Abstract
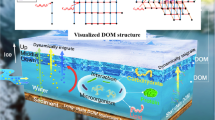
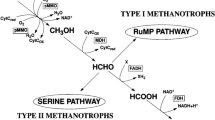
Coal seam microbes, as endogenous drivers of secondary biogenic gas production in coal seams, might be related to methane production in coal seams. In this study, we carried out anaerobic indoor culture experiments of microorganisms from three different depths of bituminous coal seams in Huainan mining area, and revealed the secondary biogas generation mechanism of bituminous coal seams by using the combined analysis of macro-genome and metabolism multi-omics. The results showed that the cumulative mass molar concentrations (Molality) of biomethane production increased with the increase of the coal seam depth in two consecutive cycles. At the genus level, there were significant differences in the bacterial and archaeal community structures corresponding to the three coal seams 1#, 6#, and 9#(p < 0.05). The volatile matter of air-dry basis (Vad) of coal was significantly correlated with differences in genus-level composition of bacteria and archaea, with correlations of R bacterial = 0.368 and R archaeal = 0.463, respectively. Functional gene analysis showed that the relative abundance of methanogenesis increased by 42% before and after anaerobic fermentation cultivation. Meanwhile, a total of 11 classes of carbon metabolism homologues closely related to methanogenesis were detected in the liquid metabolites of coal bed microbes after 60 days of incubation. Finally, the fatty acid, amino acid and carbohydrate synergistic methanogenic metabolic pathway was reconstructed based on the Kyoto Encyclopedia of Genes and Genomes (KEGG) database. The expression level of mcrA gene within the metabolic pathway of the 1# deep coal sample was significantly higher than that of the other two groups (p < 0.05 for significance), and the efficient expression of mcrA gene at the end of the methanogenic pathway promoted the conversion of bituminous coal organic matter to methane. Therefore, coal matrix compositions may be the key factors causing diversity in microbial community and metabolic function, which might be related to the different methane content in different coal seams.






Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Akimbekov NS, Digel I, Tastambek KT, Kozhahmetova M, Sherelkhan DK, Tauanov Z (2024) Hydrogenotrophic methanogenesis in coal-bearing environments: methane production, carbon sequestration, and hydrogen availability. Int J Hydrog Energy 52:1264–1277. https://doi.org/10.1016/j.ijhydene.2023.09.223
Chen T, Zheng H, Hamilton S, Rodrigues S, Golding SD, Rudolph V (2017) Characterisation of bioavailability of Surat Basin Walloon coals for biogenic methane production using environmental microbial consortia. Int J Coal Geol 179:92–112. https://doi.org/10.1016/j.coal.2017.05.017
Chen D, Zuo X, Li J, Wang X, Liu J (2020) Carbon migration and metagenomic characteristics during anaerobic digestion of rice straw. Biotechnol Biofuels 13:130. https://doi.org/10.1186/s13068-020-01770-4
Devignes CS, Carmeliet G, Stegen S (2022) Amino acid metabolism in skeletal cells. Bone Rep 17:101620. https://doi.org/10.1016/j.bonr.2022.101620
Fang X, Li J, Rui J, Li X (2015) Research progress in biochemical pathways of methanogenesis. Chin J Appl Environ Biol 21. https://doi.org/10.3724/SP.J.1145.2014.08019
