Abstract
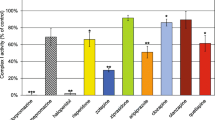
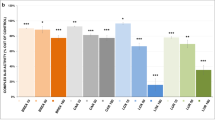
The evaluation of drug-induced mitochondrial impairment may be important in drug development as well as in the comprehension of molecular mechanisms of the therapeutic and adverse effects of drugs. The primary aim of this study was to investigate the effects of four drugs for treatment of depression (bupropion, fluoxetine, amitriptyline, and imipramine) and five drugs for bipolar disorder treatment (lithium, valproate, valpromide, lamotrigine, and carbamazepine) on cell energy metabolism. The in vitro effects of the selected psychopharmaca were measured in isolated pig brain mitochondria; the activities of citrate synthase (CS) and electron transport chain (ETC) complexes (I, II + III, and IV) and mitochondrial respiration rates linked to complex I and complex II were measured. Complex I was significantly inhibited by lithium, carbamazepine, fluoxetine, amitriptyline, and imipramine. The activity of complex IV was decreased after exposure to carbamazepine. The activities of complex II + III and CS were not affected by any tested drug. Complex I-linked respiration was significantly inhibited by bupropion, fluoxetine, amitriptyline, imipramine, valpromide, carbamazepine, and lamotrigine. Significant inhibition of complex II-linked respiration was observed after mitochondria were exposed to amitriptyline, fluoxetine, and carbamazepine. Our outcomes confirm the need to investigate the effects of drugs on both the total respiration rate and the activities of individual enzymes of the ETC to reveal the risk of adverse effects as well as to understand the molecular mechanisms leading to drug-induced changes in the respiratory rate. Our approach can be further replicated to study the mechanisms of action of newly developed drugs.








Similar content being viewed by others
Abbreviations
- AD:
-
Antidepressant
- BAD:
-
Bipolar affective disorder
- COX:
-
Cytochrome c oxidase
- CS:
-
Citrate synthase
- ETC:
-
Electron transport chain
- MPTP:
-
Mitochondrial permeability transition pore
- MS:
-
Mood-stabilizing drug
- NAA:
-
N-Acetyl-aspartate
- NIPEs:
-
Neuron inhibitor with pleotropic effects
- NSMRI:
-
Nonselective monoamine reuptake inhibitor
- OXPHOS:
-
Oxidative phosphorylation
- SSRI:
-
Selective serotonin reuptake inhibitor
References
Abdel-Razaq W, Kendall DA, Bates TE (2011) The effects of antidepressants on mitochondrial function in a model cell system and isolated mitochondria. Neurochem Res 36:327–338. https://doi.org/10.1007/s11064-010-0331-z
Agostinho FR et al (2011a) Treatment with olanzapine, fluoxetine and olanzapine/fluoxetine alters citrate synthase activity in rat brain. Neurosci Lett 487:278–281. https://doi.org/10.1016/j.neulet.2010.10.037
Agostinho FR et al (2011b) Olanzapine plus fluoxetine treatment alters mitochondrial respiratory chain activity in the rat brain. Acta Neuropsychiatr 23:282–291. https://doi.org/10.1111/j.1601-5215.2011.00569.x
Ahmadian E, Babaei H, Mohajjel Nayebi A, Eftekhari A, Eghbal MA (2017) Mechanistic approach for toxic effects of bupropion in primary rat hepatocytes. Drug Res (Stuttg) 67:217–222. https://doi.org/10.1055/s-0042-123034
Aires CC et al (2010) Inhibition of hepatic carnitine palmitoyl-transferase I (CPT IA) by valproyl-CoA as a possible mechanism of valproate-induced steatosis. Biochem Pharmacol 79:792–799. https://doi.org/10.1016/j.bcp.2009.10.011
Anan R et al (1995) Cardiac involvement in mitochondrial diseases. A study on 17 patients with documented mitochondrial DNA defects. Circulation 91:955–961
