Abstract
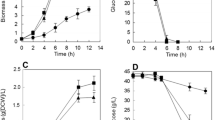
A Δsud deletion mutant of Wolinella succinogenes that lacked the periplasmic sulfide dehydrogenase (Sud) was constructed using homologous recombination. The mutant grew with sulfide and fumarate, indicating that Sud was not a component of the electron transport chain that catalyzed fumarate respiration with sulfide as an electron donor. Likewise, growth with formate and either polysulfide or sulfur was not affected by the deletion. Removal of Sud from wild-type W. succinogenes by spheroplast formation did not decrease the activity of electron transport to polysulfide. The Δpsr deletion mutant that lacks polysulfide reductase (Psr) grew by fumarate respiration with sulfide as an electron donor, indicating that Psr is not required for this activity.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 31 August 1995 / Accepted: 25 October 1995
Rights and permissions
About this article
Cite this article
Kotzian, S., Kreis-Kleinschmidt, V., Krafft, T. et al. Properties of a Wolinella succinogenes mutant lacking periplasmic sulfide dehydrogenase (Sud). Arch Microbiol 165, 65–68 (1996). https://doi.org/10.1007/s002030050298
Issue Date:
DOI: https://doi.org/10.1007/s002030050298