Flores RM, Rice CA, Stricker GD, Warden A, Ellis MS (2008) Methanogenic pathways of coal-bed gas in the Powder River Basin, United States: the geologic factor. Int J Coal Geol 76:52–75. https://doi.org/10.1016/j.coal.2008.02.005
Green MS, Flanegan KC, Gilcrease PC (2008) Characterization of a methanogenic consortium enriched from a coalbed methane well in the Powder River Basin, U.S.A. Int J Coal Geol 76:34–45. https://doi.org/10.1016/j.coal.2008.05.001
Guo H et al (2023) Analysis on methane production from various coal slime fermentations based on metagenomics. J Environ Manage 343:118058. https://doi.org/10.1016/j.jenvman.2023.118058
Hamilton SK, Golding SD, Baublys KA, Esterle JS (2014) Stable isotopic and molecular composition of desorbed coal seam gases from the Walloon Subgroup, eastern Surat Basin, Australia. Int J Coal Geol 122:21–36. https://doi.org/10.1016/j.coal.2013.12.003
Hua ZS et al (2019) Insights into the ecological roles and evolution of methyl-coenzyme M reductase-containing hot spring Archaea. Nat Commun 10:4574. https://doi.org/10.1038/s41467-019-12574-y
Jones EJ, Voytek MA, Corum MD, Orem WH (2010) Stimulation of methane generation from nonproductive coal by addition of nutrients or a microbial consortium. Appl Environ Microbiol 76:7013–7022. https://doi.org/10.1128/AEM.00728-10
Li Y, Zhang Y, Xue S (2022) pH mediated assemblage of carbon, nitrogen, and sulfur related microbial communities in petroleum reservoirs. Front Microbiol 13:952285. https://doi.org/10.3389/fmicb.2022.952285
Li Y, Zhang Y, Xue S, Liu B (2023) Actinobacteria may influence biological methane generation in coal seams. Fuel 339. https://doi.org/10.1016/j.fuel.2022.126917
Lin TH, Yang RS, Tang CH, Wu MY, Fu WM (2008) Regulation of the maturation of osteoblasts and osteoclastogenesis by glutamate. Eur J Pharmacol 589:37–44. https://doi.org/10.1016/j.ejphar.2008.04.060
Liu B et al (2019a) Variations in Microbiota communities with the ranks of coals from three Permian Mining Areas. Energy Fuels 33:5243–5252. https://doi.org/10.1021/acs.energyfuels.8b04413
Liu F et al (2019b) Characterization of organic compounds from hydrogen peroxide-treated subbituminous coal and their composition changes during microbial methanogenesis. Fuel 237:1209–1216. https://doi.org/10.1016/j.fuel.2018.10.043
Liu B, Wang Y, Li Y, Yang Y, Chen J, Zhang T (2022) Improved formation of biogenic methane by cultivable bacteria in highly volatile bituminous coals. J Clean Prod 366. https://doi.org/10.1016/j.jclepro.2022.132900
Mentch SJ, Locasale JW (2016) One-carbon metabolism and epigenetics: understanding the specificity. Ann N Y Acad Sci 1363:91–98. https://doi.org/10.1111/nyas.12956
Mercatali L et al (2016) The Effect of Everolimus in an in vitro model of Triple negative breast Cancer and osteoclasts. Int J Mol Sci 17. https://doi.org/10.3390/ijms17111827
Morita SY, Ikeda Y (2022) Regulation of membrane phospholipid biosynthesis in mammalian cells. Biochem Pharmacol 206:115296. https://doi.org/10.1016/j.bcp.2022.115296
Ohgaki R (2012) The isolation of methanogen in the low temperature and studies on the activity of produces methane J Biol 29 doi: 10.3969/j.
