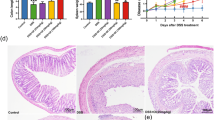
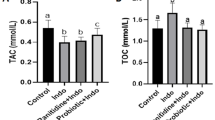
Abstract
Acute pancreatitis frequently causes intestinal barrier damage, which aggravates pancreatitis. Although Clostridium butyricum exerts anti-inflammatory and protective effects on the intestinal barrier during acute pancreatitis, the underlying mechanism is unclear. The G protein-coupled receptors 109 A (GPR109A) and adenosine monophosphate-activated protein kinase (AMPK)/ peroxisome proliferator-activated receptor-gamma coactivator 1 alpha (PGC-1α) signaling pathways can potentially influence the integrity of the intestinal barrier. Our study generated acute pancreatitis mouse models via intraperitoneal injection of cerulein and lipopolysaccharides. After intervention with Clostridium butyricum, the model mice showed reduced small intestinal and colonic intestinal barrier damage, dysbiosis amelioration, and increased GPR109A/AMPK/PGC-1α expression. In conclusion, Clostridium butyricum could improve pancreatic and intestinal inflammation and pancreatic injury, and relieve acute pancreatitis-induced intestinal barrier damage in the small intestine and colon, which may be associated with GPR109A/AMPK/PGC-1α.








Similar content being viewed by others
Data availability
Some data and methods are available from the first author upon reasonable request.
References
Beger H G, Bittner R (1986) Bacterial contamination of pancreatic necrosis. A prospective clinical study [J]. Gastroenterology 91(2):433–438. https://doi.org/10.1016/0016-5085(86)90579-2
Blad C C, Tang C, Offermanns S (2012) G protein-coupled receptors for energy metabolites as new therapeutic targets [J]. Nat Rev Drug Discov 11(8):603–619. https://doi.org/10.1038/nrd3777
BOXHOORN L, VOERMANS R P, BOUWENSE SA et al (2020) Acute pancreatitis [J]. Lancet (London, England), 396(10252): 726 – 34. https://doi.org/10.1016/s0140-6736(20)31310-6
Cantó C, Gerhart-Hines Z, FEIGE J N et al (2009) AMPK regulates energy expenditure by modulating NAD + metabolism and SIRT1 activity [J]. Nature 458(7241):1056–1060. https://doi.org/10.1038/nature07813
Capurso G, Zerboni G, Signoretti M et al (2012) Role of the gut barrier in acute pancreatitis [J]. J Clin Gastroenterol. https://doi.org/10.1097/MCG.0b013e3182652096. 46 Suppl: S46-51
Chelakkot C, Ghim J, Ryu SH (2018) Exp Mol Med 50(8):1–9. https://doi.org/10.1038/s12276-018-0126-x. Mechanisms regulating intestinal barrier integrity and its pathological implications [J]
Chiu C J, Scott H J, Gurd F N (1970). Intestinal mucosal lesion in low-flow states. II. The protective effect of intraluminal glucose as energy substrate [J]. Archives of surgery (Chicago, Ill: 1960), 101(4): 484-8. https://doi.org/10.1001/archsurg.1970.01340280036010
CUNNINGHAM K E VINCENTG, SODHI C P et al (2016) Peroxisome proliferator-activated Receptor-γ Coactivator 1-α (PGC1α) protects against experimental murine colitis [J]. J Biol Chem 291(19):10184–10200. https://doi.org/10.1074/jbc.M115.688812
D’errico I SALVATOREL (2011) Peroxisome proliferator-activated receptor-gamma coactivator 1-alpha (PGC1alpha) is a metabolic regulator of intestinal epithelial cell fate [J]. Proc Natl Acad Sci USA 108(16):6603–6608. https://doi.org/10.1073/pnas.1016354108
Elamin E E, Masclee A A, Dekker J et al (2013) Short-chain fatty acids activate AMP-activated protein kinase and ameliorate ethanol-induced intestinal barrier dysfunction in Caco-2 cell monolayers [J]. J Nutr 143(12):1872–1881. https://doi.org/10.3945/jn.113.179549