Ascher JA, Cole JO, Colin JN, Feighner JP, Ferris RM, Fibiger HC, Golden RN, Martin P, Potter WZ, Richelson E (1995) Bupropion: a review of its mechanism of antidepressant activity. J Clin Psychiatry 56:395–401
Benes FM, Matzilevich D, Burke RE, Walsh J (2006) The expression of proapoptosis genes is increased in bipolar disorder, but not in schizophrenia. Mol Psychiatry 11:241–251. https://doi.org/10.1038/sj.mp.4001758
Berger I, Segal I, Shmueli D, Saada A (2010) The effect of antiepileptic drugs on mitochondrial activity: a pilot study. J Child Neurol 25:541–545. https://doi.org/10.1177/0883073809352888
Bousman CA et al (2010) Preliminary evidence of ubiquitin proteasome system dysregulation in schizophrenia and bipolar disorder: convergent pathway analysis findings from two independent samples. Am J Med Genet B Neuropsychiatr Genet 153b:494–502. https://doi.org/10.1002/ajmg.b.31006
Castrén E (2004) Neurotrophic effects of antidepressant drugs. Curr Opin Pharmacol 4:58–64. https://doi.org/10.1016/j.coph.2003.10.004
Cataldo AM et al (2010) Abnormalities in mitochondrial structure in cells from patients with bipolar disorder. Am J Pathol 177:575–585. https://doi.org/10.2353/ajpath.2010.081068
Cebers G, Zhivotovsky B, Ankarcrona M, Liljequist S (1997) AMPA neurotoxicity in cultured cerebellar granule neurons: mode of cell death. Brain Res Bull 43:393–403
Cikankova T, Sigitova E, Zverova M, Fisar Z, Raboch J, Hroudova J (2017) Mitochondrial dysfunctions in bipolar disorder: effect of the disease and pharmacotherapy. CNS Neurol Disord Drug Targets 16:176–186. https://doi.org/10.2174/1871527315666161213110518
Cikankova T, Fisar Z, Bakhouche Y, Luptak M, Hroudova J (2019) In vitro effects of antipsychotics on mitochondrial respiration. Naunyn Schmiedeberg's Arch Pharmacol. https://doi.org/10.1007/s00210-019-01665-8
Costa C et al (2008) Electrophysiology and pharmacology of striatal neuronal dysfunction induced by mitochondrial complex I inhibition. J Neurosci 28:8040–8052. https://doi.org/10.1523/jneurosci.1947-08.2008
de Oliveira MR (2016) Fluoxetine and the mitochondria: a review of the toxicological aspects. Toxicol Lett 258:185–191. https://doi.org/10.1016/j.toxlet.2016.07.001
de Sousa RT et al (2014) Lithium increases leukocyte mitochondrial complex I activity in bipolar disorder during depressive episodes. Psychopharmacology. https://doi.org/10.1007/s00213-014-3655-6
Deuschle M (2013) Effects of antidepressants on glucose metabolism and diabetes mellitus type 2 in adults. Curr Opin Psychiatry 26:60–65. https://doi.org/10.1097/YCO.0b013e32835a4206
Finsterer J, Mahjoub SZ (2012) Mitochondrial toxicity of antiepileptic drugs and their tolerability in mitochondrial disorders. Expert Opin Drug Metab Toxicol 8:71–79. https://doi.org/10.1517/17425255.2012.644535
Fisar Z, Hroudova J, Raboch J (2010) Inhibition of monoamine oxidase activity by antidepressants and mood stabilizers. Neuro Endocrinol Lett 31:645–656
Fisar Z, Hroudova J, Singh N, Maceckova D, Koprivova A (2017) Protocols for high-resolution respirometry experiments to test the activity of electron transfer system of pig brain mitochondria. Indian Journal of Biochemistry & Biophysics 54:258–272
Folbergrova J, Jesina P, Haugvicova R, Lisy V, Houstek J (2010) Sustained deficiency of mitochondrial complex I activity during long periods of survival after seizures induced in immature rats by homocysteic acid. Neurochem Int 56:394–403. https://doi.org/10.1016/j.neuint.2009.11.011
Frey BN et al (2007) Abnormal cellular energy and phospholipid metabolism in the left dorsolateral prefrontal cortex of medication-free individuals with bipolar disorder: an in vivo 1H MRS study. Bipolar Disord 9(Suppl 1):119–127. https://doi.org/10.1111/j.1399-5618.2007.00454.x
