Abstract
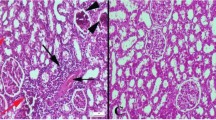
The objective of the present study was to investigate the effects of syringic acid (SA), a phenolic acid, on Nω-nitro-l-arginine methyl ester (l-NAME)-induced hypertensive rats. Hypertension was induced in adult male albino rats by oral administration of l-NAME (40 mg/kg/day) dissolved in drinking water daily for 4 weeks. Rats were treated with different doses of SA (25, 50, and 100 mg/kg body weight (b.w.)). Systolic blood pressure of control and experimental rats was recorded. Plasma nitric oxide metabolites (NOx), lipid peroxidative products such as thiobarbituric acid reactive substances, lipid hydroperoxides, conjugated dienes, and antioxidants such as superoxide dismutase, catalase, glutathione peroxidase, vitamin C, vitamin E, and reduced glutathione were estimated in erythrocytes, plasma, and tissues of experimental rats. Hepatic marker enzymes such as aspartate aminotransferase, alanine aminotransferase, and alkaline phosphatase and renal functional markers such as urea, uric acid, and creatinine were also estimated in serum. The increased levels of blood pressure, lipid peroxidation products, hepatic and renal function markers, and the decreased level of NOx and antioxidants in l-NAME-induced hypertensive rats were reversed upon SA treatment. The protective effect at the dose of the three tested doses (25, 50, and 100 mg/kg) of SA at a dose of 50 mg/kg b.w. exerts optimum protection. Biochemical findings are substantiated by the histological observation. The protective effects of SA are mediated by reducing oxidative stress and retaining the bioavailability of NO in the cardiovascular system.



Similar content being viewed by others
Abbreviations
- ALP:
-
Alkaline phosphatase
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- CAT:
-
Catalase
- CD:
-
Conjugated dienes
- CVD:
-
Cardiovascular diseases
- GPx:
-
Glutathione peroxidase
- GSH:
-
Reduced glutathione
- l-NAME:
-
Nω-nitro-l-arginine methyl ester
- LOOH:
-
Lipid hydroperoxides
- NO:
-
Nitric oxide
- NOS:
-
Nitric oxide synthase
- NOx:
-
Nitric oxide metabolites
- ROS:
-
Reactive oxygen species
- SA:
-
Syringic acid
- SBP:
-
Systolic blood pressure
- SOD:
-
Superoxide dismutase
- TBARS:
-
Thiobarbituric acid reactive substances
References
Abidi P, Afaq F, Arif JM, Lohani M, Rahman Q (1999) Chrysotile-mediated imbalance in the glutathione redox system in the development of pulmonary injury. Toxicol Lett 106:31–39
Baylis C, Mitruka B, Deng A (1992) Chronic blockade of nitric oxide synthesis in the rat produces systemic hypertension and glumerular damage. J Clin Invest 90:278–281
Bech JN, Nielsen CB, Ivarsen P, Jensen KT, Pedersen EB (1998) Dietary sodium affects systemic and renal hemodynamic response to NO inhibition in healthy humans. Am J Physiol 274:914–923
Burtis CA, Ashwood ER (1996) Tietz fundamentals of clinical chemistry. Saunders Company, London, p 881
Cayatte AJ, Palacino JJ, Horten K, Cohen RA (1994) Chronic inhibition of nitric oxide production accelerates neointima formation and impairs endothelial function in hypercholesterolemic rabbits. Arterioscler Thromb 14:753–759
Dryden GW Jr, Deaciuc I, Arteel G, McClain CJ (2005) Clinical implications of oxidative stress and antioxidant therapy. Curr Gastroenterol Rep 7:308–316
Gu D, Reynolds K, Wu X, Chen J, Duan X, Muntner P, Huang G, Reynolds RF, Su S, Whelton PK, He J, InterASIA Collaborative Group, The International Collaborative Study of Cardiovascular Disease in ASIA (2002) Prevalence, awareness, treatment, and control of hypertension in China. Hypertension 40:920–927
Guimaraes CM, Giao MS, Martinez SS, Pintado AI, Pintado ME, Bento Malcata FX (2007) Antioxidant activity of sugar molasses, including protective effect against DNA oxidative damage. J Food Sci 72:39–43
Hamberg M, Svensson J, Wakabayashi T, Samuelsson B (1974) Isolation and structure of two prostaglandins endoperoxides that cause platelet aggregation. Proc Nat Acad Sci 71:345–359
Harrison DG (1997) Cellular and molecular mechanisms of endothelial cell dysfunction. J Clin Invest 100:2153–2157
Hirota A, Taki S, Kawaii S, Yano M, Abe N (2000) 1,1-Diphenyl-2-picrylhydrazyl radical-scavenging compounds from soybean miso and antiproliferative activity of isoflavones from soybean miso toward the cancer cell lines. Biosci Biotechnol Biochem 64:1038–1040
