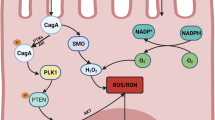
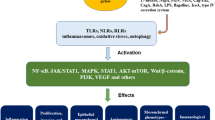
Abstract
Various forms of malignancies have been linked to Helicobacter pylori. Despite advancements in chemotherapeutic and surgical approaches, the management of cancer, particularly at advanced stages, increasingly relies on the integration of immunotherapy. As a novel, safe therapeutic modality, immunotherapy harnesses the immune system of the patient to treat cancer, thereby broadening treatment options. However, there is evidence that H. pylori infection may influence the effectiveness of immunotherapy in various types of cancer. This association is related to H. pylori virulence factors and the tumor microenvironment. This review discusses the influence of H. pylori infection on immunotherapy in non-gastrointestinal and gastrointestinal tumors, the mechanisms underlying this relationship, and directions for the development of improved immunotherapy strategies.


Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Algood HM et al (2006) Helicobacter pylori persistence: an overview of interactions between H. Pylori and host immune defenses. Clin Microbiol Rev 19(4):597–613. https://doi.org/10.1128/CMR.00006-06
Amedei A et al (2006) The neutrophil-activating protein of Helicobacter pylori promotes Th1 immune responses. J Clin Invest 116(4):1092–1101. https://doi.org/10.1172/JCI27177
Arai J et al (2024) Association between Vonoprazan and the risk of gastric cancer after Helicobacter pylori eradication. Clin Gastroenterol Hepatol. https://doi.org/10.1016/j.cgh.2024.01.037
Arnold IC et al (2018) Eosinophils suppress Th1 responses and restrict bacterially induced gastrointestinal inflammation. J Exp Med 215(8):2055–2072. https://doi.org/10.1084/jem.20172049
Bagchi S et al (2021) Immune Checkpoint inhibitors for the treatment of Cancer: clinical impact and mechanisms of response and resistance. Annu Rev Pathol 16:223–249. https://doi.org/10.1146/annurev-pathol-042020-042741
Baj J et al (2020a) Immunological aspects of the Tumor Microenvironment and epithelial-mesenchymal transition in gastric carcinogenesis. Int J Mol Sci 21(7). https://doi.org/10.3390/ijms21072544
Baj J et al (2020b) Helicobacter pylori Virulence Factors-Mechanisms of bacterial pathogenicity in the gastric microenvironment. Cells 10(1). https://doi.org/10.3390/cells10010027
Baud J et al (2013) Helicobacter pylori initiates a mesenchymal transition through ZEB1 in gastric epithelial cells. PLoS ONE 8(4):e60315. https://doi.org/10.1371/journal.pone.0060315
Berge C et al (2017) Structural insights into Helicobacter pylori cag protein interactions with host cell factors. Curr Top Microbiol Immunol 400:129–147. https://doi.org/10.1007/978-3-319-50520-6_6
Bessede E et al (2014) Helicobacter pylori generates cells with cancer stem cell properties via epithelial-mesenchymal transition-like changes. Oncogene 33(32):4123–4131. https://doi.org/10.1038/onc.2013.380
Beswick EJ et al (2007) Expression of the programmed death ligand 1, B7-H1, on gastric epithelial cells after Helicobacter pylori exposure promotes development of CD4 + CD25 + FoxP3 + regulatory T cells. Infect Immun 75(9):4334–4341. https://doi.org/10.1128/IAI.00553-07
Butt J et al (2019) Serologic Response to Helicobacter pylori proteins Associated with Risk of Colorectal Cancer among diverse populations in the United States. Gastroenterology 156(1):175–186. https://doi.org/10.1053/j.gastro.2018.09.054
Cassetta L et al (2011) Macrophage polarization in health and disease. ScientificWorldJournal 11:2391–2402. https://doi.org/10.1100/2011/213962
Chauhan N et al (2019) Helicobacter pylori VacA, a distinct toxin exerts diverse functionalities in numerous cells: an overview. Helicobacter 24(1):e12544. https://doi.org/10.1111/hel.12544