Ghaemi SN, Hsu DJ, Soldani F, Goodwin FK (2003) Antidepressants in bipolar disorder: the case for caution. Bipolar Disord 5:421–433
Haddjeri N, Szabo ST, de Montigny C, Blier P (2000) Increased tonic activation of rat forebrain 5-HT(1A) receptors by lithium addition to antidepressant treatments. Neuropsychopharmacology 22:346–356. https://doi.org/10.1016/s0893-133x(99)00138-4
Hroudova J, Fisar Z (2010) Activities of respiratory chain complexes and citrate synthase influenced by pharmacologically different antidepressants and mood stabilizers. Neuro Endocrinol Lett 31:336–342
Hroudova J, Fisar Z (2012) In vitro inhibition of mitochondrial respiratory rate by antidepressants. Toxicol Lett 213:345–352. https://doi.org/10.1016/j.toxlet.2012.07.017
Hyttel J (1994) Pharmacological characterization of selective serotonin reuptake inhibitors (SSRIs). Int Clin Psychopharmacol 9(Suppl 1):19–26
Iwamoto K, Bundo M, Kato T (2005) Altered expression of mitochondria-related genes in postmortem brains of patients with bipolar disorder or schizophrenia, as revealed by large-scale DNA microarray analysis. Hum Mol Genet 14:241–253. https://doi.org/10.1093/hmg/ddi022
Jafarian I, Eskandari MR, Mashayekhi V, Ahadpour M, Hosseini MJ (2013) Toxicity of valproic acid in isolated rat liver mitochondria. Toxicol Mech Methods 23:617–623. https://doi.org/10.3109/15376516.2013.821567
Jang EH, Park CS, Kang JH (2011) Bupropion, an atypical antidepressant, induces endoplasmic reticulum stress and caspase-dependent cytotoxicity in SH-SY5Y cells. Toxicology 285:1–7. https://doi.org/10.1016/j.tox.2011.02.006
Janicak PG (1993) The relevance of clinical pharmacokinetics and therapeutic drug monitoring: anticonvulsant mood stabilizers and antipsychotics. J Clin Psychiatry 54(Suppl):35–41 discussion 55-36
Karanikis P, Korantzopoulos P, Kountouris E, Dimitroula V, Patsouras D, Pappa E, Siogas K (2005) Kearns-Sayre syndrome associated with trifascicular block and QT prolongation. Int J Cardiol 101:147–150. https://doi.org/10.1016/j.ijcard.2004.01.027
Kato T (2006) The role of mitochondrial dysfunction in bipolar disorder. Drug News Perspect 19:597–602. https://doi.org/10.1358/dnp.2006.19.10.1068006
Kato T (2008) Role of mitochondrial DNA in calcium signaling abnormality in bipolar disorder. Cell Calcium 44:92–102. https://doi.org/10.1016/j.ceca.2007.11.005
Kato T, Kunugi H, Nanko S, Kato N (2001) Mitochondrial DNA polymorphisms in bipolar disorder. J Affect Disord 62:151–164
Kim YJ, Ko HH, Han ES, Lee CS (2007) Lamotrigine inhibition of rotenone- or 1-methyl-4-phenylpyridinium-induced mitochondrial damage and cell death. Brain Res Bull 71:633–640. https://doi.org/10.1016/j.brainresbull.2006.12.006
Kim JA, Wei Y, Sowers JR (2008) Role of mitochondrial dysfunction in insulin resistance. Circ Res 102:401–414. https://doi.org/10.1161/circresaha.107.165472
Konradi C, Eaton M, MacDonald ML, Walsh J, Benes FM, Heckers S (2004) Molecular evidence for mitochondrial dysfunction in bipolar disorder. Arch Gen Psychiatry 61:300–308. https://doi.org/10.1001/archpsyc.61.3.300
Lagrue E, Chalon S, Bodard S, Saliba E, Gressens P, Castelnau P (2007) Lamotrigine is neuroprotective in the energy deficiency model of MPTP intoxicated mice. Pediatr Res 62:14–19. https://doi.org/10.1203/PDR.0b013e31806790d7
Lai JS, Zhao C, Warsh JJ, Li PP (2006) Cytoprotection by lithium and valproate varies between cell types and cellular stresses. Eur J Pharmacol 539:18–26. https://doi.org/10.1016/j.ejphar.2006.03.076
Lherm T, Lottin F, Larbi D, Bray M, Legall C, Caen D (2000) Torsade de pointes after poisoning with fluoxetine alone. Presse Med 29:306–307
