Abstract
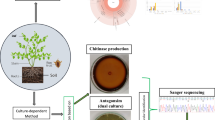
Duckweed has been highlighted as an invaluable resource because of its abilities to remove nitrogen and phosphorus from wastewater coupling with the production of high starch/protein-containing plant biomass. Duckweed recruits microbes and particularly forms a stable “core” bacterial microbiota, which greatly reduces the colonization efficiency of plant growth-promoting bacteria (PGPB). In this study, natural duckweeds were enriched in a sterilized-partially treated wastewater effluent from a poultry farm. After 24 days of cultivation, the duckweed-associated bacteria (DAB) were isolated and evaluated for their plant growth-promoting (PGP) potentials by co-cultivation with axenic Spirodela polyrhiza. Ten species were found in more than one location and could be considered candidates for the stable “core” DAB. Among them, all isolates of Acinetobacter soli, Acidovorax kalamii, Brevundimonas vesicularis, Pseudomonas toyotomiensis, and Shinella curvata increased duckweed growth in Hoagland medium. The highest PGP ability was observed in Sh. curvata W12-8 (with EPG value of 208.72%), followed by Paracoccus marcusii W7-16 (171.31%), Novosphingobium subterraneum W5-13 (156.96%), and Ac. kalamii W7-18 (156.96%). However, the highest growth promotion in the wastewater was observed when co-cultured with W7-16, which was able to increase biomass dry weight and root length of duckweed by 3.17 and 2.26 folds, respectively.








Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the article and its supplementary material.
References
Acosta K, Xu J, Gilbert S et al (2020) Duckweed hosts a taxonomically similar bacterial assemblage as the terrestrial leaf microbiome. PLoS ONE 15(2):e0228560. https://doi.org/10.1371/journal.pone.0228560
Al-Dakhil M, Ben Romdhane W, Alghamdi S, Ali AAM (2023) Differential morpho-physiological and biochemical responses of duckweed clones from saudi arabia to salinity. Plants (basel) 12(18):3206. https://doi.org/10.3390/plants12183206
Aleksandrov VG, (1958) Organo-mineral fertilizers and silicate bacteria. Dokl Akad-S.Kh Nauk 7: 43–48.
Appenroth KJ (2015) Useful Methods 2: Sterilization of Duckweed. Duckweed Forum 3(9):90–91
Appenroth KJ, Sree KS, Bog M, Ecker J, Boehm V, Lorkowski S et al (2018) Nutritional value of the duckweed species of the genus Wolffia (Lemnaceae) as human food. Front Chem 6:483. https://doi.org/10.3389/fchem.2018.00483
Bulgarelli D, Schlaeppi K, Spaepen S, Loren V, van Themaat E, Schulze-Lefert P (2013) Structure and functions of the bacterial microbiota of plants. Annu Rev Plant Biol 64:807–838. https://doi.org/10.1146/annurev-arplant-050312-120106
Bunyoo C, Roongsattham P, Khumwan S, Phonmakham J, Wonnapinij P, Thamchaipenet A (2022) Dynamic alteration of microbial communities of duckweeds from nature to nutrient-deficient condition. Plants (basel) 11(21):2915. https://doi.org/10.3390/plants11212915
Cheng JJ, Stomp AM (2009) Growing duckweed to recover nutrients from wastewaters and for production of fuel ethanol and animal feed. CLEAN - Soil Air Water 37(1):17–26. https://doi.org/10.1002/clen.200800210
Conradie TA, Pieterse E, Jacobs K (2018) Application of Paracoccus marcusii as a potential feed additive for laying hens. Poult Sci 97(3):986–994. https://doi.org/10.3382/ps/pex377
Haasdijk RA, van Ingen J (2017) Escherichia hermannii as the sole pathogen in urosepsis: case report. New Microbes New Infect 21:100–101. https://doi.org/10.1016/j.nmni.2017.11.005
Ioannou P (2019) Escherichia hermannii infections in humans: a systematic review. Trop Med Infect Dis 4(1):17. https://doi.org/10.3390/tropicalmed4010017
Ishizawa H, Kuroda M, Morikawa M, Ike M (2017) Evaluation of environmental bacterial communities as a factor affecting the growth of duckweed Lemna minor. Biotechnol Biofuels 10:62. https://doi.org/10.1186/s13068-017-0746-8
Ishizawa H, Kuroda M, Inoue D, Morikawa M, Ike M (2020a) Community dynamics of duckweed-associated bacteria upon inoculation of plant growth-promoting bacteria. FEMS Microbiol Ecol 96(7):101. https://doi.org/10.1093/femsec/fiaa101