Che Y et al (2018) Helicobacter pylori-induced exosomal MET educates tumour-associated macrophages to promote gastric cancer progression. J Cell Mol Med 22(11):5708–5719. https://doi.org/10.1111/jcmm.13847
Chen F et al (2015) New horizons in tumor microenvironment biology: challenges and opportunities. BMC Med 13. https://doi.org/10.1186/s12916-015-0278-7
Chew V et al (2012) Immune microenvironment in tumor progression: characteristics and challenges for therapy. J Oncol 2012(608406). https://doi.org/10.1155/2012/608406
Choi IJ et al (2020) Family History of Gastric Cancer and Helicobacter pylori Treatment. N Engl J Med 382(5):427–436. https://doi.org/10.1056/NEJMoa1909666
Codolo G et al (2012) HP-NAP inhibits the growth of bladder cancer in mice by activating a cytotoxic Th1 response. Cancer Immunol Immunother 61(1):31–40. https://doi.org/10.1007/s00262-011-1087-2
Codolo G et al (2019) Helicobacter pylori dampens HLA-II expression on macrophages via the Up-Regulation of miRNAs Targeting CIITA. Front Immunol 10:2923. https://doi.org/10.3389/fimmu.2019.02923
Das S et al (2006) Expression of B7-H1 on gastric epithelial cells: its potential role in regulating T cells during Helicobacter pylori infection. J Immunol 176(5):3000–3009. https://doi.org/10.4049/jimmunol.176.5.3000
Deng Y et al (2021) Helicobacter pylori infection disturbs the tumor immune microenvironment and is associated with a discrepant prognosis in gastric de novo diffuse large B-cell lymphoma. J Immunother Cancer 9(10). https://doi.org/10.1136/jitc-2021-002947
Deng R et al (2022) Effects of helicobacter pylori on tumor microenvironment and immunotherapy responses. Front Immunol 13:923477. https://doi.org/10.3389/fimmu.2022.923477
Ding L et al (2020) MiR130b from Schlafen4(+) MDSCs stimulates epithelial proliferation and correlates with preneoplastic changes prior to gastric cancer. Gut 69(10):1750–1761. https://doi.org/10.1136/gutjnl-2019-318817
Duan Z et al (2021) Targeting macrophages in cancer immunotherapy. Signal Transduct Target Ther 6(1). https://doi.org/10.1038/s41392-021-00506-6
El-Zaatari M et al (2013) Gli1 deletion prevents Helicobacter-induced gastric metaplasia and expansion of myeloid cell subsets. PLoS ONE 8(3):e58935. https://doi.org/10.1371/journal.pone.0058935
Elkrief A et al (2019) The negative impact of antibiotics on outcomes in cancer patients treated with immunotherapy: a new independent prognostic factor? Ann Oncol 30(10):1572–1579. https://doi.org/10.1093/annonc/mdz206
Fuccio L et al (2009) Meta-analysis: can Helicobacter pylori eradication treatment reduce the risk for gastric cancer? Ann Intern Med 151(2):121–128. https://doi.org/10.7326/0003-4819-151-2-200907210-00009
Gambardella V et al (2020) The role of tumor-associated macrophages in gastric cancer development and their potential as a therapeutic target. Cancer Treat Rev 86:102015. https://doi.org/10.1016/j.ctrv.2020.102015
Gobert AP et al (2001) Helicobacter pylori arginase inhibits nitric oxide production by eukaryotic cells: a strategy for bacterial survival. Proc Natl Acad Sci U S A 98(24):13844–13849. https://doi.org/10.1073/pnas.241443798
Gobert AP et al (2002) Helicobacter pylori induces macrophage apoptosis by activation of arginase II. J Immunol 168(9):4692–4700. https://doi.org/10.4049/jimmunol.168.9.4692
Gobert AP et al (2014) Heme oxygenase-1 dysregulates macrophage polarization and the immune response to Helicobacter pylori. J Immunol 193(6):3013–3022. https://doi.org/10.4049/jimmunol.1401075
Gopalakrishnan V et al (2018a) The influence of the gut microbiome on Cancer, immunity, and Cancer Immunotherapy. Cancer Cell 33(4):570–580. https://doi.org/10.1016/j.ccell.2018.03.015
