Abstract
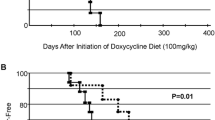
There is an ongoing debate whether the intake of soy-derived isoflavones (sISO) mediates beneficial or adverse effects with regard to breast cancer risk. Therefore, we investigated whether nutritional exposure to a sISO-enriched diet from conception until adulthood impacts on 17β-estradiol (E2)-induced carcinogenesis in the rat mammary gland (MG). August-Copenhagen-Irish (ACI) rats were exposed to dietary sISO from conception until postnatal day 285. Silastic tubes containing E2 were used to induce MG tumorigenesis. Body weight, food intake, and tumor growth were recorded weekly. At necropsy, the number, position, size, and weight of each tumor were determined. Plasma samples underwent sISO analysis, and the morphology of MG was analyzed. Tumor incidence and multiplicity were reduced by 20 and 56 %, respectively, in the sISO-exposed rats compared to the control rats. Time-to-tumor onset was shortened from 25 to 20 weeks, and larger tumors developed in the sISO-exposed rats. The histological phenotype of the MG tumors was independent of the sISO diet received, and it included both comedo and cribriform phenotypes. Morphological analyses of the whole-mounted MGs also showed no diet-dependent differences. Lifelong exposure to sISO reduced the overall incidence of MG carcinomas in ACI rats, although the time-to-tumor was significantly shortened.



Similar content being viewed by others
References
Adlercreutz H, Markkanen H, Watanabe S (1993) Plasma concentrations of phyto-oestrogens in Japanese men. Lancet 342:1209–1210
Allred CD, Allred KF, Ju YH et al (2001) Soy diets containing varying amounts of genistein stimulate growth of estrogen-dependent (MCF-7) tumors in a dose-dependent manner. Cancer Res 61:5045–5050
Allred CD, Allred KF, Ju YH et al (2004) Dietary genistein results in larger MNU-induced, estrogen-dependent mammary tumors following ovariectomy of Sprague-Dawley rats. Carcinogenesis 25:211–218. doi:10.1093/carcin/bgg198
Arai Y, Uehara M, Sato Y et al (2000) Comparison of isoflavones among dietary intake, plasma concentration and urinary excretion for accurate estimation of phytoestrogen intake. J Epidemiol 10:127–135. doi:10.2188/jea.10.127
Blei T, Soukup ST, Schmalbach K et al (2015) Dose-dependent effects of isoflavone exposure during early lifetime on the rat mammary gland: studies on estrogen sensitivity, isoflavone metabolism, and DNA methylation. Mol Nutr Food Res 59:270–283. doi:10.1002/mnfr.201400480
Chen M, Rao Y, Zheng Y et al (2014) Association between soy isoflavone intake and breast cancer risk for pre- and post-menopausal women: a meta-analysis of epidemiological studies. PLoS ONE 9:e89288. doi:10.1371/journal.pone.0089288
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate-phenol-chloroform extraction. Anal Biochem 162(1):156–159
Cohen PE, Milligan SR (1993) Silastic implants for delivery of oestradiol to mice. J Reprod Fertil 99:219–223. doi:10.1530/jrf.0.0990219
Constantinou AI, Lantvit D, Hawthorne M et al (2001) Chemopreventive effects of soy protein and purified soy isoflavones on DMBA-induced mammary tumors in female Sprague-Dawley rats. Nutr Cancer 41:75–81. doi:10.1080/01635581.2001.9680615
de Assis S, Warri A, Cruz MI, Hilakivi-Clarke L (2010) Changes in mammary gland morphology and breast cancer risk in rats. J Vis Exp. doi:10.3791/2260
Fritz WA, Coward L, Wang J, Lamartiniere CA (1998) Dietary genistein: perinatal mammary cancer prevention, bioavailability and toxicity testing in the rat. Carcinogenesis 19:2151–2158
Helle J, Kräker K, Bader MI et al (2014) Assessment of the proliferative capacity of the flavanones 8-prenylnaringenin, 6-(1.1-dimethylallyl)naringenin and naringenin in MCF-7 cells and the rat mammary gland. Mol Cell Endocrinol 392:125–135. doi:10.1016/j.mce.2014.05.014
Hosoda K, Furuta T, Ishii K (2011) Metabolism and disposition of isoflavone conjugated metabolites in humans after ingestion of kinako. Drug Metab Dispos 39:1762–1767. doi:10.1124/dmd.111.038281
Lamartiniere CA, Moore JB, Brown NM et al (1995) Genistein suppresses mammary cancer in rats. Carcinogenesis 16:2833–2840. doi:10.1093/carcin/16.11.2833
Lamartiniere CA, Zhang JX, Cotroneo MS (1998) Genistein studies in rats: potential for breast cancer prevention and reproductive and developmental toxicity. Am J Clin Nutr 68:1400S–1405S
Lamartiniere CA, Cotroneo MS, Fritz WA et al (2002) Genistein chemoprevention: timing and mechanisms of action in murine mammary and prostate. J Nutr 132:552S–558S
Lampe JW, Karr SC, Hutchins AM, Slavin JL (1998) Urinary equol excretion with a soy challenge: influence of habitual diet. Proc Soc Exp Biol Med 217:335–339
Lee SA, Shu XO, Li H et al (2009) Adolescent and adult soy food intake and breast cancer risk: results from the Shanghai Women’s Health Study. Am J Clin Nutr 89:1920–1926
Li K-M, Todorovic R, Devanesan P et al (2004) Metabolism and DNA binding studies of 4-hydroxyestradiol and estradiol-3,4-quinone in vitro and in female ACI rat mammary gland in vivo. Carcinogenesis 25:289–297. doi:10.1093/carcin/bgg191