Hoetzel A, Welle A, Schmidt R, Loop T, Humar M, Ryter SW, Geiger KK, Choi AM, Pannen BH (2008) Nitric oxide-deficiency regulates hepatic heme oxygenase-1. Nitric Oxide 18:61–69
Huang PL, Huang Z, Mashimo H, Bloch KD, Moskowitz MA, Bevan JA, Fishman MC (1995) Hypertension in mice lacking the gene for endothelial nitric oxide synthase. Nature 377:239–242
Ignarro LJ, Napoli C, Loscalzo J (2002) Nitric oxide donors and cardiovascular agents modulating the bioactivity of nitric oxide: an overview. Circ Res 90:21–28
Itoh A, Isoda K, Kondoh M, Kawase M, Kobayashi M, Tamesada M, Yagi K (2009) Hepatoprotective effect of syringic acid and vanillic acid on concanavalin A-induced liver injury. Biol Pharm Bull 32:1215–1219
Itoh A, Isoda K, Kondoh M, Kawase M, Watari A, Kobayashi M, Tamesada M, Yagi K (2010) Hepatoprotective effect of syringic acid and vanillic acid on CCl4-induced liver injury. Biol Pharm Bull 33:983–987
Jalili T, Carlstrom J, Kim S, Freeman D, Jin H, Wu TC, Litwin SE, David Symons J (2006) Quercetin-supplemented diets lower blood pressure and attenuate cardiac hypertrophy in rats with aortic constriction. J Cardiovasc Pharmacol 47:531–541
Jiang ZY, Hunt JV, Wolff SP (1992) Ferrous ion oxidation in the presence of xylenol orange for detection of lipid hydroperoxide in low density lipoprotein. Anal Biochem 202:384–389
Kampa M, Alexaki VI, Notas G, Nifli AP, Nistikaki A, Hatzoglou A, Bakogeorgou E, Kouimtzoglou E, Blekas G, Boskou D, Gravanis A, Castanas E (2004) Antiproliferative and apoptotic effects of selective phenolic acids on T47D human breast cancer cells: potential mechanisms of action. Breast Cancer Res 6:63–74
Khokhar S, Apenten RKO (2003) Iron binding characteristics of phenolic compounds: some tentative structure–activity relations. Food Chem 81:133–140
Kumar S, Prahalathan P, Raja B (2011) Antihypertensive and antioxidant potential of vanillic acid, a phenolic compound in L-NAME induced hypertensive rats: a dose-dependence study. Redox Rep 16:208–215
Kumar S, Saravanakumar M, Raja B (2010) Efficacy of piperine, an alkaloidal constituent of pepper on nitric oxide, antioxidants and lipid peroxidation markers in L-NAME induced hypertensive rats. Int J Res Pharm Sci 3:300–307
Loria P, Lonardo A, Carulli L, Verrone AM, Ricchi M, Lombardini S, Rudilosso A, Ballestri S, Carulli N (2005) The metabolic syndrome and non-alcoholic fatty liver disease. Aliment Pharmacol Ther 22:31–36
Mahdi AA (2002) Free radicals and other antioxidants. A textbook of biochemistry by S.P. Singh, thirdth edn. CBS, New Delhi, pp 545–555
Mates JM, Sanchez-Jimenez F (1999) Antioxidant enzymes and their implications in pathophysiologic processes. Front Biosci 4:339–345
Mattila P, Kumpulainen J (2002) Determination of free and total phenolic acids in plant-derived foods by HPLC with diode-array detection. J Agric Food Chem 50:3660–3667
Mittal BV, Singh AK (2010) Hypertension in the developing world: challenges and opportunities. Am J Kidney Dis 55:590–598
Nakmareong S, Kukongviriyapan U, Pakdeechote P, Donpunha W, Kukongviriyapan V, Kongyingyoes B, Sompamit K, Phisalaphong C (2011) Antioxidant and vascular protective effects of curcumin and tetrahydrocurcumin in rats with L-NAME induced hypertension. Naunyn-Schmied Arch Pharmacol 383:519–529
Nguelefack-Mbuyoa PE, Nguelefackb TB, Dongmoc B, Afkird S, Azebazee AGB, Dimoa T, Legssyerd A, Kamanyi A, Ziyyat A (2008) Anti-hypertensive effects of the methanol/methylene chloride stem bark extract of mammea africana in L-NAME-induced hypertensive rats. J Ethnopharmacol 117:446–450
Niehaus WG, Samuelson B (1968) Formation of malondialdehyde from phospholipid arachidonate during microsomal lipid peroxidation. Eur J Pharmacol 6:126–130
Pechanova O, Bernatova I, Babal P, Martinez MC, Kysela S, Stvrtina S, Andriantsitohaina R (2004) Red wine polyphenols prevent cardiovascular alterations in L-NAME-induced hypertension. J Hyperten 22:1551–1559
Pereira LM, Bezerra DG, Machado DL, Mandarim-de-lacerda CA (2004) Enalapril attenuates cardiorenal damage in nitric-oxide-deficient spontaneously hypertensive rats. Clin Sci 106:337–343
Prahalathan P, Kumar S, Raja B (2012) Morin a bioflavonoid, rescues kidney and heart from DOCA-salt hypertensive rats: a biochemical and histopathological study. Metabolism 61:1087–1099