Gopalakrishnan V et al (2018b) Gut microbiome modulates response to anti-PD-1 immunotherapy in melanoma patients. Science 359(6371):97–103. https://doi.org/10.1126/science.aan4236
Gur C et al (2019) The Helicobacter pylori HopQ outermembrane protein inhibits immune cell activities. Oncoimmunology 8(4). https://doi.org/10.1080/2162402X.2018.1553487
Han Z et al (2023) Helicobacter pylori, gastrointestinal microbiota, and immunotherapy. Helicobacter 28(1):e12942. https://doi.org/10.1111/hel.12942
Hardbower DM et al (2016) Arginase 2 deletion leads to enhanced M1 macrophage activation and upregulated polyamine metabolism in response to Helicobacter pylori infection. Amino Acids 48(10):2375–2388. https://doi.org/10.1007/s00726-016-2231-2
Helicobacter et al (2001) Gastric cancer and Helicobacter pylori: a combined analysis of 12 case control studies nested within prospective cohorts. Gut 49(3):347–353. https://doi.org/10.1136/gut.49.3.347
Hooi JKY et al (2017) Global prevalence of Helicobacter pylori infection: systematic review and Meta-analysis. Gastroenterology 153(2):420–429. https://doi.org/10.1053/j.gastro.2017.04.022
Horvat A et al (2018) Helicobacter pylori pathogen regulates p14ARF tumor suppressor and autophagy in gastric epithelial cells. Oncogene 37(37):5054–5065. https://doi.org/10.1038/s41388-018-0343-8
Hou M et al (2020) TLR agonist rHP-NAP as an adjuvant of dendritic cell-based vaccine to Enhance Anti-melanoma Response. Iran J Immunol 17(1):14–25. https://doi.org/10.22034/iji.2020.80291
Hu Y et al (2019) Downregulation of tumor suppressor RACK1 by Helicobacter pylori infection promotes gastric carcinogenesis through the integrin beta-1/NF-kappaB signaling pathway. Cancer Lett 450:144–154. https://doi.org/10.1016/j.canlet.2019.02.039
Huang L et al (2019) Penicillin–binding protein 1A mutation–positive Helicobacter pylori promotes epithelial–mesenchymal transition in gastric cancer via the suppression of microRNA–134. Int J Oncol 54(3):916–928. https://doi.org/10.3892/ijo.2018.4665
Huang Y et al (2022) The molecular mechanisms and therapeutic strategies of EMT in tumor progression and metastasis. J Hematol Oncol 15(1):129. https://doi.org/10.1186/s13045-022-01347-8
Jia Z et al (2022) Positive H. pylori status predicts better prognosis of non-cardiac gastric cancer patients: results from cohort study and meta-analysis. BMC Cancer 22(1). https://doi.org/10.1186/s12885-022-09222-y
Jin C et al (2022) CAR T cells expressing a bacterial virulence factor trigger potent bystander antitumour responses in solid cancers. Nat Biomed Eng 6(7):830–841. https://doi.org/10.1038/s41551-022-00875-5
Kim W et al (2021) PD-1 signaling promotes tumor-infiltrating myeloid-derived suppressor cells and gastric tumorigenesis in mice. Gastroenterology 160(3):781–796. https://doi.org/10.1053/j.gastro.2020.10.036
Koch MRA et al (2023) CagA-specific gastric CD8(+) tissue-resident T cells control Helicobacter pylori during the early infection phase. Gastroenterology 164(4):550–566. https://doi.org/10.1053/j.gastro.2022.12.016
Krzysiek-Maczka G et al (2019) Involvement of epithelial-mesenchymal transition-inducing transcription factors in the mechanism of Helicobacter pylori-induced fibroblasts activation. J Physiol Pharmacol 70(5). https://doi.org/10.26402/jpp.2019.5.08
Kusters JG et al (2006) Pathogenesis of Helicobacter pylori infection. Clin Microbiol Rev 19(3):449–490. https://doi.org/10.1128/CMR.00054-05
Lee DG et al (2014) Helicobacter pylori CagA promotes snail-mediated epithelial-mesenchymal transition by reducing GSK-3 activity. Nat Commun 5:4423. https://doi.org/10.1038/ncomms5423
