Abstract
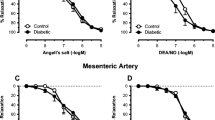
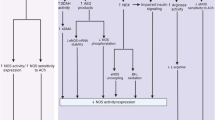
Diabetes is a risk factor of ischemic heart disease, cerebral ischemia, and atherosclerosis, in which endothelial dysfunction plays a role in the pathogenesis. We examined vascular responses in the aorta of pre-diabetic db/db mice with normoglycemia, hyperlipidemia, and hyperinsulinemia (6 weeks old), and diabetic db/db mice with hyperglycemia, hyperlipidemia, and hyperinsulinemia (11 weeks old) in comparison with age-matched non-diabetic db/+ mice. Prostaglandin F2α (PGF2α)-induced contraction was significantly enhanced in the aorta of diabetic but not pre-diabetic db/db mice compared to age-matched non-diabetic db/+ mice. Acetylcholine (ACh), adenosine-5′-diphosphate (ADP), NaF, a G protein activator and A-23187, a Ca-ionophore, caused endothelium-dependent and nitric oxide (NO)-mediated relaxation, and sodium nitroprusside (SNP), an NO donor, caused endothelium-independent relaxation in the pre-contracted aorta of db/db mice. Maximal endothelium-dependent ACh-induced relaxation was reduced in diabetic but not pre-diabetic db/db mice compared to age-matched db/+ mice, while maximal SNP-induced relaxation was not different between diabetic and non-diabetic mice. ACh-induced relaxation in diabetic db/db mice was not affected by ozagrel, a thromboxane A2 (TXA2) synthetase inhibitor, or acetylsalicylic acid (aspirin), a cyclooxygenase inhibitor, suggesting no involvement of endogenous TXA2 or prostanoids in the reduction of relaxation. Maximal endothelium-dependent ADP-, A-23187-, and NaF-induced relaxation was not reduced in diabetic db/db mice. EC50 values for ACh- and SNP-induced relaxation were increased in diabetic but not pre-diabetic db/db mice, suggesting decreases in sensitivity to NO in diabetic mice. Two-week treatment with KV-5070, a PPARγ agonist, lowered plasma glucose, triglyceride (TG), and insulin but not cholesterol, and reversed the reduced ACh-induced relaxation. In conclusion, ACh-induced endothelium-dependent relaxation is impaired in diabetic db/db mice, probably due to the dysfunction of ACh receptors and/or receptor–G protein coupling. Endothelial dysfunction was not genetic and was considered to be initiated primarily by hyperglycemia, and was improved by anti-diabetic treatment with a PPARγ agonist.







Similar content being viewed by others
References
Akbari CM, Saouaf R, Barnhill DF, Newman PA, LoGerfo FW, Veves A (1998) Endothelium-dependent vasodilatation is impaired in both microcirculation and macrocirculation during acute hyperglycemia. J Vasc Surg 28:687–694
Bohlen HG, Lash JM (1993) Topical hyperglycemia rapidly suppresses EDRF-mediated vasodilation of normal rat arterioles. Am J Physiol 265:H219–H225
Buvinic S, Briones R, Huidobro-Toro JP (2002) P2Y1 and P2Y2 receptors are coupled to the NO/cGMP pathway to vasodilate the rat arterial mesenteric bed. Br J Pharmacol 136:847–856
Cameron NE, Cotter MA (1992) Impaired contraction and relaxation in aorta from streptozotocin-diabetic rats: role of polyol pathway. Diabetologia 35:1011–1019
Chinen I, Shimabukuro M, Yamakawa K, Higa N, Matsuzaki T, Noguchi K, Ueda S, Sakanashi M, Takasu N (2007) Vascular lipotoxicity: endothelial dysfunction via fatty-acid-induced reactive oxygen species overproduction in obese Zucker diabetic fatty rats. Endocrinology 148:160–165
Chiou WF, Shum AY, Liao JF, Chen CF (1997) Studies of the cellular mechanisms underlying the vasorelaxant effects of rutaecarpine, a bioactive component extracted from an herbal drug. J Cardiovasc Pharmacol 29:490–498
Flavahan NA, Vanhoutte PM (1990) Pertussis toxin inhibits endothelium-dependent relaxations evoked by fluoride. Eur J Pharmacol 178:121–124
Guerci B, Kearney-Schwartz A, Bohme P, Zannad F, Drouin P (2001a) Endothelial dysfunction and type 2 diabetes. Part 1: physiology and methods for exploring the endothelial function. Diabetes Metab (Paris) 27:425–434
Guerci B, Bohme P, Kearney-Schwartz A, Zannad F, Drouin P (2001b) Endothelial dysfunction and type 2 diabetes. Part 2: altered endothelial function and the effects of treatment in type 2 diabetes mellitus. Diabetes Metab (Paris) 27:436–447
Guo Z, Su W, Allen S, Pang H, Daugherty A, Smart E, Gong MC (2005) Cox-2 up-regulation and vascular smooth muscle contractile hyperreactivity in spontaneous diabetic db/db mice. Cardiovasc Res 67:723–735
Howarth AG, Wiehler WB, Pannirselvam M, Jiang Y, Berger JP, Severson D, Anderson TJ, Triggle CR (2006) A nonthiazolidinedione peroxisome proliferator-activated receptor gamma agonist reverses endothelial dysfunction in diabetic (db/db −/−) mice. J Pharmacol Exp Ther 316:364–370
Kamata K, Kojima S (1997) Characteristics of contractile responses of aorta to norepinephrine in db/db mice. Res Commun Mol Pathol Pharmacol 96:319–328