Glaubitz J, Wilden A, Frost F et al (2023) Activated regulatory T-cells promote duodenal bacterial translocation into necrotic areas in severe acute pancreatitis [J]. Gut 72(7):1355–1369. https://doi.org/10.1136/gutjnl-2022-327448
Guilloteau P, Martin L, Eeckhaut V, et al (2010) From the gut to the peripheral tissues: the multiple effects of butyrate [J]. Nutr Res Rev 23(2):366–384. https://doi.org/10.1017/s0954422410000247
Guo W, Li W, Su Y et al (2021) GPR109A alleviate mastitis and enhances the blood milk barrier by activating AMPK/Nrf2 and autophagy [J]. Int J Biol Sci 17(15):4271–4284. https://doi.org/10.7150/ijbs.62380
Hagihara M, KUROKI Y, ARIYOSHI T et al (2020) Clostridium butyricum modulates the Microbiome to protect intestinal barrier function in mice with antibiotic-Induced dysbiosis [J]. iScience 23(1):100772. https://doi.org/10.1016/j.isci.2019.100772
Hoque R, Mehal W Z (2015) Inflammasomes in pancreatic physiology and disease [J]. Am J Physiol Gastrointest Liver Physiol 308(8):G643–G651. https://doi.org/10.1152/ajpgi.00388.2014
Jäger S, Handschin C, St-Pierre J et al (2007) AMP-activated protein kinase (AMPK) action in skeletal muscle via direct phosphorylation of PGC-1alpha [J]. Proc Natl Acad Sci USA 104(29):12017–12022. https://doi.org/10.1073/pnas.0705070104
Jin M, Zhang H, Wu M et al (2022) Colonic interleukin-22 protects intestinal mucosal barrier and microbiota abundance in severe acute pancreatitis [J]. FASEB journal: official publication of the Federation of American Societies for Experimental Biology. 36(3):e22174. https://doi.org/10.1096/fj.202101371R
Kim MS, Shigenaga J K, Moser A H et al (2005) Suppression of estrogen-related receptor alpha and medium-chain acyl-coenzyme A dehydrogenase in the acute-phase response [J]. J Lipid Res 46(10):2282–2288. https://doi.org/10.1194/jlr.M500217-JLR200
Kuo W T, Odenwald M A, Turner J R et al (2022) Tight junction proteins occludin and ZO-1 as regulators of epithelial proliferation and survival [J]. Ann N Y Acad Sci 1514(1):21–33. https://doi.org/10.1111/nyas.14798
Lei Y, Tang L, Liu S et al (2021) Parabacteroides produces acetate to alleviate heparanase-exacerbated acute pancreatitis through reducing neutrophil infiltration [J]. Microbiome 9(1):115. https://doi.org/10.1186/s40168-021-01065-2
Lerch MM, Gorelick FS (2013) Models of acute and chronic pancreatitis [J]. Gastroenterology 144(6):1180–1193. https://doi.org/10.1053/j.gastro.2012.12.043
Lesina M, Wörmann SM, Neuhöfer P et al (2014) Interleukin-6 in inflammatory and malignant diseases of the pancreas [J]. Semin Immunol 26(1):80–87. https://doi.org/10.1016/j.smim.2014.01.002
Li Z, Mccafferty K J, JUDD RL (2021) Role of HCA(2) in regulating intestinal homeostasis and suppressing Colon carcinogenesis [J]. Front Immunol 12:606384. https://doi.org/10.3389/fimmu.2021.606384
Li H, Sun J, Du J et al (2018) Clostridium butyricum exerts a neuroprotective effect in a mouse model of traumatic brain injury via the gut-brain axis [J]. Neurogastroenterol Motil 30(5):e13260. https://doi.org/10.1111/nmo.13260
Li X, Jamal M, Guo P, Biomedicine et al (2019) Pharmacotherapy = Biomedecine Pharmacotherapie 118:109363. https://doi.org/10.1016/j.biopha.2019.109363
Li X Y, He C, Zhu Y et al (2020) Role of gut microbiota on intestinal barrier function in acute pancreatitis [J]. World J Gastroenterol 26(18):2187–2193. https://doi.org/10.3748/wjg.v26.i18.2187