Li N et al (2018) Helicobacter pylori CagA promotes epithelial mesenchymal transition in gastric carcinogenesis via triggering oncogenic YAP pathway. J Exp Clin Cancer Res 37(1):280. https://doi.org/10.1186/s13046-018-0962-5
Lin L et al (2019) Chronic CagA-positive Helicobacter pylori infection with MNNG stimulation synergistically induces mesenchymal and cancer stem cell-like properties in gastric mucosal epithelial cells. J Cell Biochem 120(10):17635–17649. https://doi.org/10.1002/jcb.29031
Lina TT et al (2015) Helicobacter pylori cag pathogenicity island’s role in B7-H1 induction and immune evasion. PLoS ONE 10(3). https://doi.org/10.1371/journal.pone.0121841
Liu M et al (2018) Recent updates on cancer immunotherapy. Precis Clin Med 1(2):65–74. https://doi.org/10.1093/pcmedi/pby011
Magahis PT et al (2023) Impact of Helicobacter pylori infection status on outcomes among patients with advanced gastric cancer treated with immune checkpoint inhibitors. J Immunother Cancer 11(10). https://doi.org/10.1136/jitc-2023-007699
Malfertheiner P et al (2023) Helicobacter pylori infection. Nat Rev Dis Primers 9(1). https://doi.org/10.1038/s41572-023-00431-8
MaruYama T et al (2022) TGF-beta and Cancer immunotherapy. Biol Pharm Bull 45(2):155–161. https://doi.org/10.1248/bpb.b21-00966
Mejias-Luque R et al (2017) Immune evasion strategies and persistence of Helicobacter pylori. Curr Top Microbiol Immunol 400:53–71. https://doi.org/10.1007/978-3-319-50520-6_3
Nishizuka SS et al (2023) Anti-helicobacter Pylori antibody status is associated with cancer mortality: a longitudinal analysis from the Japanese DAIKO prospective cohort study. PLOS Glob Public Health 3(2):e0001125. https://doi.org/10.1371/journal.pgph.0001125
Noori M et al (2023) Impact of Helicobacter pylori infection on the efficacy of immune checkpoint inhibitors for cancer treatment: a meta-analysis. Immunotherapy 15(9):657–667. https://doi.org/10.2217/imt-2022-0250
Oster P et al (2022a) The efficacy of Cancer Immunotherapies is compromised by Helicobacter pylori Infection. Front Immunol 13:899161. https://doi.org/10.3389/fimmu.2022.899161
Oster P et al (2022b) Helicobacter pylori infection has a detrimental impact on the efficacy of cancer immunotherapies. Gut 71(3):457–466. https://doi.org/10.1136/gutjnl-2020-323392
Pan Y et al (2020) Tumor-Associated macrophages in Tumor Immunity. Front Immunol 11. https://doi.org/10.3389/fimmu.2020.583084
Panagioti E et al (2021) Immunostimulatory bacterial antigen-armed oncolytic measles virotherapy significantly increases the potency of anti-PD1 checkpoint therapy. J Clin Invest 131(13). https://doi.org/10.1172/JCI141614
Qin BD et al (2021) Effects of concomitant proton pump inhibitor use on immune checkpoint inhibitor efficacy among patients with advanced cancer. Oncoimmunology 10(1). https://doi.org/10.1080/2162402X.2021.1929727
Queiroz DM et al (2011) dupA polymorphisms and risk of Helicobacter pylori-associated diseases. Int J Med Microbiol 301(3):225–228. https://doi.org/10.1016/j.ijmm.2010.08.019
Rahimian G et al (2014) Virulence factors of Helicobacter pylori vacA increase markedly gastric mucosal TGF-beta1 mRNA expression in gastritis patients. Microb Pathog 67–68. https://doi.org/10.1016/j.micpath.2013.12.006
Ralser A et al (2023) Helicobacter pylori promotes colorectal carcinogenesis by deregulating intestinal immunity and inducing a mucus-degrading microbiota signature. Gut 72(7):1258–1270. https://doi.org/10.1136/gutjnl-2022-328075
Reyes VE et al (2019) Helicobacter pylori deregulates T and B cell signaling to trigger Immune Evasion. Curr Top Microbiol Immunol 421:229–265. https://doi.org/10.1007/978-3-030-15138-6_10