Ritter D et al (2015) Enhanced microbial coalbed methane generation: a review of research, commercial activity, and remaining challenges. Int J Coal Geol 146:28–41. https://doi.org/10.1016/j.coal.2015.04.013
Ruan L, Zhang S, Chen X, Liang W, Xie Q (2022) Role of anti-angiogenic factors in the pathogenesis of breast cancer: a review of therapeutic potential. Pathol Res Pract 236:153956. https://doi.org/10.1016/j.prp.2022.153956
Sechman H, Kotarba MJ, Dzieniewicz M, Romanowski T, Fiszer J (2017) Evidence of methane and carbon dioxide migration to the near surface zone in the area of the abandoned coal mines in Wałbrzych District (Lower Silesian Coal Basin, SW Poland) based on periodical changes of molecular and isotopic compositions. Int J Coal Geol 183:138–160. https://doi.org/10.1016/j.coal.2017.10.011
Shao P, Wang A, Wang W (2019) Effect of chemical structure of lignite and high-volatile bituminous coal on the generation of biogenic coalbed methane. Fuel 245:212–225. https://doi.org/10.1016/j.fuel.2019.02.061
Shimizu S, Akiyama M, Naganuma T, Fujioka M, Nako M, Ishijima Y (2007) Molecular characterization of microbial communities in deep coal seam groundwater of northern Japan. Geobiology 5:423–433. https://doi.org/10.1111/j.1472-4669.2007.00123.x
Susilawati R, Esterle JS, Golding SD, Mares TE (2015) Microbial Methane potential for the South Sumatra Basin coal: formation water screening and coal substrate bioavailability. Energy Procedia 65:282–291. https://doi.org/10.1016/j.egypro.2015.01.051
Tao M, Shi B, Li J, Wang W, Li X, Gao B (2007) Secondary biological coalbed gas in the Xinji area, Anhui province, China: evidence from the geochemical features and secondary changes. Int J Coal Geol 71:358–370. https://doi.org/10.1016/j.coal.2006.12.002
Wang Y, Wegener G, Hou J, Wang F, Xiao X (2019) Expanding anaerobic alkane metabolism in the domain of Archaea. Nat Microbiol 4:595–602. https://doi.org/10.1038/s41564-019-0364-2
Wang H et al (2022) Bioelectrochemically assisting anaerobic digestion enhanced methane production under low-temperature. Renewable Energy 194:1071–1083. https://doi.org/10.1016/j.renene.2022.05.118
Wang X et al (2024) Biogenic methane clumped isotope signatures: insights from microbially enhanced coal bed methane. Fuel 365. https://doi.org/10.1016/j.fuel.2024.131307
Xianbo S, Ying X, Yu W, Da-ping X, Xin C (2011) Effect of salinity and pH on biogenic methane production of low-rank coal. J CHINA COAL Soc 36. https://doi.org/10.13225/j.cnki.jccs.2011.08.002
XiangZhao SYLZM (2024) Establishment and optimization of technique for direct desulfation from Gracilaria plant’s Agar by Priestia megaterium. Food Res Develop 45. https://doi.org/10.12161/j.issn.1005%20436521.2024.02.025
Xu F et al (2023) The status and development strategy of coalbed methane industry in China. Pet Explor Dev 50:765–783. https://doi.org/10.1016/s1876-3804(23)60427-6
Yongyong J, Yuanyuan C, Ziyu Y (2022) Comparative analysis of methane adsorption characteristics of different coal ranks coal body. Shaanxi coal
Zhang J, Liang Y, Pandey R, Harpalani S (2015) Characterizing microbial communities dedicated for conversion of coal to methane in situ and ex situ. Int J Coal Geol 146:145–154. https://doi.org/10.1016/j.coal.2015.05.001
Zhang J, Bi Z, Liang Y (2018) Development of a nutrient recipe for enhancing methane release from coal in the Illinois basin. Int J Coal Geol 187:11–19. https://doi.org/10.1016/j.coal.2018.01.001
Zheng H, Chen T, Rudolph V, Golding SD (2017) Biogenic methane production from Bowen Basin coal waste materials. Int J Coal Geol 169:22–27. https://doi.org/10.1016/j.coal.2016.09.006
Acknowledgements
The paper was supported by National Key R&D Program of China(NO.2023YFC3009002); Independent Research fund of Joint National-Local Engineering Research Centre for Safe and Precise Coal Mining(Anhui University of Science and Technology)(NO. EC2023006); The Key Research and Development Projects in Anhui Province(NO.2022l07020020); National Natural Science Foundation of China (No. 52274171); National Natural Science Foundation of China (Grant No. 52104113).
Author information
Authors and Affiliations
Contributions
X.Z., B.L., S.X., J.C., C.Z.,Y.Y.,Y.Z.,J.W., and J.Z. conducted the bulk of the data analysis for the study and co-wrote the manuscript. X.Z.,B.L.,S.X.andC.Z. provided the funding for the study, were involved in the conceptualization of the study, as well as assisted in the writing of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Yusuf Akhter.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, X., Liu, B., Xue, S. et al. Mechanisms of secondary biogenic coalbed methane formation in bituminous coal seams: a joint experimental and multi-omics study. Arch Microbiol 206, 263 (2024). https://doi.org/10.1007/s00203-024-03990-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-024-03990-w