Magee PJ, Rowland I (2012) Soy products in the management of breast cancer. Curr Opin Clin Nutr Metab Care 15:586–591. doi:10.1097/MCO.0b013e328359156f
Mehrara E, Forssell-Aronsson E, Ahlman H, Bernhardt P (2007) Specific growth rate versus doubling time for quantitative characterization of tumor growth rate. Cancer Res 67:3970–3975. doi:10.1158/0008-5472.CAN-06-3822
Messina M, Hilakivi-Clarke L (2009) Early intake appears to be the key to the proposed protective effects of soy intake against breast cancer. Nutr Cancer 61:792–798. doi:10.1080/01635580903285015
Möller FJ, Diel P, Zierau O et al (2010) Long-term dietary isoflavone exposure enhances estrogen sensitivity of rat uterine responsiveness mediated through estrogen receptor alpha. Toxicol Lett 196:142–153
Molzberger AF, Vollmer G, Hertrampf T et al (2012) In utero and postnatal exposure to isoflavones results in a reduced responsivity of the mammary gland towards estradiol. Mol Nutr Food Res 56:399–409. doi:10.1002/mnfr.201100371
Molzberger AF, Soukup ST, Kulling SE, Diel P (2013) Proliferative and estrogenic sensitivity of the mammary gland are modulated by isoflavones during distinct periods of adolescence. Arch Toxicol 87:1129–1140. doi:10.1007/s00204-012-1009-x
Murrill WB, Brown NM, Zhang J-X et al (1996) Prepubertal genistein exposure suppresses mammary cancer and enhances gland differentiation. Carcinogenesis 17:1451–1457. doi:10.1093/carcin/17.7.1451
Nagata C (2010) Factors to consider in the association between soy isoflavone intake and breast cancer risk. J Epidemiol 20:83–89. doi:10.2188/jea.JE20090181
National Toxicology Program (2008) Toxicology and carcinogenesis studies of genistein (CAS No. 446-72-0) in Sprague-Dawley rats (feed study). Natl Toxicol Program Tech Rep Ser (545):1–240
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Russo J, Gusterson BA, Rogers AE, Russo IH, Wellings SR, van Zwieten MJ (1990) Comparative study of human and rat mammary tumorigenesis. Lab Investig J Tech Methods Pathol 62:244–278
Santell RC, Kieu N, Helferich WG (2000) Genistein inhibits growth of estrogen-independent human breast cancer cells in culture but not in athymic mice. J Nutr 130:1665–1669
Seo H-S, DeNardo DG, Jacquot Y et al (2006) Stimulatory effect of genistein and apigenin on the growth of breast cancer cells correlates with their ability to activate ER alpha. Breast Cancer Res Treat 99:121–134. doi:10.1007/s10549-006-9191-2
Setchell KD, Brown NM, Desai PB et al (2003) Bioavailability, disposition, and dose-response effects of soy isoflavones when consumed by healthy women at physiologically typical dietary intakes. J Nutr 133:1027–1035
Shu XO, Jin F, Dai Q et al (2001) Soyfood intake during adolescence and subsequent risk of breast cancer among chinese women. Cancer Epidemiol Biomark Prev 10:483–488
Shu XO, Zheng Y, Cai H et al (2009) Soy food intake and breast cancer survival. JAMA 302:2437–2443
Shull JD, Spady TJ, Snyder MC et al (1997) Ovary-intact, but not ovariectomized female ACI rats treated with 17beta-estradiol rapidly develop mammary carcinoma. Carcinogenesis 18:1595–1601. doi:10.1093/carcin/18.8.1595
Soukup ST, Al-Maharik N, Botting N, Kulling SE (2014) Quantification of soy isoflavones and their conjugative metabolites in plasma and urine: an automated and validated UHPLC-MS/MS method for use in large-scale studies. Anal Bioanal Chem. doi:10.1007/s00216-014-8034-y
Taylor CK, Levy RM, Elliott JC, Burnett BP (2009) The effect of genistein aglycone on cancer and cancer risk: a review of in vitro, preclinical, and clinical studies. Nutr Rev 67:398–415. doi:10.1111/j.1753-4887.2009.00213.x
Turan VK, Sanchez RI, Li JJ et al (2004) The effects of steroidal estrogens in ACI rat mammary carcinogenesis: 17β-estradiol, 2-hydroxyestradiol, 4-hydroxyestradiol, 16α-hydroxyestradiol, and 4-hydroxyestrone. J Endocrinol 183:91–99. doi:10.1677/joe.1.05802
Watson KL, Stalker L, Jones RA, Moorehead RA (2015) High levels of dietary soy decrease mammary tumor latency and increase incidence in MTB-IGFIR transgenic mice. BMC Cancer. doi:10.1186/s12885-015-1037-z
Acknowledgments
The authors are grateful to Dr. Carolin Kleider for help in determining E2 levels in the implants and for performing the UHPLC-MS/MS analysis of estrogens in plasma, to Antje Beyer for support with the animal experiments and technical assistance, and to Dr. Katja Schmalbach for assistance with the biostatistical analysis. This work is part of the joint research project, IsoCross, entitled, ‘Isoflavones: Cross-species comparison of metabolism, estrogen sensitivity, epigenetics and carcinogenesis’ that is funded by the German Research Foundation (Grants: VO 410/12-1, KU 1079/10–1, LE 1329/10–1).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Human and animal participants
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Möller, F.J., Pemp, D., Soukup, S.T. et al. Soy isoflavone exposure through all life stages accelerates 17β-estradiol-induced mammary tumor onset and growth, yet reduces tumor burden, in ACI rats. Arch Toxicol 90, 1907–1916 (2016). https://doi.org/10.1007/s00204-016-1674-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00204-016-1674-2