Abstract
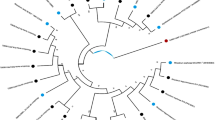
A novel actinobacterium strain, designated HUAS 2-6 T, was isolated from the rhizosphere soil of Camellia oleifera Abel collected from Taoyuan County, Northwestern Hunan Province, South China. This strain was subjected to a polyphasic taxonomic study. Strain HUAS 2-6 T is characterized by morphology typical of members of the genus Streptomyces, with deep purplish vinaceous aerial mycelia and deep dull lavender substrate mycelia. Strain HUAS 2-6 T, based on the full-length 16S rRNA gene sequence analysis, exhibited the highest similarities to S. puniciscabiei S77T (99.31%), S. filipinensis NBRC 12860 T (99.10%), S. yaanensis CGMCC 4.7035 T (99.09%), S. fodineus TW1S1T (99.08%), S. broussonetiae CICC 24819 T (98.76%), S. achromogenes JCM 4121 T (98.69%), S. barringtoniae JA03T (98.69%), and less than 98.70% with other validly species. In phylogenomic tree, strain HUAS 2-6 T was clustered together with S. broussonetiae CICC 24819 T, suggesting that they were closely related to each other. However, average nucleotide identity (ANI) and digital DNA–DNA hybridisation (dDDH) between them were much less than the species cutoff values (ANI 96.7% and dDDH 70%). Moreover, in phenotypic and chemotaxonomic characteristics, strain HUAS 2-6 T is distinct from S. broussonetiae CICC 24819 T. On the basis of the polyphasic data, strain HUAS 2-6 T is proposed to represent a novel species, Streptomyces camelliae sp. nov. (= MCCC 1K04729T = JCM 35918 T).



Similar content being viewed by others
Data availability
The NCBI accession numbers for the full-length 16S rRNA gene sequence and genome sequence of strain HUAS 2-6T are OQ363169 and CP115300, respectively.
Code availability
Not applicable.
Abbreviations
- ISP:
-
International Streptomyces Project
- MCCC:
-
Marine Culture Collection of China
References
Atlas RM (1993) Handbook of Microbiological Media. Edited by Parks LC. Boca Raton, FL: CRC Press
Blodgett JAV, Oh DC, Cao S, Currie CR, Kolter R, Clardy J (2010) Common biosynthetic origins for polycyclic tetramate macrolactams from phylogenetically diverse bacteria. Proc Natl Acad Sci 107:11692–11697. https://doi.org/10.1073/pnas.1001513107
Collins MD, Pirouz T, Goodfellow M, Minnikin DE (1977) Distribution of menaquinones in actinomycetes and corynebacteria. J Gen Microbiol 100:221–230
Egli A, Fuller J, Humar A, Lien D, Weinkauf J, Nador R et al (2012) Emergence of Aspergillus calidoustus infection in the era of posttransplantation azole prophylaxis. Transplantation Journal 94:403–410. https://doi.org/10.1097/tp.0b013e31825992f0
Farris JS (1972) Estimating phylogenetic trees from distance matrices. Am Nat 106:645–668. https://doi.org/10.1086/282802
Felsenstein J (1981) Evolutionary trees from DNA sequences: a maximum likelihood approach. J Mol Evol 17:368–376. https://doi.org/10.1007/BF01734359
Gallagher KA, Fenical W, Jensen PR (2010) Hybrid isoprenoid secondary metabolite production in terrestrial and marine actinomycetes. Curr Opin Biotechnol 21:794–800. https://doi.org/10.1016/j.copbio.2010.09.010
Hasegawa T, Takizawa M, Tanida S (1983) A rapid analysis for chemical grouping of aerobic actinomycetes. J Gen Appl Microbiol 29:319–322. https://doi.org/10.2323/jgam.29.319
Howard SJ (2014) Multi-resistant aspergillosis due to cryptic species. Mycopathologia 178:435–439. https://doi.org/10.1007/s11046-014-9774-0
Hu SR, Li KQ, Zhang YF, Wang YF, Fu L, Xiao Y et al (2022) New insights into the threshold values of multilocus sequence analysis, average nucleotide identity and digital DNA–DNA hybridization in delineating Streptomyces species. Front Microbiol 13:910277. https://doi.org/10.3389/fmicb.2022.910277