Ishizawa H, Ogata Y, Hachiya Y et al (2020b) Enhanced biomass production and nutrient removal capacity of duckweed via two-step cultivation process with a plant growth-promoting bacterium, Acinetobacter calcoaceticus P23. Chemosphere 238:124682. https://doi.org/10.1016/j.chemosphere.2019.124682
Kajadpai N, Angchuan J, Khunnamwong P, Srisuk N (2023) Diversity of duckweed (Lemnaceae) associated yeasts and their plant growth promoting characteristics. AIMS Microbiol 9(3):486–517. https://doi.org/10.3934/microbiol.2023026
Khairina Y, Jog R, Boonmak C, Toyama T, Oyama T, Morikawa M (2021) Indigenous bacteria, an excellent reservoir of functional plant growth promoters for enhancing duckweed biomass yield on site. Chemosphere 268:129247. https://doi.org/10.1016/j.chemosphere.2020.129247
Kitanaka H, Sasano MA, Yokoyama S, Suzuki M, Jin W, Inayoshi M, Hori M, Wachino J, Kimura K, Yamada K, Arakawa Y (2014) Invasive infection caused by carbapenem-resistant Acinetobacter soli. Japan Emerg Infect Dis 9:1574–1576. https://doi.org/10.3201/eid2009.140117
Kristanti RA, Kanbe M, Hadibarata T, Toyama T, Tanaka Y, Mori K (2012) Isolation and characterization of 3-nitrophenol-degrading bacteria associated with rhizosphere of Spirodela polyrrhiza. Environ Sci Pollut Res Int 19(5):1852–1858. https://doi.org/10.1007/s11356-012-0836-x
Lam E, Acosta K (2019) Useful methods: Cefotaxime: a useful antibiotic for duckweed culture management. Duckweed Forum 7(25):52–54
Leeprasert L, Chonudomkul D, Boonmak C (2022) Biocalcifying potential of ureolytic bacteria isolated from soil for biocementation and material crack repair. Microorganisms 10(5):963. https://doi.org/10.3390/microorganisms10050963
Leinberger J, Holste J, Bunk B et al (2021) High Potential for secondary metabolite production of Paracoccus marcusii CP157, isolated from the crustacean Cancer pagurus. Front Microbiol 12:688754. https://doi.org/10.3389/fmicb.2021.688754
Lu B, Wang B, Pan X et al (2023) First case of bloodstream infection caused by NDM-positive Escherichia hermannii. BMC Infect Dis 23:355. https://doi.org/10.1186/s12879-023-08336-3
Massa F, Defez R, Bianco C (2022) Exploitation of plant growth promoting bacteria for sustainable agriculture: hierarchical approach to link laboratory and field experiments. Microorganisms 10(5):865. https://doi.org/10.3390/microorganisms10050865
Matsuzawa H, Tanaka Y, Tamaki H, Kamagata Y, Mori K (2010) Culture-dependent and independent analyses of the microbial communities inhabiting the giant duckweed (Spirodela polyrhiza) rhizoplane and isolation of a variety of rarely cultivated organisms within the phylum Verrucomicrobia. Microbes Environ 25(4):302–308. https://doi.org/10.1264/jsme2.me10144
Naik R, Gupte S (2023) Characterization of pigment produced by high carotenoid yielding bacteria Paracoccus marcusii RSPO1 and evaluation of its anti-diabetic, anti-microbial and antioxidant properties. Nat Prod Res. https://doi.org/10.1080/14786419.2023.2208358
Parnell JJ, Berka R, Young HA, Sturino JM, Kang Y, Barnhart DM, DiLeo MV (2016) From the lab to the farm: an industrial perspective of plant beneficial microorganisms. Front Plant Sci 7:1110. https://doi.org/10.3389/fpls.2016.01110
Pellegrino FL, Vieira VV, Baio PV, dos Santos RM, dos Santos AL, Santos NG, Meohas MM, Santos RT, de Souza TC, da Silva Dias RC, Santoro-Lopes G, Riley LW, Moreira BM (2011) Acinetobacter soli as a cause of bloodstream infection in a neonatal intensive care unit. J Clin Microbiol 49(6):2283–2285. https://doi.org/10.1128/JCM.00326-11
Pikovskaya RI (1948) Mobilization of phosphorus in soil in connection with vital activity of some microbial species. Mikrobiologiya 17:362–370
Radić S, Stipaničev D, Cvjetko P et al (2011) Duckweed Lemna minor as a tool for testing toxicity and genotoxicity of surface waters. Ecotoxicol Environ Saf 74(2):182–187. https://doi.org/10.1016/j.ecoenv.2010.06.011