Kamata K, Nakajima M, Sugiura M (1999) Effects of superoxide dismutase on the acetylcholine-induced relaxation response in cholesterol-fed and streptozotocin-induced diabetic mice. J Smooth Muscle Res 35:33–46
Khurana S, Chacon I, Xie G, Yamada M, Wess J, Raufman J-P, Kennedy RH (2004) Vasodilatory effects of cholinergic agonists are greatly diminished in aorta from M3R−/− mice. Eur J Pharmacol 493:127–132
Kurahashi K, Shirahase H, Nakamura S, Tarumi T, Koshino Y, Wang A-M, Nishihashi T, Shimizu Y (2001) Nicotine-induced contraction in the rat coronary artery: possible involvement of the endothelium, reactive oxygen species and COX-1 metabolites. J Cardiovasc Pharmacol 38(Suppl 1):S21–S25
Kwon SC (2001) Mechanisms of acetylcholine-induced vasorelaxation in high K+-stimulated rabbit renal arteries. J Vet Med Sci 63:41–44
Lam TY, Seto SW, Lau YM, Au LS, Kwan YW, Ngai SM, Tsui KW (2006) Impairment of the vascular relaxation and differential expression of caveolin-1 of the aorta of diabetic +db/+db mice. Eur J Pharmacol 546:134–141
Matsui H, Kobayashi H, Azukizawa S, Kasai M, Yoshimi A, Shirahase H (2001) Novel heterocyclic compounds and salts thereof and medicinal use of the same. World Intellectual Property Organization, Patent WO 01/40192 A1, Kyoto Pharmaceutical Industries, Ltd., Kyoto Japan
Matsumoto T, Kakami M, Noguchi E, Kobayashi T, Kamata K (2007) Imbalance between endothelium-derived relaxing and contracting factors in mesenteric arteries from aged OLETF rats, a model of type 2 diabetes. Am J Physiol Heart Circ Physiol 293:H1480–H1490
Miller VM, Flavahan NA, Vanhoutte PM (1991) Pertussis toxin reduces endothelium-dependent and independent responses to alpha-2-adrenergic stimulation in systemic canine arteries and veins. J Pharmacol Exp Ther 257:290–293
Natali A, Toschi E, Baldeweg S, Casolaro A, Baldi S, Sironi AM, Yudkin JS, Ferrannini E (2005) Haematocrit, type 2 diabetes, and endothelium-dependent vasodilatation of resistance vessels. Eur Heart J 26:464–471
Okon EB, Chung AWY, Zhang H, Laher I, van Breemen C (2007) Hyperglycemia and hyperlipidemia are associated with endothelial dysfunction during the development of type 2 diabetes. Can J Physiol Pharm 85:562–567
Pannirselvam M, Wiehler WB, Anderson T, Triggle CR (2005) Enhanced vascular reactivity of small mesenteric arteries from diabetic mice is associated with enhanced oxidative stress and cyclooxygenase products. Br J Pharmacol 144:953–960
Piercy V, Taylor SG (1998) A comparison of spasmogenic and relaxant responses in aortae from C57/BL/KsJ diabetic mice with those from their non-diabetic litter mates. Pharmacology 56:267–275
Pirotton S, Communi D, Motte S, Janssens R, Boeynaems JM (1996) Endothelial P2-purinoceptors: subtypes and signal transduction. J Auton Pharmacol 16:353–356
Samata K, Kimura T, Satoh S, Watanabe H (1986) Chemical removal of the endothelium by saponin in the isolated dog femoral artery. Eur J Pharmacol 128:85–91
Shibano T, Codina J, Birnbaumer L, Vanhoutte PM (1992) Guanosine 5′-O-(3-thiotriphoaphate) causes endothelium-dependent, pertussis toxin-sensitive relaxations in porcine coronary arteries. Biochem Biophys Res Commun 189:324–329
Shige H, Ishikawa T, Suzukawa M, Ito T, Nakajima K, Higashi K, Ayaori M, Tabata S, Ohsuzu F, Nakamura H (1999) Endothelium-dependent flow-mediated vasodilation in the postprandial state in type 2 diabetes mellitus. Am J Cardiol 84:1272–1274
Skyrme-Jones RA, O’Brien RC, Luo M, Meredith IT (2000) Endothelial vasodilator function is related to low-density lipoprotein particle size and low-density lipoprotein vitamin E content in type 1 diabetes. J Am Coll Cardiol 35:292–299
Taylor PD, Poston L (1994) The effect of hyperglycemia on function of rat isolated mesenteric resistance artery. Br J Pharmacol 113:801–808
Watts GF, O’Brien SF, Silvester W, Millar JA (1996) Impaired endothelium-dependent and independent dilatation of forearm resistance arteries in men with diet-treated non-insulin-dependent diabetes: role of dyslipidaemia. Clin Sci (London) 91:567–573
Zanchi A, Delacretaz E, Taleb V, Gaillard R, Jeanrenaud B, Brunner HR, Waeber B (1995) Endothelial function of the mesenteric arteriole and mechanical behaviour of the carotid artery in rats with insulin resistance and hypercholesterolaemia. J Hypertens 13:1463–1470
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Miike, T., Kunishiro, K., Kanda, M. et al. Impairment of endothelium-dependent ACh-induced relaxation in aorta of diabetic db/db mice—possible dysfunction of receptor and/or receptor–G protein coupling. Naunyn-Schmied Arch Pharmacol 377, 401–410 (2008). https://doi.org/10.1007/s00210-008-0261-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00210-008-0261-3