Jiang CR, Ruan JS (1982) Two new species and a new variety of Ampullarella. Acta Microbiol Sin 22:207–211. https://doi.org/10.13343/j.cnki.wsxb.1982.03.011
Kim MK, Kang HJ, Roh SG, Park JS, Kim SB (2019) Streptomyces fodineus sp. nov., an actinobacterium with antifungal activity isolated from mine area soil. Int J Syst Evol Microbiol 69:1350–1354. https://doi.org/10.1099/ijsem.0.003353
Kluge AG, Farris JS (1969) Quantitative phyletics and the evolution of anurans. Syst Zool 18:1–32
Koshy A, Dhevendaran K, Georgekutty MI, Natarajan P (1997) Asparaginase activity in Streptomyces plicatus isolated from the alimentary canal of the fish Gerres filamentous Cuvier. J Mar Biotechnol 5:181–185
Kroiss J, Kaltenpoth M, Schneider B, Schwinger MG, Hertweck C, Maddula RK et al (2010) Symbiotic streptomycetes provide antibiotic combination prophylaxis for wasp offspring. Nat Chem Biol 6:261–263. https://doi.org/10.1038/nchembio.331
Lamoth F (2016) Aspergillus fumigatus-related species in clinical practice. Front Microbiol 7:683. https://doi.org/10.3389/fmicb.2016.00683
Lane DJ (1991) 16S/23S rRNA sequencing. In: Stackebrandt E, Goodfellow M (eds) Nucleic Acid Techniques in Bacterial Systematics. Wiley, New York, USA, pp 115–175
Lechevalier MP, Lechevalier H (1970) Chemical composition as a criterion in the classification of aerobic actinomycetes. Int J Syst Bacteriol 20:435–443. https://doi.org/10.1099/00207713-20-4-435
Meier-Kolthoff JP, Auch AF, Klenk HP, Göker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinformatics 14:60. https://doi.org/10.1186/1471-2105-14-60
Meier-Kolthoff JP, Göker M (2019) TYGS is an automated high-throughput platform for state-of-the-art genome-based taxonomy. Nat Commun 10:2182. https://doi.org/10.1038/s41467-019-10210-3
Mo P, Li KQ, Zhou JH, He J, Zou WS, Gao J (2023) Nocardiopsis changdeensis sp. nov., an endophytic actinomycete isolated from the roots of Eucommia ulmoides Oliv. J Antibiot 76:191–197. https://doi.org/10.1038/s41429-023-00596-0
Mo P, Liu J, Zhao YL, Xu ZG (2020) Streptomyces phaeolivaceus sp. nov. and Streptomyces broussonetiae sp. nov., isolated from the leaves and rhizosphere soil of Broussonetia papyrifera. Int J Syst Evol Microbiol 70:6458–6467. https://doi.org/10.1099/ijsem.0.004556
Mo P, Yu YZ, Zhao JR, Gao J (2017) Streptomyces xiangtanensis sp. nov., isolated from a manganese-contaminated soil. Antonie Van Leeuwenhoek 110:297–304. https://doi.org/10.1007/s10482-016-0797-z
Park DH, Kim JS, Kwon SW, Wilson C, Yu YM, Hur JH, Lim CK (2003) Streptomyces luridiscabiei sp. nov., Streptomyces puniciscabiei sp. nov. and Streptomyces niveiscabiei sp. nov., which cause potato common scab disease in Korea. Int J Syst Evol Microbiol 53:2049–2054. https://doi.org/10.1099/ijs.0.02629-0
Reasoner DJ, Geldreich EE (1985) A new medium for the enumeration and subculture of bacteria from potable water. Appl Environ Microbiol 49:1–7. https://doi.org/10.1128/AEM.49.1.1-7.1985
Richter M, Rosselló-Móra R, Oliver Glöckner F, Peplies J (2016) JSpeciesWS: a web server for prokaryotic species circumscription based on pairwise genome comparison. Bioinformatics 32:929–931. https://doi.org/10.1093/bioinformatics/btv681
Ridgway R (1912) Color standards and color nomenclature. Ridgway, Washington, DC, pp 1–43 ((plate I–LII))
Ruan J, Huang Y (2011) Rapid identification and systematics of actinobacteria. Science press, China, Beijing