Schenk RU, Hildebrandt AC (1972) Medium and techniques for induction and growth of monocotyledonous and dicotyledonous plant cell cultures. Canad J Bot 50(1):199–204. https://doi.org/10.1139/b72-026
Schwyn B, Neilands JB (1987) Universal chemical assay for the detection and determination of siderophores. Anal Biochem 160(1):47–56. https://doi.org/10.1016/0003-2697(87)90612-9
Soda S, Ohchi T, Piradee J, Takai Y, Ike M (2015) Duckweed biomass as a renewable biorefinery feedstock: Ethanol and succinate production from Wolffia globosa. Biomass Bioenerg 81:364–368. https://doi.org/10.1016/j.biombioe.2015.07.020
Stout L, Nüsslein K (2010) Biotechnological potential of aquatic plant-microbe interactions. Curr Opin Biotechnol 21(3):339–345. https://doi.org/10.1016/j.copbio.2010.04.004
Suzuki W, Sugawara M, Miwa K, Morikawa M (2014) Plant growth-promoting bacterium Acinetobacter calcoaceticus P23 increases the chlorophyll content of the monocot Lemna minor (duckweed) and the dicot Lactuca sativa (lettuce). J Biosci Bioeng 118(1):41–44. https://doi.org/10.1016/j.jbiosc.2013.12.007
Toyama T, Yu N, Kumada H, Sei K, Ike M, Fujita M (2006) Accelerated aromatic compounds degradation in aquatic environment by use of interaction between Spirodela polyrrhiza and bacteria in its rhizosphere. J Biosci Bioeng 101(4):346–353. https://doi.org/10.1263/jbb.101.346
Toyama T, Hanaoka T, Tanaka Y, Morikawa M, Mori K (2018) Comprehensive evaluation of nitrogen removal rate and biomass, ethanol, and methane production yields by combination of four major duckweeds and three types of wastewater effluent. Bioresour Technol 250:464–473. https://doi.org/10.1016/j.biortech.2017.11.054
Toyama T, Mori K, Tanaka Y, Ike M, Morikawa M (2022) Growth promotion of giant duckweed Spirodela polyrhiza (Lemnaceae) by Ensifer sp. SP4 through enhancement of nitrogen metabolism and photosynthesis. Mol Plant Microbe Interact 35(1):28–38. https://doi.org/10.1094/MPMI-06-21-0157-R
Yamaga F, Washio K, Morikawa M (2010) Sustainable biodegradation of phenol by Acinetobacter calcoaceticus P23 isolated from the rhizosphere of duckweed Lemna aoukikusa. Environ Sci Technol 44(16):6470–6474. https://doi.org/10.1021/es1007017
Yamakawa Y, Jog R, Morikawa M (2018) Effects of co-inoculation of two different plant growth-promoting bacteria on duckweed. Plant Growth Regul 86:287–296. https://doi.org/10.1007/s10725-018-0428-y
Yoneda Y, Yamamoto K, Makino A, Tanaka Y, Meng X-Y, Hashimoto J, Shin-ya K, Satoh N, Fujie M, Toyama T, et al. (2021) Novel plant-associated acidobacteria promotes growth of common floating aquatic plants, duckweeds. Microorgan 9(6):1133. https://doi.org/10.3390/microorganisms9061133
Zhou Y, Stepanenko A, Kishchenko O, Xu J, Borisjuk N (2023) Duckweeds for phytoremediation of polluted water. Plants (basel) 12(3):589. https://doi.org/10.3390/plants12030589
Funding
This work was funded by the Kasetsart University Research and Development Institute (KURDI) under the project FF(KU)4.64 “Livestock wastewater treatment and biomass production of duckweed using duckweed growth promoting-associated microbes”.
Author information
Authors and Affiliations
Contributions
CB provided conceptualization, funding acquisition, project administration, resources, supervision, data curation, formal analysis, investigation, methodology, writing—original draft, and editing. SK performed the data curation and investigation. BB performed the data curation and investigation. MM provided project administration and resources. KD provided project administration and resources. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Communicated by Yusuf Akhter.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Boonmak, C., Kettongruang, S., Buranathong, B. et al. Duckweed-associated bacteria as plant growth-promotor to enhance growth of Spirodela polyrhiza in wastewater effluent from a poultry farm. Arch Microbiol 206, 43 (2024). https://doi.org/10.1007/s00203-023-03778-4
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03778-4