Abstract
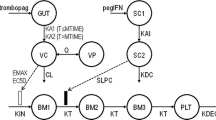
Neutropenia is a hematologic disorder commonly reported in patients with chronic hepatitis C viral (HCV) infection. The objective of the present analysis is to describe the change in neutrophil count resulting from peglated interferon alpha 2-a (PEG-IFN α-2a) therapy in HCV-infected patients. A population pharmacodynamic model will be developed. We also plan to identify patient characteristics that contribute to the development of PEG-IFN α-2a-induced neutropenia in hepatitis C patients. A population pharmacodynamic modeling approach was applied to a cohort of patients (n = 292) with chronic HCV infection. Modeling was performed using NONMEM 6. Data was obtained from two phases III studies sponsored by Hoffmann-La Roche. Covariate screening was applied to evaluate various demographic and clinical characteristics as possible predictors of pharmacodynamic parameter during model development. A total of 4517 neutrophil counts from 292 subjects were analyzed by the proposed population pharmacodynamic model. A constant residual error model was used to the log-transformed neutrophil count. Platelet baseline count and uric acid level were identified as predictors of neutrophil pharmacodynamic model. Increased baseline platelet count is expected to result in higher neutrophil baseline. A higher neutrophil baseline is also expected in patients with increased uric acid level. In conclusion, a mechanistic pharmacodynamic model was developed. The effect of various covariates was included in the model. This allows the prediction of neutrophil count following antiviral therapy in patients with hepatitis C infection. Clinical studies: NV15942 and NV15801





Similar content being viewed by others
References
Alter MJ, Mast EE (1994) The epidemiology of viral hepatitis in the United States. Gastroenterol Clin N Am 23:437–455
Alzubiedi S, Saleh MI (2017) Predictors of severe thrombocytopenia secondary to peginterferon alfa-2a treatment in subjects with hepatitis C virus infection. Am J Ther 24(6):e670–e675
Bekkering FC, Neumann AU, Brouwer JT, Levi-Drummer RS, Schalm SW (2001) Changes in anti-viral effectiveness of interferon after dose reduction in chronic hepatitis C patients: a case control study. BMC Gastroenterol 1:14
Camma C, Giunta M, Andreone P, Craxi A (2001) Interferon and prevention of hepatocellular carcinoma in viral cirrhosis: an evidence-based approach. J Hepatol 34:593–602
Dancey JT, Deubelbeiss KA, Harker LA, Finch CA (1976) Neutrophil kinetics in man. J Clin Invest 58:705–715
Fischbach F, Fischbach FT, Dunning MB (2014) A manual of laboratory and diagnostic tests. Wolters Kluwer Health, Philadelphia
Friberg LE, Henningsson A, Maas H, Nguyen L, Karlsson MO (2002) Model of chemotherapy-induced myelosuppression with parameter consistency across drugs. J Clin Oncol Off J Am Soc Clin Oncol 20:4713–4721
Fried MW, Shiffman ML, Reddy KR, Smith C, Marinos G, Goncales FL Jr, Haussinger D, Diago M, Carosi G, Dhumeaux D, Craxi A, Lin A, Hoffman J, Yu J (2002) Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med 347:975–982
Juarez-Navarro A, Vera-de-Leon L, Navarro J, Chirino-Sprung R, Diaz-Hernandez M, Casillas-Davila L, Dehesa-Violante M (2005) Incidence and severity of infections according to the development of neutropenia during combined therapy with pegylated interferon-a2a plus ribavirin in chronic hepatitis C infection. Methods Find Exp Clin Pharmacol 27:317–322
Lavanchy D (2011) Evolving epidemiology of hepatitis C virus. Clin Microbiol Infect 17:107–115
Nachnani JS, Rao GA, Bulchandani D, Pandya PK, Alba LM (2010) Predictors of hematological abnormalities in patients with chronic hepatitis C treated with interferon and ribavirin. Ann Hematol 89:121–125
Pillay J, den Braber I, Vrisekoop N, Kwast LM, de Boer RJ, Borghans JA, Tesselaar K, Koenderman L (2010) In vivo labeling with 2H2O reveals a human neutrophil lifespan of 5.4 days. Blood 116:625–627
Ruggiero C, Cherubini A, Ble A, Bos AJ, Maggio M, Dixit VD, Lauretani F, Bandinelli S, Senin U, Ferrucci L (2006) Uric acid and inflammatory markers. Eur Heart J 27:1174–1181
Russo F, Bacosi M, Miglioresi L, Ricci GL (2000) Leucopenia is a side effect of combination therapy for hepatitis C infection. Am J Gastroenterol 95:1100–1101
Saleh M (2014) Predictors of long term opioid withdrawal outcome after short-term stabilization with buprenorphine. Eur Rev Med Pharmacol Sci 18:3935–3942
Saleh MI (2016) Clinical predictors associated with warfarin sensitivity. Am J Ther 23:e1690–e1694
Saleh MI (2017) A Bayesian approach for population pharmacokinetic modeling of Pegylated interferon α-2a in hepatitis C patients. Clin Pharmacokinet 56(11):1396–1379
Saleh MI, Obeidat AR, Anter HA, Khanfar AA (2015) Eltrombopag dose predictors in thrombocytopenic subjects with hepatitis C virus infection. Clin Exp Pharmacol Physiol 42:1030–1035
Sheehan V, Weir A, Waters B (2013) Severe neutropenia in patients with chronic hepatitis C: a benign condition. Acta Haematol 129:96–100
Sheehan VA, Weir A, Waters B (2014) Hepatitis C and neutropenia. Curr Opin Hematol 21:58–63
Soto E, Staab A, Tillmann C, Trommeshauser D, Fritsch H, Munzert G, Troconiz IF (2010) Semi-mechanistic population pharmacokinetic/pharmacodynamic model for neutropenia following therapy with the Plk-1 inhibitor BI 2536 and its application in clinical development. Cancer Chemother Pharmacol 66:785–795
Stroup TS, Lieberman JA (2010) Antipsychotic trials in schizophrenia: the CATIE project. Cambridge University Press, Cambridge
Takama H, Tanaka H, Nakashima D, Ueda R, Takaue Y (2006) Population pharmacokinetics of intravenous busulfan in patients undergoing hematopoietic stem cell transplantation. Bone Marrow Transplant 37:345–351
Wang B, Ludden TM, Cheung EN, Schwab GG, Roskos LK (2001) Population pharmacokinetic-pharmacodynamic modeling of filgrastim (r-metHuG-CSF) in healthy volunteers. J Pharmacokinet Pharmacodyn 28:321–342
Wang X, Gao F, Yuan G, Shi K, Huang Y, Chen Y, Qiu R, Sun L, Liu J, Hu C, Zhou Y (2016) Ten-year follow-up analysis of chronic hepatitis C patients after getting sustained virological response to pegylated interferon-alpha and ribavirin therapy. J Viral Hepat 23(12):971–976
Acknowledgments
All authors approved the final version of the article, including the authorship list.
Author contribution statement
MS and NH conceived and designed research. MS developed NONMEM code and designed modeling experiment. NH analyzed data. MS and NH wrote the manuscript. All authors read and approved the manuscript.
Funding
No funding has been received for the conduct of this study and/or preparation of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Saleh, M.I., Hindi, N.N. A population pharmacodynamic model characterizing neutropenia associated with pegylated interferon alpha 2-a therapy in patients with chronic hepatitis C viral infection. Naunyn-Schmiedeberg's Arch Pharmacol 391, 953–963 (2018). https://doi.org/10.1007/s00210-018-1517-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-018-1517-1