Liu T et al (2014) Inhibiting mitochondrial Na+/Ca2+ exchange prevents sudden death in a guinea pig model of heart failure. Circ Res 115:44–54. https://doi.org/10.1161/circresaha.115.303062
Macritchie K, Geddes JR, Scott J, Haslam D, de Lima M, Goodwin G (2003) Valproate for acute mood episodes in bipolar disorder. Cochrane Database Syst Rev:CD004052. https://doi.org/10.1002/14651858.CD004052
Maron BJ, Towbin JA, Thiene G, Antzelevitch C, Corrado D, Arnett D, Moss AJ, Seidman CE, Young JB, American Heart Association., Council on Clinical Cardiology, Heart Failure and Transplantation Committee., Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups., Council on Epidemiology and Prevention (2006) Contemporary definitions and classification of the cardiomyopathies: an American Heart Association Scientific Statement from the Council on Clinical Cardiology, Heart Failure and Transplantation Committee; Quality of Care and Outcomes Research and Functional Genomics and Translational Biology Interdisciplinary Working Groups; and Council on Epidemiology and Prevention. Circulation 113:1807–1816. https://doi.org/10.1161/circulationaha.106.174287
Maurer IC, Schippel P, Volz HP (2009) Lithium-induced enhancement of mitochondrial oxidative phosphorylation in human brain tissue. Bipolar Disord 11:515–522. https://doi.org/10.1111/j.1399-5618.2009.00729.x
Munakata K et al (2004) Mitochondrial DNA 3644T-->C mutation associated with bipolar disorder. Genomics 84:1041–1050. https://doi.org/10.1016/j.ygeno.2004.08.015
Ogasahara S, Engel AG, Frens D, Mack D (1989) Muscle coenzyme Q deficiency in familial mitochondrial encephalomyopathy. Proc Natl Acad Sci U S A 86:2379–2382
Pesta D, Gnaiger E (2012) High-resolution respirometry: OXPHOS protocols for human cells and permeabilized fibers from small biopsies of human muscle. Methods Mol Biol (Clifton, NJ) 810:25–58. https://doi.org/10.1007/978-1-61779-382-0_3
Pinna G, Broedel O, Eravci M, Stoltenburg-Didinger G, Plueckhan H, Fuxius S, Meinhold H, Baumgartner A (2003) Thyroid hormones in the rat amygdala as common targets for antidepressant drugs, mood stabilizers, and sleep deprivation. Biol Psychiatry 54:1049–1059
Ponchaut S, Draye JP, Van Hoof F, Veitch K (1991) Loss of hepatic cytochrome aa3 during chronic valproate treatment: dissociation of proton pumping and electron transport in complex IV. Biochem Soc Trans 19:253S
Ponchaut S, van Hoof F, Veitch K (1992) In vitro effects of valproate and valproate metabolites on mitochondrial oxidations. Relevance of CoA sequestration to the observed inhibitions. Biochem Pharmacol 43:2435–2442
Post RM, Weiss SR (2011) Tolerance to the prophylactic effects of carbamazepine and related mood stabilizers in the treatment of bipolar disorders. CNS Neurosci Ther 17:649–660. https://doi.org/10.1111/j.1755-5949.2010.00215.x
Redrobe JP, Bourin M (1999) Evidence of the activity of lithium on 5-HT1B receptors in the mouse forced swimming test: comparison with carbamazepine and sodium valproate. Psychopharmacology 141:370–377
Rustin P, Chretien D, Bourgeron T, Gerard B, Rotig A, Saudubray JM, Munnich A (1994) Biochemical and molecular investigations in respiratory chain deficiencies. Clin Chim Acta 228:35–51
Santos NA, Medina WS, Martins NM, Mingatto FE, Curti C, Santos AC (2008) Aromatic antiepileptic drugs and mitochondrial toxicity: effects on mitochondria isolated from rat liver. Toxicol in Vitro 22:1143–1152. https://doi.org/10.1016/j.tiv.2008.03.004
Silva MF et al (2008) Valproic acid metabolism and its effects on mitochondrial fatty acid oxidation: a review. J Inherit Metab Dis 31:205–216. https://doi.org/10.1007/s10545-008-0841-x