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4:406–425. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Scott JJ, Oh DC, Yuceer MC, Klepzig KD, Clardy J, Currie CR (2008) Bacterial protection of beetle-fungus mutualism. Science 322:63. https://doi.org/10.1126/science.1160423
Shirling EB, Gottlieb D (1966) Methods for characterisation of Streptomyces species. Int J Syst Bacteriol 16:313–340. https://doi.org/10.1099/00207713-16-3-313
Stackebrandt E, Ebers J (2006) Taxonomic parameters revisited: tarnished gold standards. Microbiol Today 33:152–155
Tamura K, Stecher G, Kumar S (2021) MEGA11: molecular evolutionary genetics analysis version 11. Mol Biol Evol 38:3022–3027. https://doi.org/10.1093/molbev/msab120
Vincent L, Richard D, Olivier G (2015) FastME 2.0: a comprehensive, accurate, and fast distance-based phylogeny inference program. Mol Biol Evol 32:2798–2800. https://doi.org/10.1093/molbev/msv150
Wayne LG, Brenner DJ, Colwell RR, Grimont PAD, Kandler O, Krichevsky MI et al (1987) International committee on systematic bacteriology. Report of the ad hoc committee on the reconciliation of approaches to bacterial systematics. Int J Syst Bacteriol 37:463–464. https://doi.org/10.1099/00207713-37-4-463
Williams ST, Goodfellow M, Alderson G, Wellington EM, Sneath PH, Sackin MJ (1983) Numerical classification of Streptomyces and related genera. J Gen Microbiol 129:1743–1813. https://doi.org/10.1099/00221287-129-6-1743
Xie Y, Wu G, Tang J, Luo R, Patterson J, Siyuan L et al (2014) SOAPdenovo-trans: denovo transcriptome assembly with short RNA-seq reads. Bioinformatics 30:1660–1666. https://doi.org/10.1093/bioinformatics/btu077
Xu LH, Li WJ, Liu ZH, Jiang CL (2007) Actinomycetes systematics: principles, methods and practices. Science Press, Beijing, China
Yoon SH, Ha SM, Kwon S, Lim J, Kim Y, Seo H, Chun J (2017) Introducing EzBioCloud: a taxonomically united database of 16S rRNA and whole genome assemblies. Int J Syst Evol Microbiol 67:1613–1617. https://doi.org/10.1099/ijsem.0.001755
Zhou DQ (2020) Essential microbiology, 4th edn. Higher Education Press, Beijing
Acknowledgements
We would like to thank Wan Shuang, Xu Wangjie, Zhu Zhisheng, Liu Xiupian and Gu Li for providing help.
Funding
This work was supported by a grant from Scientific Research Fund of Hunan Provincial Education Department (grant number 22A0487 and 23B0659), the Hunan Natural Science Foundation (grant number 2022JJ50249 and 2024JJ7300), the Innovation and Entrepreneurship Training Program for College Students, and the Central Guidance on Local Science and Technology Development Fund of Hunan Province (grant number 2023ZYQ077).
Author information
Authors and Affiliations
Contributions
Conceptualization and methodology: PM, AH D; Software and analysis: YX Z, YP, YZ, XX X, CX L, PM; Validation: PM, AH D; Investigation: YX Z, XX X, CX L, YP, YZ, TT, JL, YR M; Writing-original draft preparation: YX Z, XX X, YP; Supervision: AH D; Project administration: PM, AH D; Funding acquisition: AH D; YX Z, XX X, CX L, YP, YZ, PM, TT, JL, YR M, AH D have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent to participate
Not applicable.
Consent for publication
All authors are consent for publication in Archives of Microbiology.
Additional information
Communicated by Praveen Rahi.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yaxi, Z., Aihua, D., Xiaoxiao, X. et al. Streptomyces camelliae sp. nov., isolated from rhizosphere soil of Camellia oleifera Abel. Arch Microbiol 206, 256 (2024). https://doi.org/10.1007/s00203-024-03987-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-024-03987-5