Li Z R Pengl, Green RS et al (2009) Butyrate enhances the intestinal barrier by facilitating tight junction assembly via activation of AMP-activated protein kinase in Caco-2 cell monolayers [J]. J Nutr 139(9):1619–1625. https://doi.org/10.3945/jn.109.104638
Ma X, Fan P X, Li LS et al (2012) Butyrate promotes the recovering of intestinal wound healing through its positive effect on the tight junctions [J]. J Anim Sci 90(Suppl 4):266–268. https://doi.org/10.2527/jas.50965
MA L, Shen Q, Lyu W et al (2022) Clostridium butyricum and its derived extracellular vesicles modulate gut homeostasis and ameliorate Acute Experimental colitis [J]. Microbiol Spectr 10(4):e0136822. https://doi.org/10.1128/spectrum.01368-22
Nguyen TD, Hållenius FF, Lin X et al (2020) Monobutyrin and Monovalerin affect brain short-chain fatty acid profiles and tight-Junction protein expression in ApoE-Knockout rats Fed High-Fat diets [J]. Nutrients 12(4). https://doi.org/10.3390/nu12041202
Niu Panll, FANG W (2019) Mol Nutr Food Res 63(13):e1801419. https://doi.org/10.1002/mnfr.201801419. X, et al. Clostridium butyricum Strains Suppress Experimental Acute Pancreatitis by Maintaining Intestinal Homeostasis [J]
Piao X, Liu B, Sui X et al (2020) Picroside II Improves Severe Acute Pancreatitis-Induced Intestinal Barrier Injury by Inactivating Oxidative and Inflammatory TLR4-Dependent PI3K/AKT/NF-κB Signaling and Improving Gut Microbiota [J]. Oxidative medicine and cellular longevity, 2020: 3589497. https://doi.org/10.1155/2020/3589497
Rowart P, Wu J, Caplan M J et al (2018) Implications of AMPK in the formation of epithelial tight junctions [J]. Int J Mol Sci 19(7). https://doi.org/10.3390/ijms19072040
Schwarz M, Thomsen J, Meyer H et al (2000) Frequency and time course of pancreatic and extrapancreatic bacterial infection in experimental acute pancreatitis in rats [J]. Surgery 127(4):427–432. https://doi.org/10.1067/msy.2000.104116
Shimizu T, Shiratori K, Sawada T et al (2000) Recombinant human interleukin-11 decreases severity of acute necrotizing pancreatitis in mice [J]. Pancreas 21(2):134–140. https://doi.org/10.1097/00006676-200008000-00005
Singh N, Gurav A, Sivaprakasam S et al (2014) Activation of Gpr109a, receptor for niacin and the commensal metabolite butyrate, suppresses colonic inflammation and carcinogenesis [J]. Immunity 40(1):128–139. https://doi.org/10.1016/j.immuni.2013.12.007
Stoeva M K, Garcia-So J, Justice N et al (2021) Butyrate-producing human gut symbiont, Clostridium butyricum, and its role in health and disease [J]. Gut Microbes 13(1):1–28. https://doi.org/10.1080/19490976.2021.1907272
Sun Y Y, Li M, Li YY et al (2018) The effect of Clostridium butyricum on symptoms and fecal microbiota in diarrhea-dominant irritable bowel syndrome: a randomized, double-blind, placebo-controlled trial [J]. Sci Rep 8(1):2964. https://doi.org/10.1038/s41598-018-21241-z
Thangaraju M, Cresci G A, Liu K et al (2009) GPR109A is a G-protein-coupled receptor for the bacterial fermentation product butyrate and functions as a tumor suppressor in colon [J]. Cancer Res 69(7):2826–2832. https://doi.org/10.1158/0008-5472.Can-08-4466
Xiao Z, Liu L, Jin Y et al (2021a) Clostridium tyrobutyricum protects against LPS-Induced Colonic inflammation via IL-22 signaling in mice [J]. Nutrients 13(1). https://doi.org/10.3390/nu13010215
Xiao Z, Liu L, Pei X et al (2021b) A potential probiotic for Diarrhea: Clostridium tyrobutyricum protects against LPS-Induced Epithelial Dysfunction via IL-22 produced by Th17 cells in the ileum [J]. Front Immunol 12:758227. https://doi.org/10.3389/fimmu.2021.758227