Soeiro-de-Souza MG, Dias VV, Figueira ML, Forlenza OV, Gattaz WF, Zarate CA Jr, Machado-Vieira R (2012) Translating neurotrophic and cellular plasticity: from pathophysiology to improved therapeutics for bipolar disorder. Acta Psychiatr Scand 126:332–341. https://doi.org/10.1111/j.1600-0447.2012.01889.x
Srere (1969) Citrate synthase: [EC 4.1.3.7 citrate oxaloacetate-lyase (CoA acetylating)]. Methods Enzymol 13:3–11
Stork C, Renshaw PF (2005) Mitochondrial dysfunction in bipolar disorder: evidence from magnetic resonance spectroscopy research. Mol Psychiatry 10:900–919. https://doi.org/10.1038/sj.mp.4001711
Strakowski SM, Delbello MP, Adler CM (2005) The functional neuroanatomy of bipolar disorder: a review of neuroimaging findings. Mol Psychiatry 10:105–116. https://doi.org/10.1038/sj.mp.4001585
Taylor V, MacQueen G (2006) Associations between bipolar disorder and metabolic syndrome: a review. J Clin Psychiatry 67:1034–1041. https://doi.org/10.4088/jcp.v67n0704
Trounce IA, Kim YL, Jun AS, Wallace DC (1996) Assessment of mitochondrial oxidative phosphorylation in patient muscle biopsies, lymphoblasts, and transmitochondrial cell lines. Methods Enzymol 264:484–509
Valvassori SS et al (2018) Increased oxidative stress in the mitochondria isolated from lymphocytes of bipolar disorder patients during depressive episodes. Psychiatry Res 264:192–201. https://doi.org/10.1016/j.psychres.2018.03.089
van Reedt Dortland AK, Giltay EJ, van Veen T, Zitman FG, Penninx BW (2010) Metabolic syndrome abnormalities are associated with severity of anxiety and depression and with tricyclic antidepressant use. Acta Psychiatr Scand 122:30–39. https://doi.org/10.1111/j.1600-0447.2010.01565.x
Varriale P (2001) Fluoxetine (Prozac) as a cause of QT prolongation. Arch Intern Med 161:612
Vieweg WV, Wood MA (2004) Tricyclic antidepressants, QT interval prolongation, and torsade de pointes. Psychosomatics 45:371–377. https://doi.org/10.1176/appi.psy.45.5.371
Walden J, Schaerer L, Schloesser S, Grunze H (2000) An open longitudinal study of patients with bipolar rapid cycling treated with lithium or lamotrigine for mood stabilization. Bipolar Disord 2:336–339
Weinbach EC, Costa JL, Nelson BD, Claggett CE, Hundal T, Bradley D, Morris SJ (1986) Effects of tricyclic antidepressant drugs on energy-linked reactions in mitochondria. Biochem Pharmacol 35:1445–1451
Zanetti MV, Otaduy MC, de Sousa RT, Gattaz WF, Busatto GF, Leite CC, Machado-Vieira R (2015) Bimodal effect of lithium plasma levels on hippocampal glutamate concentrations in bipolar II depression: a pilot study. Int J Neuropsychopharmacol:18. https://doi.org/10.1093/ijnp/pyu058
Zarate CA Jr, Singh J, Manji HK (2006) Cellular plasticity cascades: targets for the development of novel therapeutics for bipolar disorder. Biol Psychiatry 59:1006–1020. https://doi.org/10.1016/j.biopsych.2005.10.021
Acknowledgments
The authors thank Zdeněk Hanuš for his assistance.
Funding
This study is supported by a grant GA ČR 17-07585Y from the Grant Agency, Czech Republic, and by a grant 17-05292S from the Czech Science Foundation, Czech Republic.
Author information
Authors and Affiliations
Contributions
JH and ZF conceived and designed the research. TC, ZF, and JH conducted experiments, analyzed data, and wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cikánková, T., Fišar, Z. & Hroudová, J. In vitro effects of antidepressants and mood-stabilizing drugs on cell energy metabolism. Naunyn-Schmiedeberg's Arch Pharmacol 393, 797–811 (2020). https://doi.org/10.1007/s00210-019-01791-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-019-01791-3