Routy B et al (2018) Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science 359(6371):91–97. https://doi.org/10.1126/science.aan3706
Sarajlic M et al (2020) H. Pylori modulates DC functions via T4SS/TNFalpha/p38-dependent SOCS3 expression. Cell Commun Signal 18(1):160. https://doi.org/10.1186/s12964-020-00655-1
Seeneevassen L et al (2021) Gastric Cancer: advances in Carcinogenesis Research and New Therapeutic Strategies. Int J Mol Sci 22(7). https://doi.org/10.3390/ijms22073418
Shi Y et al (2022) Effect of Helicobacter pylori on immunotherapy is gaining more attention. Helicobacter , 27(5), e12925. https://doi.org/10.1111/hel.12925
Shiota S et al (2010) Systematic review and meta-analysis: the relationship between the Helicobacter pylori dupA gene and clinical outcomes. Gut Pathog 2(1):13. https://doi.org/10.1186/1757-4749-2-13
Song L et al (2021) Helicobacter pylori Immunoproteomic profiles in gastric Cancer. J Proteome Res 20(1):409–419. https://doi.org/10.1021/acs.jproteome.0c00466
Suzuki S et al (2019) Development of Helicobacter pylori treatment: how do we manage antimicrobial resistance? World J Gastroenterol 25(16):1907–1912. https://doi.org/10.3748/wjg.v25.i16.1907
Talebi Bezmin Abadi A et al (2016) Role of dupA in virulence of Helicobacter pylori. World J Gastroenterol 22(46):10118–10123. https://doi.org/10.3748/wjg.v22.i46.10118
Talebi Bezmin Abadi A et al (2018) Helicobacter pylori therapy and clinical perspective. J Glob Antimicrob Resist 14:111–117. https://doi.org/10.1016/j.jgar.2018.03.005
Tegtmeyer N et al (2017) Subversion of host kinases: a key network in cellular signaling hijacked by Helicobacter pylori CagA. Mol Microbiol 105(3):358–372. https://doi.org/10.1111/mmi.13707
Teymournejad O et al (2014) In vitro suppression of dendritic cells by Helicobacter pylori OipA. Helicobacter 19(2):136–143. https://doi.org/10.1111/hel.12107
Tonneau M et al (2022) Helicobacter pylori serology is associated with worse overall survival in patients with melanoma treated with immune checkpoint inhibitors. Oncoimmunology 11(1). https://doi.org/10.1080/2162402X.2022.2096535
Vetizou M et al (2015) Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science 350(6264):1079–1084. https://doi.org/10.1126/science.aad1329
Wada A et al (2004) Helicobacter pylori vacuolating cytotoxin, VacA, is responsible for gastric ulceration. J Biochem 136(6):741–746. https://doi.org/10.1093/jb/mvh181
Wang T et al (2015) Antitumor and immunomodulatory effects of recombinant fusion protein rMBP-NAP through TLR-2 dependent mechanism in tumor bearing mice. Int Immunopharmacol 29(2):876–883. https://doi.org/10.1016/j.intimp.2015.08.027
Wang PF et al (2018) Prognostic role of pretreatment circulating MDSCs in patients with solid malignancies: a meta-analysis of 40 studies. Oncoimmunology 7(10). https://doi.org/10.1080/2162402X.2018.1494113
Wang M et al (2023) The role of the gut microbiota in gastric cancer: the immunoregulation and immunotherapy. Front Immunol 14:1183331. https://doi.org/10.3389/fimmu.2023.1183331
Wroblewski LE et al (2013) Helicobacter pylori in gastric carcinogenesis: mechanisms. Gastroenterol Clin North Am 42(2):285–298. https://doi.org/10.1016/j.gtc.2013.01.006
Wu YY et al (2010) Increased programmed death-ligand-1 expression in human gastric epithelial cells in Helicobacter pylori infection. Clin Exp Immunol 161(3):551–559. https://doi.org/10.1111/j.1365-2249.2010.04217.x
Wu Q et al (2013) Association between Helicobacter pylori infection and the risk of colorectal neoplasia: a systematic review and meta-analysis. Colorectal Dis 15(7):e352–e364. https://doi.org/10.1111/codi.12284