Xu RC, Miao W T, Xu JY et al (2022) Neuroprotective effects of Sodium Butyrate and Monomethyl Fumarate Treatment through GPR109A Modulation and Intestinal Barrier Restoration on PD mice [J]. Nutrients 14(19). https://doi.org/10.3390/nu14194163
Yan Q, Jia L, Wen B et al (2022) Clostridium butyricum protects against pancreatic and intestinal Injury after severe Acute pancreatitis via downregulation of MMP9 [J]. Front Pharmacol 13:919010. https://doi.org/10.3389/fphar.2022.919010
Yu S, Xiong Y, Xu J et al (2020) Identification of dysfunctional gut microbiota through rectal swab in patients with different severity of Acute pancreatitis [J]. Dig Dis Sci 65(11):3223–3237. https://doi.org/10.1007/s10620-020-06061-4
Zhang YJ, Wu Q (2021) Sulforaphane protects intestinal epithelial cells against lipopolysaccharide-induced injury by activating the AMPK/SIRT1/PGC-1ɑ pathway [J]. Bioengineered 12(1):4349–4360. https://doi.org/10.1080/21655979.2021.1952368
Zhang X M, Zhang Z Y, Zhang C H et al (2018) Biomed Environ Sci: BES 31(1):81–86. https://doi.org/10.3967/bes2018.010. Intestinal Microbial Community Differs between Acute Pancreatitis Patients and Healthy Volunteers [J]
Zhao HB, Yan Q Q Jial et al (2020) Effect of Clostridium butyricum and Butyrate on Intestinal Barrier functions: study of a rat model of severe Acute Pancreatitis with Intra-abdominal hypertension [J]. Front Physiol 11:561061. https://doi.org/10.3389/fphys.2020.561061
Zheng B, Cantley LC (2007) Regulation of epithelial tight junction assembly and disassembly by AMP-activated protein kinase [J]. Proc Natl Acad Sci USA 104(3):819–822. https://doi.org/10.1073/pnas.0610157104
Zhu Y, He C, Li X et al (2019) Gut microbiota dysbiosis worsens the severity of acute pancreatitis in patients and mice [J]. J Gastroenterol 54(4):347–358. https://doi.org/10.1007/s00535-018-1529-0
Zhu Y, Mei Q, Fu Y, Biomedicine et al (2021) Pharmacotherapy = Biomedecine Pharmacotherapie 141:111850. https://doi.org/10.1016/j.biopha.2021.111850
Funding
Our study was funded by the National Natural Science Foundation of China (No. 81870438), National Natural Science Foundation of Guangdong Province (No. 2022A1515010321), Guangzhou Technology Projects (N0.2023A03J0954), and Guangzhou Technology Projects (No. 202102020099).
Author information
Authors and Affiliations
Contributions
Guiqing Deng: Investigation, Formal analysis, Writing-Original Draft. Biyan Wen: Methodology, Project administration. Lin Jia: Conceptualization, Project administration. Jiaxin Liu: Software.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Our study was approved by the Medical Ethics Committee of South China University of Technology. Permission certificate: SYXK (Guang dong)2022 − 0180; Validity: 2022/11/16-2027/11/15; Address: Laboratory Animal Center, B2 Building, South China University of Technology, Guangzhou University City, Guangzhou, China.
Conflict of interest
There are no conflicts of interest among the authors.
Additional information
Communicated by Dmitriy Volokhov.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Deng, G., Wen, B., Jia, L. et al. Clostridium butyricum upregulates GPR109A/AMPK/PGC-1α and ameliorates acute pancreatitis-associated intestinal barrier injury in mice. Arch Microbiol 206, 265 (2024). https://doi.org/10.1007/s00203-024-04001-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-024-04001-8