Pushpavalli G, Veeramani C, Pugalendi KV (2010) Influence of chrysin on hepatic marker enzymes and lipid profile against d-galactosamine-induced hepatotoxicity rats. Food Chem Toxicol 48:1654–1659
Quam L, Smith R, Yach D (2006) Rising to the global challenge of the chronic disease epidemic. Lancet 368:1221–1223
Ramachandran V, Raja B (2010) Protective effects of syringic acid against acetaminophen induced hepatic damage in albino rats. J Basic Clin Physiol Pharmacol 21:369–385
Ramachandran V, Saravanan R, Raja B (2011) Attenuation of oxidative stress by syringic acid on acetaminophen-induced nephrotoxic rats. Comp Clin Pathol. doi:10.1007/s00580-011-1327-z
Rao KS, Recknagel RO (1968) Early onset of lipid peroxidation in rat liver after carbon tetrachloride administration. Exp Mol Pathol 9:271–278
Retelny VS, Neuendorf A, Roth JL (2008) Nutrition protocols for the prevention of cardiovascular disease. Nutr Clin Pract 23:468–476
Rice-Evans CA, Miller NJ, Paganga G (1996) Structure-antioxidant activity relationships of flavonoids and phenolic acids. Free Radic Biol Med 20:933–956
Rosselli M, Keller PJ, Dubey RK (1998) Role of nitric oxide in the biology, physiology and pathophysiology of reproduction. Hum Reprod Update 4:3–24
Saravanakumar M, Kumar S, Raja B (2010) Antihypertensive and antioxidant potential of borneol - a natural terpene in L-NAME-induced hypertensive rats. Int J Pharma Biol Arch 1:271–279
Saravanakumar M, Raja B (2011) Veratric acid, a phenolic acid attenuates blood pressure and oxidative stress in L-NAME induced hypertensive rats. Eur J Pharma 671:87–94
Senthil S, Chandramohan G, Pugalendi KV (2007) Isomers (oleanolic and ursolic acids) differ in their protective effect against isoproterenol-induced myocardial ischemia in rats. Int J Cardiol 119:131–133
Sharifi AM, Akbarloo N, Darabi R (2005) Investigation of local ACE activity and structural alterations during development of L-NAME-induced hypertension. Pharmacol Res 52:438–444
Silambarasan T, Raja B (2012) Diosmin, a bioflavonoid reverses alterations in blood pressure, nitric oxide, lipid peroxides and antioxidant status in DOCA-salt induced hypertensive rats. Eur J Pharm 679:81–89
Taniyama Y, Griendling KK (2003) Reactive oxygen species in the vasculature: molecular and cellular mechanism. Hypertension 42:1075–1081
Toba H, Nakagawa Y, Miki S, Shimizu T, Yoshimura A, Inoue R, Asayama J, Kobara M, Nakata T (2005) Calcium channel blockades exhibit anti-inflammatory and antioxidative effects by augmentation of endothelial nitric oxide synthase and the inhibition of angiotensin converting enzyme in the NG-Nitro-L-arginine methyl ester-induced hypertensive eat aorta: vasoprotective effects beyond the blood pressure-lowering effects of amlodipine and manidipine. Hypertens Res 28:689–700
Ulker S, McKeown PP, Bayraktutan U (2003) Vitamins reverse endothelial dysfunction through regulation of eNOS and NAD(P)H oxidase activities. Hypertension 41:534–539
Veeramani C, Aristatle B, Pushpavalli G, Pugalendi KV (2011) Effects of Melothria maderaspatana leaf extract on antioxidant status in sham-operated and uninephrectomized DOCA-salt hypertensive rats. Saudi J Biolog Sci 18:99–105
Wenz G, Han BH, Muller A (2006) Experimental and theoretical studies on the inclusion complexation of syringic acid with α, β, γ and Heptakis (2, 6-di-O-methyl)-β-cyclodextrin. Chem Res 106:782–817
Wu XY, Liu YH, Sheng W, Sun J, Qin G (1997) Chemical constituents of Isatis indigotica. Planta Med 63:55–57
Yu BP (1994) Cellular defenses against damage from reactive oxygen species. Physiol Rev 74:139–162
Acknowledgments
We thank Annamalai University for the financial assistance in the form of “University Research Fellowship” to Mr. S. Kumar. The authors wish to record their sincere thanks to Dr. P. Viswanathan, Professor, Department of Pathology, Rajah Muthiah Medical College and Hospital, Annamalai University, Tamilnadu, India for his assistance in histopathological studies.
Conflict of interest
We declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kumar, S., Prahalathan, P. & Raja, B. Syringic acid ameliorates l-NAME-induced hypertension by reducing oxidative stress. Naunyn-Schmiedeberg's Arch Pharmacol 385, 1175–1184 (2012). https://doi.org/10.1007/s00210-012-0802-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-012-0802-7