Wustner S et al (2015) Helicobacter pylori gamma-glutamyltranspeptidase impairs T-lymphocyte function by compromising metabolic adaption through inhibition of cMyc and IRF4 expression. Cell Microbiol 17(1):51–61. https://doi.org/10.1111/cmi.12335
Xiang X et al (2021) Plasmacytoid dendritic cell-derived type I Interferon is involved in Helicobacter pylori infection-Induced differentiation of Schlafen 4-Expressing myeloid-derived suppressor cells. Infect Immun 89(11):e0040721. https://doi.org/10.1128/IAI.00407-21
Yan L et al (2022) Effect of Helicobacter pylori Eradication on Gastric Cancer Prevention: updated Report from a Randomized Controlled Trial with 26.5 years of follow-up. Gastroenterology 163(1):154–162. https://doi.org/10.1053/j.gastro.2022.03.039
Yu H et al (2014) Helicobacter pylori promotes epithelial-mesenchymal transition in gastric cancer by downregulating programmed cell death protein 4 (PDCD4). PLoS ONE 9(8). https://doi.org/10.1371/journal.pone.0105306
Zabaleta J et al (2004) Helicobacter pylori arginase inhibits T cell proliferation and reduces the expression of the TCR zeta-chain (CD3zeta). J Immunol 173(1):586–593. https://doi.org/10.4049/jimmunol.173.1.586
Zhang X et al (2013) Structural, enzymatic and biochemical studies on Helicobacter pylori arginase. Int J Biochem Cell Biol 45(5):995–1002. https://doi.org/10.1016/j.biocel.2013.02.008
Zhang J et al (2014) Outer membrane inflammatory protein A, a new virulence factor involved in the pathogenesis of Helicobacter pylori. Mol Biol Rep 41(12):7807–7814. https://doi.org/10.1007/s11033-014-3673-9
Zhang Y et al (2020) The history and advances in cancer immunotherapy: understanding the characteristics of tumor-infiltrating immune cells and their therapeutic implications. Cell Mol Immunol 17(8):807–821. https://doi.org/10.1038/s41423-020-0488-6
Zhang MJ et al (2021a) Helicobacter pylori infection as a potential favorable factor for immune checkpoint inhibitor therapy for gastric cancer. Invest New Drugs 39(5):1436–1438. https://doi.org/10.1007/s10637-021-01122-5
Zhang Z et al (2021b) Helicobacter pylori induces gastric cancer via down-regulating miR-375 to inhibit dendritic cell maturation. Helicobacter 26(4). https://doi.org/10.1111/hel.12813
Zhao Y et al (2016) Helicobacter pylori infection and colorectal carcinoma risk: a meta-analysis. J Cancer Res Ther 12(Supplement):15–18. https://doi.org/10.4103/0973-1482.191621
Zhou J et al (2022) The impact of antibiotic use on clinical features and survival outcomes of cancer patients treated with immune checkpoint inhibitors. Front Immunol 13:968729. https://doi.org/10.3389/fimmu.2022.968729
Funding
This work was supported by Jiangsu Postgraduate Practice Innovation Program (Grant numbers SJCX23-1408); Translational Medicine Innovative Talent Program, North Jiangsu Clinical Research Institute, Nanjing Medical University (Grant numbers YZHR201904).
Author information
Authors and Affiliations
Contributions
Junyi Zhou and Minna Zhang wrote and prepared the manuscript. Honggang Wang, Xiaomin Zhong and Xiaozhong Yang reviewed the manuscript. All authors contributed to the article submission and agreed to the published version of manuscript.
Corresponding authors
Ethics declarations
Ethical approval
Not applicable.
Consent for publication
Not applicable.
Consent to participate
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Yusuf Akhter.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhou, J., Zhang, M., Wang, H. et al. Role of Helicobacter pylori virulence factors and alteration of the Tumor Immune Microenvironment: challenges and opportunities for Cancer Immunotherapy. Arch Microbiol 206, 167 (2024). https://doi.org/10.1007/s00203-024-03908-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-024-03908-6