivastava D, Yang HT, Xiao X, Li H, Wang Y (2016) The long noncoding RNA Chaer defines an epigenetic checkpoint in cardiac hypertrophy. Nat Med 22:1131–1139
Wang K, Jin W, Song Y, Fei X (2017) LncRNA RP11-436H11.5, functioning as a competitive endogenous RNA, upregulates BCL-W expression by sponging miR-335-5p and promotes proliferation and invasion in renal cell carcinoma. Molecular cancer 16:166
Xu TP, Liu XX, Xia R, Yin L, Kong R, Chen WM, Huang MD, Shu YQ (2015) SP1-induced upregulation of the long noncoding RNA TINCR regulates cell proliferation and apoptosis by affecting KLF2 mRNA stability in gastric cancer. Oncogene 34:5648–5661
Yang JH, Li JH, Shao P, Zhou H, Chen YQ, Qu LH (2011) starBase: a database for exploring microRNA-mRNA interaction maps from Argonaute CLIP-Seq and Degradome-Seq data. Nucleic Acids Res 39:D202–D209
Yang F, Dong A, Mueller P, Caicedo J, Sutton AM, Odetunde J, Barrick CJ, Klyachkin YM, Abdel-Latif A, Smyth SS (2012) Coronary artery remodeling in a model of left ventricular pressure overload is influenced by platelets and inflammatory cells. PLoS One 7:e40196
Zhu XH, Yuan YX, Rao SL, Wang P (2016) LncRNA MIAT enhances cardiac hypertrophy partly through sponging miR-150. Eur Rev Med Pharmacol Sci 20:3653–3660
Funding
This study was supported by the following grants: National Natural Science Foundation of China (grant no.81970204), The National Thirteenth Five-Year Project (grant no.2016YFC0903100), The Science research foundation of Shanxi Province Health and Family Planning Commission (grant no. 2017038 and 2018039), The Central Leading Local Science and Technology Development Special Fund Project (grant no.YDZX20191400004850), The Science research foundation of Shanxi Science and Technology Department (grant no. 201801D221422), 6. Ph.D Programs Foundation of the First Clinical Medical College of Shanxi Medical University (grant no. YB161704), and Key Laboratory of Cellular Physiology (Shanxi Medical University), Ministry of Education.
Author information
Authors and Affiliations
Contributions
Study design: QH. Experiments and data collection: HS, HL, FZ, HX, and YZ. Data analysis and interpretation: HS. Manuscript writing: QH.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare that they have no competing interests.
Ethical approval and consent to participate
All animal experimental procedures in this study were conducted in accordance with the guidelines of the animal ethical committee for animal experimentation in China, and the experimental design was approved by Shanxi Medical University.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, H., Shi, H., Zhang, F. et al. LncRNA Tincr regulates PKCɛ expression in a miR-31-5p-dependent manner in cardiomyocyte hypertrophy. Naunyn-Schmiedeberg's Arch Pharmacol 393, 2495–2506 (2020). https://doi.org/10.1007/s00210-020-01847-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-020-01847-9